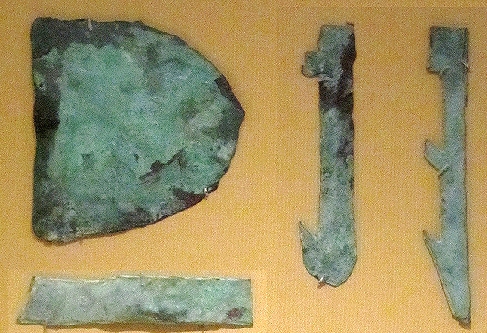|
|
| |
Mythical Concepts of Human Ages |
 |
There are many attempts to bring order into human history by defining "ages"
or periods that have some common denominator. It goes without saying that whoever made that attempt was biased by his or
her own culture and history. That's why I and everybody else from the "western" world have usually only heard
about "ages of man" as defined by western guys and by western thought.
As
usual (in Western culture), one starts with the ancient Greeks. |
|
 |
Hesiod, a Greek poet writing around
750 BC to 650 BC, came up with 5 ages:
- Golden Age. People and Gods lived together in harmony, nobody had to work,
and peace prevailed.
Hesiod obviously was a writer of "fantasy" stuff.
- Silver Age. Men in the Silver age lived for one hundred years under the dominion
of their mothers and not quite as peacefully and harmonious as in the golden age.
As much as I love my mother, I tend
to agree. 100 years?! Can't be all that harmonious and peaceful.
- Bronze Age. Things are more modern. War was common, arms, tools and even houses
were forged of bronze. People after death went to "Hades", a kind of hell.
- Heroic Age. Things improved partially. Demigods and heroes did noble deeds.
Humans died and went on occasion to "Elysium", a kind of paradise.
- Iron Age. The age Hesiod lived in. Complete disaster, everything goes down the
drain.
A bit like modern Greece today (2012).
In other words: Things were always better in the past, also known as "the good old times". This viewpoint
wasn't new in 600 BC. You may laugh at this but Hesiod's "ages" was
influential to the way people perceived themselves and to the arts for a long time. |
 |
After the old Greeks we look to the old Romans.
Ovid, a Roman poet who lived from 43 BC – 17/18 AD, adapted
Hesiod's system but omitted the heroic age. |
|
 |
He also ties the remaining four ages a bit more to the development of civilization and technology.
Like all elderly persons he shared Hesiod's opinion that things became worse as he grew older.
|
 |
The early Christians certainly had
to say something about this too. |
|
 |
Saint Jerome, formerly Saint Hierom (347 – 420), the
guy who translated "the Bible" into Latin (the Vulgate), quite full
of himself and given to accuracy, gave precise dates on Hesiod's ages:
- Golden Age: 1710 to 1674 BC
- Silver Age: 1674 to 1628 BC
- Bronze Age: 1628 to 1472 BC
- Heroic Age: 1460 to 1103 BC
- Iron Age: 1103 BC, still going on
Makes you wonder how believable his other stuff will be. Of course, the less you actually know about what you are
writing, the more you tend to give numbers with many digits and thus supposed precision. Saint Jerome started a time-honored
tradition here that is still alive and thriving. |
 |
Saint Augustine
(354 - 430) begged to differ. He conceived Six Ages that actually had some roots in the Jewish tradition.
|
|
 |
His ideas became central to the church. Augustine's six ages of history, with each age lasting
approximately 1000 years, were widely believed to be factual and thus dominated the writing of history in the Middle Ages.
Here they are:
- First Age: from the beginning of the human race; i.e. from Adam, down to
Noah.
- Second Age: from Noah to Abraham.
- Third Age: from Abraham to King David.
- Fourth Age: from David to the captivity of the Jews in Babylonia.
- Fifth Age: from Babylonia to the advent of Jesus Christ.
- Sixth Age: coming of Christ to now.
Tough luck that the sixth age lasts already more than 2.000 years. |
|
 |
What we learn from this is that medieval history, including art and sword lore, is often deeply
rooted in wishful thinking or plain nonsense. One should bear that in mind when contemplating old
stories about iron, steel and swords. |
 |
One can safely assume that other cultures / religions had their own "ages",
and that they weren't doing much better. Indeed, as Wikipedia knows, old Indian writings also refer to ages linked to major
Deities and metals: Satya (Golden), Treta (Silver), Dwapara (Bronze) and Kali (Iron). |
|
 |
In stark contrast to Augustine's 1000 years per age, the Indians allow a grant
total of 4.32 million years for the four ages in the Hindu-Vedic line. |
 |
Interestingly, as far as metals are associated with ages, there seems to be a
general agreement that a golden age is the best, and an iron age is the worst. |
|
 |
Considering that gold is utterly useless
except for displaying wealth and power, while iron is extremely useful to all of us
who must work for a living, it becomes clear what kind of people came up with those "human age" concepts. Not
the kind like you and me that had to work for a living, but the kind who took away the fruits of our (and our forebears)
labor in exchange for letting us live.
Once more it is important to bear in mind that history in general was written
by the winners. Be it the winners in a war or just in the race for social settings, ranks or class. |
| |
|
|
Scientific Concepts of Human Ages |
 |
Just to be clear: the scientific concept of
Human Ages is not all that scientific either. The idea was to classify general human development by the progress made in
material development for tools.
In reality, the concept of Human Ages served mainly as a battle ground for various
budding scientists in the 19th century. Untold pages to the topic can be found in the Net. Take this quote from the very
entertaining wikipedia page "Three-age system": |
|
 |
"In 1874 at the Stockholm meeting of the International Congress of Anthropology
and Prehistoric Archaeology, a suggestion was made by A. Bertrand that no distinct age of bronze had existed, that the bronze
artifacts discovered were really part of the Iron Age. Hans Hildebrand in refutation pointed to two Bronze Ages and a transitional
period in Scandinavia. John Evans denied any defect of continuity between the two and asserted there were three Bronze Ages,
"the early, middle and late bronze age." |
|
 |
Here is another quote from that site that we will need later on:
"In his 1865 book, Prehistoric Times, Lubbock divided the Stone Age in Europe, and possibly nearer Asia and Africa,
into the Palaeolithic and the Neolithic:
I That of the Drift... This we may call the 'Palaeolithic' Period.
II.
The later, or polished Stone Age ... in which, however, we find no trace ... of any metal, excepting
gold, ... This we may call the 'Neolithic' Period.
III. The Bronze Age, in which bronze was used for arms and
cutting instruments of all kinds.
IV. The Iron Age, in which that metal had superseded bronze." |
|
 |
Scientists do love a good controversy, especially if it makes no sense to anybody else. Witness:
"The European definition of the Iron Age as distinguished from the Bronze age is still not all that clear, yet it
is a marvel of clarity compared to that of the Middle East. There, the first stage of what they call the Iron Age is defined
by the absence of iron! They obviously don't know their three age system out there, or, for that matter, over in the US
...". Writes one Hernik Thrane in the Opening Lecture to the 1999
Sandbjerg Conference in Denmark |
 |
What survived, after long and often completely pointless and bitter debates, and
what I learned in school, are the following "Ages": - Stone age
- Bronze age
- Iron age
There is now also a "copper age" but still no gold and/or silver age, although these metals were of some
importance (to the rich and powerful). Not for making tools to work wood or stone with, to be sure, but as powerful tokens
for representing power and wealth.
Those ages, however, are no longer only tied to the use of stone or metals for tools,
but to several parameters of human civilization as given in the table below. It compiles the data in the Wikipedia article
: "Three-age system". Note that the Wikipedia articles gives no dates; the dates in the table here are my best
guess. Note that there is some overlap. |
| | |
|
|
|
| Age | Time | Period Name |
Tools | Economy / Society |
Stone
Age |
2 Mio BC
-
300.000
-
12.000 BC |
Palaeolithic |
Handmade tools and objects
found in nature
– cudgel, club,
sharpened stone,
chopper, handaxe, scraper, spear,
harpoon, needle,
scratch awl.
"Art" as early as 30.000 BC |
Hunting and gathering
in small bands of 25–100 people |
-
6.000 BC |
Mesolithic
(other name:
epipalaeolithic) |
Stone tools employed in
composite devices
– harpoon, bow and arrow.
Other devices such as fish
– basket, boats
Occasional use of native copper
|
Intensive hunting and
gathering, porting of
wild animals and seeds
of wild plants for
domestic use and planting.
Temporary villages |
-
3.000 BC |
Neolithic |
Polished stone tools,
devices useful in subsistence
farming and defense
– chisel, hoe, plough, yoke,
reaping-hook, grain pourer,
loom, earthenware (pottery) and weapons.
First use of (smelted) copper and Gold
|
Neolithic Revolution - domestication of plants
and animals used in
agriculture
and herding,
Supplementary gathering,
hunting, and fishing. Warfare.
Permanent settlements up
to to walled
cities.
Chiefdoms; "Kings" |
Bronze
Age |
6.000
-
2.000 |
Copper Age |
Copper tools,
potter's wheel |
Civilization, including craft,
trade.
Urban centers surrounded
by politically attached
communities.
City-states |
3.000 BC
-
500 AD |
Bronze Age |
Bronze tools |
Iron
Age |
1.000 BC
-
now |
Iron Age |
Iron tools;
Steel tool |
National economy
presided over by the
government.
Cities
connected by
roads.
Capital city.
Countries, empires |
| Steel |
|
| |
| |
| |
 |
It is clear why "official" sources like Wikipedia are reluctant to supply dates.
There are three main reasons: - The exact age of artifacts is not always known with sufficient precision.
- Different cultures were different.
The ancient Egyptians, for example, built the big pyramids around 2.500 BC mostly
with stone and copper tools, even so bronze was well known in other cultures at this time and
in Egypt. We can be reasonably sure, however, that they had plenty of gold in use.
- There was always an overlap between different materials and technologies and not a sudden transition. For example, bronze
and iron coexisted for more than thousand years, essentially until today.
It is thus simply nonsense to give a precise date to the beginning and end of some age. It is also not very helpful
to let a "metal age" begin with the oldest artifact made from copper, bronze or iron that has been found so far. The "ages" thus more or less refer to the bulk use of the metal.
The times
I give below thus serve only as a crude "guide to the eye". My additions are in blue |
 |
Here is a time line of "The Ages" set against some key words for
some cultures / regions. |
| |
| |
| |
 |
Bronze Age in the Near East and Europe with fine divisions
"Kassite" refers to the "New Babylon" | |
Source: Data from Wikipedia |
|
| |
| |
|
 |
The funny thing is that all those bronze ages ended somewhat abruptly in this representation.
One wonders what comes next. Well, there was never a sharp transition between the Ages. One problem with organizing time
this way and defining "ages" seem to be that historians confuse the (often rather sudden) end of a civilization,
and therefore the scarcity of certain artifacts after some date, with the end of a technology.
That is not to say that technologies could not also end or become "lost". |
| |
| |
| |
Critical View of "The Ages" |
 |
Let's look a bit closer at each of those ages - with the eye of a Material Scientist.
Let me make clear from the outset that this is a kind of naive view, based on straight reasoning following some straight
logic. Early humans, however, may have subscribed to different world views that are rather alien to us. They might not have
liked gold before about 5.000 BC, for example, for reasons completely their own.
In later chapters I will
to some small extent modify some of the simple views put forward here and give reasons for that. In order not to overwhelm
you right at the beginning with sophisticated details, I'll keep it simple here: I will also be a bit provocative on occasion,
to stir you up a bit and make you think. |
 |
The Stone Age starts about 2.5 million years ago, when humans and their predecessors
began to use stones for hard tools, possibly with a sharp edge. They also used bone and wood but few of those implements
survived. "During the Stone Age, metalworking was entirely beyond human capability" says Wikipedia - but
I have my doubts. See below and also the quote above.
In the final millennia of the
stone age our forebears produced very fine stone tools. They also did not just pick up the odd flintstone here or there
for their arrow tips, they dug for it or even mined it as early as 30.000 BC. |
|
 |
Major mining for flintstones (and obsidian) was done 6.000 BC - 3.000 BC and possibly later. This period
overlapped already with the copper and bronze "ages". In Egypt, for example, stone tools including complex flint
stone products were used on a large scale to built the pyramids around 2500 BC. |
| |
|
|
|
 |
| Flintstone knifes from the tomb of Ninetjer |
| Source: SOKAR No 18; 2009, p. 21 |
|
| | |
|
|
 |
Ninetjer was the third Pharao of the 2nd dynasty. He ruled from 2785 BC - 2742
BC. The flintstone "razor blades" shown above were obviously still of some value; they were unearthed in 2008.
|
 |
When we consider "The Ages", we usually forget about the Far East and
the "New World". Me too, in most of what I have to say here. So just one little observation: Pretty much all Meso
and South American Cultures tortured and sacrificed enormous numbers of captives, up to the time when they were eclipsed
by the Spaniards after 1500. And all this beheading and cutting out of the heart was done with obsidian
blades as shown below. They are just as good or typically far better than copper knifes for this kind of work. They
also built their pyramids with stone tools. The Aztec, Maya, Inca and so on must not be considered to be stone-age, however.
They produced highly sophisticated pottery for example, not
to mention plenty of high-culture artifacts like big government, big buildings, tyrants, and strange religions. |
| | |
| |
 |
| Aztec sacrificial knifes | |
Source: Photographed 2014 in the Templa Major museum in Mexico City |
|
| | |
 |
Real stone age people, at least if he or she lived in Suebia like me, did not
just make flint stone tools. He also painted the walls of his cave with art some 15.000 years ago, and long before that
- 30.000 to 40.000 years ago - he made complex ivory sculptures including a figurine known as Venus of the "Hohle Fels" (hollow rock). And he or she made music. With flutes from bird bones.
I can't resist the temptation to claim several "firsts" for my early countrymen; use the link for details. |
|
|
|
 |
What I'm driving at is: given all this sophistication more than 30.000 years
ago, it is utterly inconceivable (for us) that those people would not pick up those pretty yellow or brown-green-reddish
stones - gold (Au) or copper (Cu) nuggets - that they encountered here and there, and then made something from it.
Even
the North American Indians, who had never any metal technology to speak off, did use the native copper pieces they found
in the so-called "Old Copper Complex"
around Lake Superior. The "Copper Complex people" learned to hammer the copper and produce a variety of spearpoints,
tools and decorative objects; they also traded copper goods to obtain other exotic materials. The "Copper Complex culture"
can be dated back to at least 3.000 BC. |
|
| |
| |
|
| | |
|
|
 |
Left: Gold crystals from California; several octahedral fcc single crystals are visible Right: Many
small gold piece sintered together by geological forces. "Sintering"
here is a kind of natural "hammer welding"
or cold-welding; all you need is some pressure. |
 |
I'm thus reasonable to assume that elemental gold (Au), silver (Ag) and Copper
(Cu) was used by "stone age" guys whenever it could be found in its pure metallic state. That did not happen all
that frequently and not everywhere, so whoever was lucky enough to find some of the good stuff, treasured it. Well; it is
a reaonable assumption but probably wrong to some extent. I'll come back to this. |
|
 |
Given an overlap of the stone age and the "copper age" (sometimes seen
as part of the "bronze age") for many thousand years, and at least 2.000 years of overlap between the bronze age
and the iron age, one might also propose a different system that doesn't look so much on the kind
of metal found, but on the technologies people used to work with metals of any kind. |
 |
In other words, the Copper age or, to use the
fancy term: Chalcolithic age (ancient Greek once more: khalkos = copper + lithos =
stone) need not be the first metal age. In my system it doesn't matter what kind of
metal was used, what matters is how the metal was produced and worked.
|
 |
The first metal age then was the "native
metal age". You may also use the qualifiers "pure", "sterling" or "elemental". The English
language lacks a definite word for metals that are found as metals and not as metal ore, in contrast to German where we have "gediegen". |
|
 |
Starting in this way, a system follows quite naturally. Here it is:
- Working with elemental metals.
- Carbon smelting of simple metals and casting.
- Carbon smelting of difficult metals and forging.
- Electro-smelting of very difficult metals.
|
 |
I avoided the use of the word "age". The technologies mentioned above
may have occurred at quite different times in different parts of the world, may have overlapped, may have been known but
not used by some cultures and may not even have the sequence given above in time. Copper may have be been smelted before
gold was cast, for example. An "age" also refers to something that was important for the civilizations living
in it. Working with native metals, or with metals produced by smelting just occasionally on a small scale, may not have
been important, though. |
|
 |
Of course, plenty of other people have had thoughts along similar lines. There is an increasing
number of good books to the topic here, and there is even a relatively new field of science dedicated to the issue called
"archeometallurgy". This
link leads to just one of the many papers to this subject that illustrates what is going on in that new field of science.
The transition from any of the technologies enumerated above to the next one demands a major breakthrough in metal technology.
All I want to do here is to raise your awareness for what that means.
So let's go through my list and compare it to
the "classical" copper / bronze / iron ages. |
|
|
|
Working with Elemental Metals |
 |
That stage of human development started when somebody somewhere picked up a piece
of elemental gold or copper, possibly silver, from the ground or pried it from a rock, and banged it into some shape with
a stone. Maybe that person even produced a little sculpture or whatever. The three metals mentioned above, and possibly
mixtures or alloys like "electrum", a naturally occurring alloy of gold and silver,
are pretty much all you can find as far as elemental metals go. Finding silver would be least likely, by the way.
One
might include elemental meteorite iron in this list but it is exceedingly rare and there are very few proven
cases of its use; consult the fabulous book "Iron and steel in ancient times" from Vagn Fabricius
Buchwald or this link for details. |
|
 |
In some areas of the globe it wasn't all that difficult to find those metals
in long bygone days. As far as Europe is concerned, native copper could be found in Cyprus, Crete, Turkey and many other
places. In fact, the name "copper" goes back the Roman "Cyprium"
(later "Cuprum"), meaning "the stuff from Cyprus".
It is difficult nowadays to pick up some gold
or copper while hiking around. Whatever was easy to pick up, has been picked up long ago. Of course, in some areas you couldn't
find anything at all, while in others places there were rich pickings. My suebian
forebears, for example, couldn't find much elemental metals around the "Schwäbische Alb"; the geology
just isn't right. |
 |
We don't know if and when a first gold or copper object was made
by some stone age artist. The first old gold objects unearthed by archeologists are
from around 4 500 BC (see below), the first copper objects from about 8 000 to 9 000 BC. |
|
 |
How can that be? Wouldn't whoever collected copper also collect gold? Did nobody pick up
and use gold before 5 500 BC? The answer is simple: we don't know! Either early men did not work with gold, or he did and
we just didn't find the evidence yet.
There is an easy explanation for the latter point. That little gold Venus that some stone age guy perhaps had made some 30.000 years ago,
is now part of your wedding ring. In contrast to ivory, stone or ceramic objects, a gold object, after it has served its
intended use, can be reused. Since gold was also far more precious than ivory, you can be quite sure that it was reused,
indeed. The same principle applies for silver and copper.
That this happened in more modern times is easy to demonstrate.
The 4th dynasty Egyptians, living from 2 620 BC - 2 470 BC, produced around 25 million tons of dressed stone,
including huge amounts of precisely shaped hard granite or other hard stone; see the examples below. With those stones they
built the pyramids and scores of temples etc. - using only stone and copper tools. They
were well into the "carbon smelting of simple metals and casting" technology or the classical "bronze age"
by then, and thus knew how to make copper in bulk. For hollowing out huge granite sarcophagi or delicate vessels they used
hollow copper drills plus quartz sand as abrasive; see below. All in all there
must have been many thousands of copper tools in constant use for many hundred of years. Here are a few pictures: |
| |
| |
| |
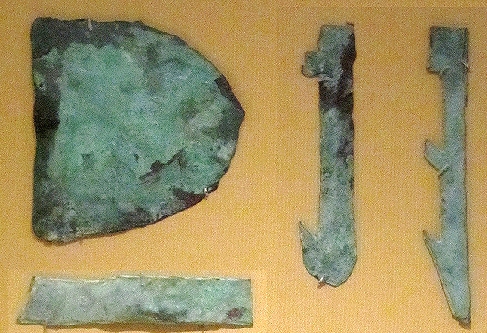 |
| Model copper tools form the grave of King Kasekhemui (ca. 2.650 BC) |
| Source: Pictures taken at the Metropolitan Museum New York. |
|
| |
| |
| |
 |
These things are only models, like much of the other stuff going into the
grave with the Boss. I would guess that the copper came from a smelter but was rather hammered into shape than cast. |
| |
| |
| |
 |
 | | Copper axe and copper bread forms |
| Source: Picture taken at the Roemer-Pelizaeus Museum; Hildesheim, Germany. |
|
| |
| |
| |
 |
The copper axe here is the real thing but according to the museum from a much later time:
6th - 12 th dynasty, i.e. 2 320 BC - 1 780 BC, than the models above. That copper was used for bread forms indicates that
copper was plentiful - but not so plentiful that a hollow copper drill was left for us to find. |
| |
| |
| |
 |
| Small Egyptian stone vases hollowed out with copper tools |
| Source: Roemer-Pelizaeus Museum; Hildesheim, Germany. |
|
| |
| |
|
 |
Those small vases made from very hard stone. There is a general agreement that the first step
was to drill the stone with a hollow copper drill. Huge amounts of vases and pots like these, made more than 4700 years
ago, have been found. |
 |
So how many of those copper tools do you find in all the museums
today? You probably could carry off the lot in a few suitcases - and it wouldn't contain a copper drill, none has been found so far! As far as stone tools are concerned, we have many tons of the
things. You can easily find some yourself when you wander around the pyramid areas and keep your eyes open.
Note that
I'm not implying (like a great many Internet sources) that those guys had some tricky
way for working stone without copper tools. I personally know that you can cut granite with soft copper plus an abrasive
powder. I have done it myself (with hard silicon instead of granite). I'm only implying that the metals were almost completely
recycled in the early days. When they were put in the grave with the owner, that grave was almost always robbed as a matter
of course.
This is one of the reasons why I can't supply many pictures with copper tool examples here. However, I
do have an example of a pot partially hollowed pour by (hollow) copper drills - here
it is. I found it in Mexico, of all places.
The other reason is that museums usually do not offer a lot of information
with respect to old metal objects. And if they do, it's often
puzzling or wrong.
Anyway, here is one picture possibly showing gold and copper objects that were not
cast and thus belong to the "age" in question: |
| |
|
|
|
|
| |
| |
|
 |
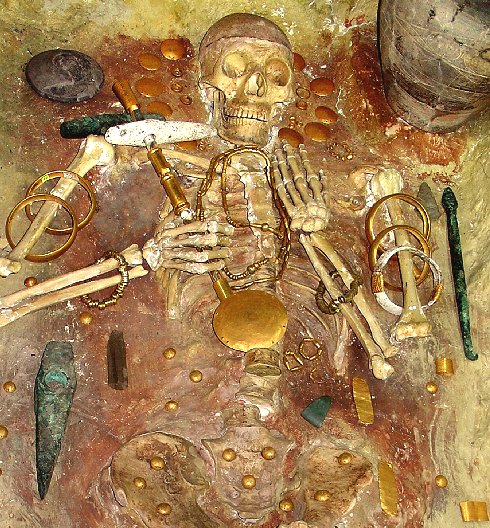
You see some of the world's oldest gold objects unearthed so far; they are dated to 4700 BC
- 4200 BC (or 4400 BC - 4100 BC) You also see copper objects but there are even older ones around. |
 |
The Varna Necropolis, accidentally discovered in October 1972 and located in present-day
Bulgaria, is a burial site that is internationally considered to be of prime importance for world prehistory. It contains
a lot of the oldest gold artifacts unearthed so far. It also contains copper objects (the greenish stuff) and stone tools
(also green). Note that smelting of metals like lead and possibly also copper was most likely know for a few centuries before
the Varna time. |
|
 |
The gold objects do not look cast to me. Some scientists tend to agree 2. So how come that many of those gold objects have the same size considering that nuggets are of
different sizes? Moreover, the copper parts are certainly far larger than the biggest elemental copper piece you can find.
The answer might be: The larger gold and copper pieces were hammer-welded together. That should be possible, especially if you heat the pieces before banging away. In
fact, many naturally occurring gold nuggets seem to have been welded together from small parts by geological forces.
|
 |
We might surmise that's that what the Varna people did with their gold, and possibly
copper. They must have had already some experience with this. That's why I guess that this "oldest gold found"
comes rather from the end of the "Cold Working of Elemental Metals" technology,
and that elemental metals were used long before this. |
|
 |
When I wrote this part I was inclined to assign a high probability to the "hammer welding
and hammer forming" hypothesis. I no longer do. Things are just more complicated - find out by reading on to chapter
10.
It would be interesting to examine the Varna pieces to see if hammer welding can be verified. It can and has been
done in other cases. |
 |
So when did our ancestors start to work with elemental metals? Who knows. The
oldest gold artifacts, as shown above, date from around 4.500 BC. The oldest copper artifacts go back to about 9500 BC in
the Middle East. It wouldn't surprise me very much, however, if older ones will be found some day for all the reasons discussed.
But I don't think it is very likely. |
| | |
|
|
Casting of
Elemental Metals |
 |
Casting metals, I surmise, had to wait until
pottery had advanced to a point where some kind of firing was used that allowed temperatures up to 1100 oC (2012
oF). The reason is simple: If you want to cast a liquid metal, you need a crucible that can take the temperature!
If you fire a ceramic bowl at temperatures higher than the melting point of the metal to be cast and it survives, chances
are that it will make a good crucible.
Let's look at some relevant melting points (the precise melting points of the
alloys depend, of course, on their composition). |
| |
|
|
Material
Melting point |
Copper
(Cu) | Gold
(Au) |
Silver
(Ag) | Lead
(Pb) |
Zinc
(Zn) | Tin
(Sn) |
Electrum
(Au+Ag) |
Bronze
(Cu+Sn)
Brass
(Cu+Zn) | |
oC | 1083,6 | 1064,58 |
962,08 | 327,65 |
419.0 | 232,12 |
1050 - 980 | 1000 - 900 |
| oF | 1982.5 |
1948.2 | 1763.7 |
621.8 | 786.2 |
449.8 | 1922 -1796 | 1832 - 1652 |
|
| | |
 |
Whichever way you look at it, casting of Gold (Au) or copper (Cu) demands temperatures
well above 1000 oC (1832 oF). You won't get that kind of temperature by just setting
some wood on fire on a hearth. You need a
kiln or some other special design. |
|
 |
Some kind of pottery is very old. There is a Venus figurine from a palaeolithic settlement in Dolní
Vestonice, Moravia, now a part of Czechoslovakia. It is between 27.000 to 31.000 years old and thus several thousands
years younger than the suebian ivory venus mentioned above. It is prettier,
however.
The Dolní Vestonice Venus is the among the oldest known ceramics
in the world. It has a height of 111 millimeters (4.4 in), and a width of 43 millimeters (1.7 in) at its widest point, and
it is made of a clay body fired at a relatively low temperature that wouldn't have molten the elemental metals. |
| |
Venus figurine making happened all over Europe (and in other places) 30.000 years ago (give
ore take a few thousand years) but metal melting was probably not yet possible. Just for the hell of it, I give you a Venus collection in the link. |
 |
Back to metal casting. Proper kilns (meaning a closed hot container) came relatively
late. The oldest known method for making pottery at high temperatures was pit firing.
The raw pots were nestled together in a pit in the ground, and covered with combustible materials. The filled pit is then
set on fire and: temperatures of 900 °C (1652 oF) were possible this way. With luck and some added sophistication
(e.g. cover the pit to some extent with a layer of clay), even 1 100 °C (2 012 oF) could be reached. |
|
 |
Putting some gold or copper inside a pot-to-be-made thus might have produced molten metal
on occasion. That is not good enough, however, because casting does not only require
to melt the metal, but also to keep it molten, to produce a suitable mold, and to find a way of pouring the very hot melt
into the mold. The whole process chain appears to be a bit too difficult for routine use with simple pit firing.
For
metal casting, a different kind of high-temperature "furnace" needed to be invented. Considering, that gold was
never cast in huge quantities, and that even copper tools rarely were heavier than 1 kg or so, the whole thing could be
much smaller than the contraption you needed for making pottery.
The smart thing to do was to blow lots of air into
a relatively small charcoal fire that heats a crucible. That's what those old Egyptians did at least 4.400 years ago; here
is that picture again: |
| | |
| |
 | | Melting Gold (??) in Ancient Egypt |
|
| | |
|
| |
|
What you see is a relief in the tomb of Mereruka in Saqqara, Egypt. It dates
to the 6th dynasty, 2 450 BC - 2 350 BC. Ancient smiths (or more likely their apprentices / employees / slaves) use blowpipes
to supply air to a kind of hearth, getting the temperature up. Maybe they intended to melt gold or copper, maybe they were
smelting copper. |
 |
As soon as a given culture had discovered the technology of casting elemental metals, it was bound to discover smelting. It might even have been the other
way around. So let's not dwell on this topic any longer but move on to: |
| | |
| |
Carbon Smelting
of Simple Metals and Casting |
 |
Let's consider my terms: "Carbon Smelting"
simply means that carbon (C; or more precisely carbon monoxide CO) has a higher affinity to oxygen than the metal in question.
In human terms that simply means that carbon is able to rip off the oxygen from some metal ore, producing carbon dioxide
(CO2) and the elemental metal. Some metals, for example aluminum, titanium, sodium or and silicon, will never
allow that - and that's why I call them "difficult metals". |
|
 |
Ore (German: "Erz"), by the way, is an old
term that for our purposes means; "rocks or earth containing a chemical compound
of the element you want plus God knows what else. Etymologically speaking, "ore
" and "erz" may have their roots in "earth" (German "Erde"), the old-English "ar"
= brass, copper, bronze, or the Old German "aruz", meaning copper and containing "rot" = red.
|
 |
Simple metals essentially mean metals
that i) could be smelted with carbon as reducing agent, and ii) have a melting
point below about 1100 oC (2.120 oF). |
|
 |
Aluminum (Al) and magnesium (Mg), for example, have melting points of 661 oC (1.220
oF) or 649 oC (1.200 oF), respectively, but cannot
be smelted with carbon so they are not simple metals for our purpose here.
Iron
(Fe), cobalt (Co) and nickel (Ni), to give another example, can be smelted with carbon
but have melting points well above 1.400 oC (2550 oF), so they don't qualify as simple
metals either. |
 |
This is the point were I need to exorcise a demon plaguing a
lot of archeologists or museum curators: |
|
| |
| |
Smelting has nothing to do with melting!
|
|
| |
|
|
 |
An ore is not
some porous rock containing tiny pieces of the elemental metal (exception: noble metals) but a chemical compound of the metal, usually an oxide or sulfide. An ore is as different from the metal, as a
piece of rust is from iron, elemental sodium from rock salt, or hydrogen and oxygen gas from water.
Smelting means to
break that chemical compound apart into the pure metal and some residue; chemically this is called reducing
the ore. "Smelting" water, for example, would mean to break it apart into oxygen and hydrogen. This is not easily
done and it certainly does not happen at temperatures around the melting point of solid water (then called ice).
For
smelting metals you also may need temperatures higher than the melting point of the
metal. Smelting lead, for example, needs temperatures far higher (at least 800 oC (1472 oF) than its
melting point at 328 oC (622 oF).
On the other hand, you can smelt some metals from their ores
at temperatures far below the melting temperature of the metal. Iron smelting was done
this way for more than two millennia. However, you won't get your metal in the liquid state in this case, and that makes
the total process chain from smelting your metal to the final product far more difficult – witness iron and steel
technology, the topic of this Hyperscript.
And, as a last point, you don't melt
the ore either. If you do, you just might get liquid ore. You know that. Melting hydrogen ore also known as ice (solid H2O)
does not produce hydrogen (H) and oxygen (O) but just water or liquid H2O. Maybe more to the point: melting rock
salt (NaCl) at 801 °C (1 474 °F) produces molten salt and certainly not the metal sodium (Na). And adding carbon
in any form won't help either.
What you do when smelting ores with carbon is to immerse your ore in an atmosphere of
carbon monoxide (CO), a gas. You produce carbon monoxide by first burning carbon, typically
in the form of charcoal, with oxygen to carbon dioxide (CO2). Next the CO2 reacts with hot
carbon to carbon monoxide in an oxygen deprived part of your smelter. This needs a certain
minimal temperature - not much will happen below about 1 000 °C (1 832 °F). The carbon monoxide reacts with the
ore, taking off its oxygen to form carbon dioxide (CO2), leaving back the metal. This reaction may occur at rather
low temperatures that have nothing to do with the melting point of the metal. |
|
 |
Smelting is an extremely complex process if you look at it scientifically.
Yes, I know that you, just like your ancestors, are perfectly capable to produce a metal
by simply igniting a mix of charcoal, ore, and possibly some "flux" - even so you don't know a thing about what
is going on. That doesn't mean it's simple. A lot of people know how to make a baby without having the faintest notion of
how that works in detail. More to that (smelting, not baby-making) in chapter 10 |
 |
As soon as the "age" of smelting began in earnest, serious mining must have started too. Usually you can't pick up enough ore just from shoveling it off the
ground; except if you go for "bog iron".
Moreover, charcoal
production in bulk was also needed. Any wood fire not too well tended produces some charcoal, but that would not have been
enough for serious smelting. |
|
 |
Mining is a tricky enterprise. You need to identify sites with rich deposits of the ore you
want, and you have to dig deep into the earth and rock to get it out. For digging you needed tools, some light, and luck
with the ground water and the air supply.
I haven't found much literature about ancient charcoal
production. Of course, charcoal has been around a long time; it is typically used for dating archeological sites
with the C14 method. But then it might be just the residue
of normal fires.
In ancient Egypt (as in modern one) there weren't enough trees to support the production of charcoal
in bulk, and that may be one of the reasons why so few copper tools have been found. Copper
production by smelting must have been expensive because both ore and charcoal had to be imported. Recycling was just an
economic necessity. Note that for smelting you typically need more (char)coal than ore. That's the reason why modern steel
production usually takes place in coal mining areas, with the ore shipped in. It's cheaper than the other way around. |
| | |
|
|
|
 |
| Oldest know mine for copper ore |
|
Source: Internet (disasturbation.wordpress.com/ 2010/06/29/holey/) |
|
| | |
|
|
 |
The picture shows an underground shaft in the Timna Valley in the Negev Desert / Israel, thought
to be the oldest mine for the copper (Cu) ore (malachite in this case), going back
to the 6th millennium BC. There are even older mines for flintstone. |
 |
Only "(s)melting" of gold and the other truly noble metals like platinum is different. Noble metals rarely occur as chemical compounds (that's why they are
called "noble"). So in these cases you actually really just "melt" the metal out of the "ore"
that contains it in elemental form. In case of platinum, however, the melting point is 1772 oC (3222 oF),
so it's not so easy to do. There are other methods, however,
that may have been used in ancient times for extracting gold from the rocks that contain it (e.g. by alloying it with mercury
(Hg)). |
|
 |
In contrast to gold (Au), silver (Ag) is typically smelted. You can find elemental silver in many places but it is quite rare and mostly you go
for its ore, typically a sulfide, often mixed up with lead ore. You do know that silver it not noble enough to withstand
the lure of sulfur (S). Your Grandma never let you eat a sulfur-rich egg with her silver spoons on the grounds that it it
would quickly tarnish, i.e. form that blackish coating that needs to be cleaned off once in a while, even if you don't eat
eggs with your silver.
Well, here is some elemental silver. It is very pretty and you can get an idea why some cultures
treasured it more than gold. |
| |
|
|
|

| Elemental or native silver
Source: Internet |
|
| |
|
 |
We might expect then that first smelting started unintentionally
with ores of easy-to-smelt metals like tin (Sn; probably cassiterite,
the name for tin oxide SnO2), lead (Pb, probably galena,
the name for lead sulfide PbS), silver (Ag, probably silver sulfid Ag2S,
often found together with galena), zinc (Zn; probably sphalerite, the name for zinc sulfide ZnS),
and finally copper (Cu, coming in many
different minerals, the most prominent ones being: cuprite (CuO), malachite (CuCO3(OH)2), azurite
(Cu3(CO3)2(OH)2), chalcopyrite (CuFeS2), chrysocolla ((Cu,Al)HSi2O5(OH)·nH2O)
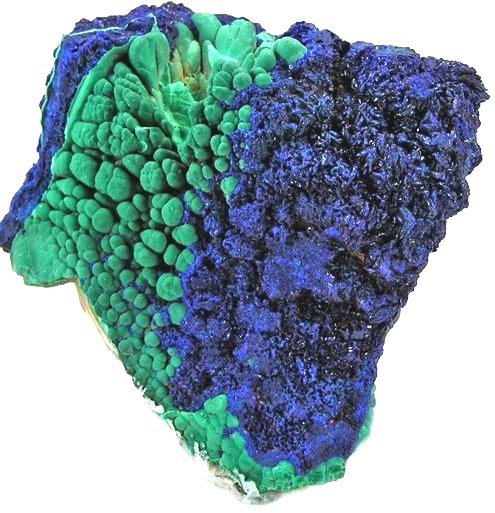
and tennantite Cu12As4S13). Most copper minerals, in particular malachite and azurite,
have bright colors (green or deep blue, respectively) and were used as gemstones for millennia. Here is a picture: |
| |
|
|
|
|
| | |
|
 |
We can be reasonably sure that the first smelting happened accidentally. It's
just not conceivable that some ancient guys, after having had a few beers (of course there was always some kind of beer
around, you just can't have a decent civilization otherwise), were inspired enough
to come up with the idea ("hey, how about smelting a few metals now"?) |
| |
 |
It is often assumed that accidental smelting could have happened if some lead
or copper ore happened to be in that fire pit used for barbecuing mammoth spare ribs. it also might have happened when some
early potter adorned his raw pottery with nice looking stones. If you take one of the pretty copper ores to decorate your
"green" ware, and your pottery pit fire happens to be very hot, you might get copper this way.
|
| |
I did subscribe to that view myself some time ago - but now I'm not so sure anymore.
All of the above could have happened, indeed, but accidental smelting like that is far more unlikely than it appears at
a first glance. Not impossible but unlikely.
Nevertheless, it must have happened, and it probably happened more than
once before the first ancient engineer noticed that you could make metals this way intentionally.
You had to be in a relaxed state of mind to come up with this idea and drinking beer certainly helped. So did some already
advanced "pyrotechnology", the use of fire to do
high temperature processing. | |
|
 |
The big question is: When, what, where and who? When
did the first intentional smelting take place, where
was that, who made that discovery - just one guy from whom it spread, or many guys independently
- and finally what kind of simple-to-smelt metal was produced first ? Nobody knows for
sure. If the malachite mine shown above is really from the 6th century BC, it would give a date for early copper smelting.
But the ancient miners may have dug out the malachite not for smelting copper but for making jewelry with it. |
 |
Looking in the Internet or in books, you find a lot of dates and finds claiming
for a "first". However, thanks to the advent of archeometallurgy,
a lot if not all of reported "firsts" are now disputed. |
|
 |
1. Example:
"At the ancient Hittite
city of Catal Hüyük in Turkey, beads of lead have been uncovered that date back to about 6500 years BC",
is a statement you can find many times. Lead could only have been obtained by smelting, so here is the answer to the question
above?
Maybe - but: "At Çatal Höyük, Turkey, a mid to late seventh millennium bead was thought
to be of smelted lead, but is now known to be of shaped galena." (G. Sperl, "Zur Urgeschichte des Bleies",
Zeitschrift für Metallkunde, 81, 1990, 799 - 801). This is a quote from a very good blog that you can access in this link, and that has the most recent information about the
topic.
Galena or lead sulfide (PbS), by the way, does look a lot like elemental lead; it's thus easy
to make that mistake. Here is a picture of galena: |
|
| |
| | |
 |
| Galena or lead sulfide (PbS) |
| Source: Wikipedia |
|
| | |
|
| |
 |
Galena usually is mixed with some silver sulfide (Ag2S) or Acanthite
and that's why lead and silver mining / smelting go usually together. |
| |
|
| |
 |
2. Example:
Quote "The first
smelted copper artifact was thought to be a copper mace head from Can Hasan, Turkey, dated to 5000 BC".
Well - no. That mace head is now considered to be made from native copper that was hammer welded. There is some indication of the seam on the left hand side.
Just how you hammer some copper
into a kind of hollow sphere is a different question. I'm sure my grandfather, who was a smith, would have known how to
do this.
Read more in this link. |
|
|
|
 |
3. Example:
"The oldest known piece of worked copper is often quoted as being a copper pendant from the burial site of Zawi
Chemi / Shanidar Cave, northern Iraq, dated to the middle of the ninth millennium BC. However, this is not worked copper
but ground and polished copper ore, probably from Turkey to the north. It is made up of malachite and chrysocolla but happens
to contain a fair amount of native copper".
That's a quote from this
source. Even more here. |
|
| |
 |
4. Example:
The eminent scientists Sherby and Wadsworth, well-known to students
of ancient steel, got so fed up with the prevalent age dogma, that they wrote a full paper 1)
proposing that the iron age not only precedes the bronze age but may even go back to Neanderthal times, i.e. to 300.000
to 40.000 BC.
I don't believe that for a second - but that is believe and not knowledge. |
 |
Be that as it may, smelting of copper and so on was definitely done as far back
as 5.500 BC, almost 2000 years before the bronze age "proper" started.
It is clear that if you could smelt copper intentionally, you could also melt and possibly
alloy it.
That doesn't mean, however, that everything was now made by casting.
Below is an example of a rather "new" and complex copper artifact from 2.400 BC - 2.000 BC that does not look cast to me. |
|
 |
However - I don't know for sure. All that the Metropolitan
Museum in New York City has to say to this object is: |
| |
| |
| |
Wagon drawn by bulls
Copper
Anatolia
Early Bronze II - III, 2400-2000 BC
Four wheeled
wagons driven by a deity and drawn by a team of four animals in cultic processions are often depicted in Anatolian seals. |
|
| | |
|
|
 |
No mention of how it was produced is made, and no picture of those "Anatolian seals"
is provided either. Well, here is that wagon; more about the problems with exhibits like that in this
link. |
| | |
|
|
|

|
| Copper (?) wagon drawn by bulls.m
Cast or hammered? | |
Source: Photographed in the Metropolitam Museum New York |
|
| | |
|
 |
Making bronze means you need to do do some alloying,
mixing two or more metals. Alloying can be done in two ways. Accidentially or
intentionally |
|
 |
Accidential alloying happens if your ore contains not
only copper compounds but also, for example arsenic (As) or antimony (Sb) compounds. This is actually rather common. In
the smelting process, metallic arsenic (As) may also be produced and dissolves in the copper. What you get is what is called
an arsenic
bronze if the arsenic concentration reaches at least a few percent. |
|
 |
Intentional alloying happens if you either add intentionally
some special ores to your copper ore, or if you produce an alloying element separately, for example tin (Sn), and then melt
a mix of copper and tin to make tin bronze. |
 |
The first smelters must have noticed that results depended on what you put into
your smelter. If you got some kind of bronze instead of relatively pure copper, you couldn't fail to notice that it was
harder than copper and thus more useful. Why an alloy is always
harder than the pure metal you will learn if you keep reading this Hyperscript. |
|
 |
Again, it is not so clear when and where serious alloying technology started. The best guess
is between 4 000 BC and 3 000 BC. The pyramid builders in Egypt (2 620 BC - 2 160 BC), however, appear not to have used
bronze as outlined above.
Around 2.000 BC, tin ore was being mined on purpose for
the production of bronze. That is not so easy because tin ore is rather rare, and even rich cassiterite
contains only 5% tin. But by 2.000 BC some cultures must have mastered the bulk production of good tin bronze. That means
that there must have been several centuries before that, when the technology was there but not yet fully developed.
It is one thing to know about some technology, and another thing to use it. The Egyptians may not have had easy access to
tin (tin ore is far rarer than copper ore), or simply kept on doing whatever they did the old-fashioned way.
The (North)
European "battle axe culture" (2 900 BC – » 2 300 BC) used stone axes modeled on copper axes, even with imitation "mold marks" carved in the stone. So they
new about the superior technology but couldn't do it for some reason. This is nothing new. A lot of present-day cultures
know about microelectronic chips or solar cells but can't make it either. |
 |
The next two pictures show two typical copper / bronze artifacts
you find in museums. These two are also from the Metropolitan Museum in New York. |
| | |
| |
 | | Copper figure with Ibex horns |
| Source: Photographed in the Metropolitam Museum New York |
|
| | |
|
|
 |
The text in the exhibition states only hat this is "arsenical copper"
(whatever that means) from Iran or Mesopotamia, from around 3 000 BC.This figure was obviously cast but
this is not mentioned in the exhibition. In the Internet we learn that it is a Proto-Elamite copper alloy,
and quite a bit about the the context of the figure, including: "This solid-cast
sculpture is one of a pair...". |
| |
| |
| |
 | Silver bull holding a spouted vessel
Proto-Elamite; Susa, Iran; Around 3100 BC-2850 BC
More Elamite silver |
| Source: Photographed in the Metropolitan Museum New York, 20011 |
|
| | |
|
|
 |
The bull is from southwestern Iran; proto-Elamite period, 3 100 BC - 2 900 BC. it also has some pebbles inside that make it a rattle.
It looks
cast to me, probably in several individual pieces that then were joined together. The museum does not provide any information
about this in the exhibit or in the Internet. |
 |
Around 2 000 BC, when proper tin-bronze technology was well established, the next
technological breakthrough occurred and the next "age" came up. |
| | |
|
| |
"Age"
of Carbon Smelting of Difficult Metals and Forging |
 |
Difficult metals in this context
means by default that i) smelting could still be done with carbon as
reducing agent and ii) the melting points of the metals to be produced is well above 1100 oC (2012 oF). |
|
 |
That certainly covers iron, the topic of
this Hyperscript. Its melting point is 1 535 oC (2 795 oF). It also covers, for example, nickel (Ni;
1 453 oC (2 647 oF)), cobalt (Co; 1 495 oC (2 723 oF), or chromium (Cr; 1 857
oC (3 375 oF).
Note that ancient man did not make and use these metals - but this is the category
they would belong to. |
 |
I'm not going to say much about iron technology here; this is what the whole Hyperscript
is for. I'm just going to make a few points to stay within the system. |
|
 |
First: Smelting
of iron is not much more difficult than smelting copper - as long as you get everything just
right. Lots of people with some interest in early technology now do it routinely as a weekend group activity. The reason
is that you do not have to reach the melting temperature of iron, a mere 800 oC (1.472 oF) or so will
suffice.
Second: The low-temperature smelting process, however, is not very efficient.
The ratio of what you put in and what you get out is rather bad.
Third: What you get is not iron but, with
luck, a "bloom": a lump of spongy and porous iron and steel, intimately mixed with slag and God knows what else.
Fourth: The real technological break-through thus involved not only some reliable
procss to making a bloom but also everything one might subsume under "forging". This
contains a lot of processing steps, from banging your red-hot mess of iron and dirt (the "bloom") obtained right
after smelting with a hammer in order to compact it and to squeeze out some dirt and slag, up to the final forging of extremely
complex and fine objects like swords, with many steps in between. Don't forget: you couldn't cast iron or steel! |
|
 |
Read the main part of the Hyperscript to learn more about this. |
 |
Here are some pictures of early iron objects: |
| | |
|
|
|
 |
 |
| Iron dagger of King Tut |
Iron Sword from Luristan |
| Source: Kairo Museum |
Source: Potoraphed in the Metropolitan Museum, New York, 2011
|
|
| | |
|
|
 |
The iron dagger of famous King Tutankhamun
is one of the oldest well-preserved iron objects in the world; it dates from around 1.330 BC. It has never been properly
investigated!
Well, that is no longer true. In 2016 the dagger was investigated (by X-ray fluorescence without touching
it) and found to be made from meteoritic iron! More about
that here. |
|
 |
All the museum has to say to the "iron sword" is:
Sword with figure on handle,
Iron
Western Iran, Luristan
8th - 7th century BC.
To me, the "handle"
of the iron sword in the Metropolitan looks very much like
it was cast. Then it can't be iron; must be bronze. If it was no cast but forged, this would be remarkable and should be
mentioned. We have just one more example of the lackadaisical
attitude museums take when it comes to anything remotely "technical"
and not artistic. I will have to say much more about the Luristan swords later on. They all come from illegal digging about
100 years ago. In recent years they have been recognized as being very special; they seem to be also about 300 years older
than what is stated above. The Metropolitan, by the way, is no longer exhibiting "its" sword in 2013. |
| | |
|
|
|
 | | Iron axe head with silver parts
| | Source: Metropolitan Museum New York |
|
| |
|
|
 |
It's Scythian work form the 5th - 6th century BC and about 500 years younger than the Luristan
sword. It is, like most iron objects that old, just a piece of rust by now, not unlike the knife
from what is now Moldova. |
| |
|
|
"Age"
of Electro-Smelting of Very Difficult Metals. |
 |
Some metals (and other elements) like to be associated with oxygen even more then
carbon does. They simply will not yield their oxygen (or whatever else they are bonded with) to carbon under any circumstances.
The most important metals / elements in that group are aluminum (Al) and silicon (Si) but all alkali
metals like sodium (Na) and potassium (K) and earth alkali
metals like calcium (Ca) or magnesium (Mg) also belong into that group. |
|
 |
Removing the oxygen or whatever from this elements means to "reduce"
them, which in turn means to transfer some electrons to the element. That is what carbon does if the element is susceptible
to carbon smelting. If carbon can't do that, you could actually use aluminum as reducing agent. You can "smelt"
silicon from its oxide (SiO2 or quartz) with aluminum powder; I've actually done that - but you need aluminum
first. |
|
 |
Using electricity is by far the easiest way to force some electrons on the aluminum atoms
in a compound, and that's the way it's done for all the "very difficult metals".
Aluminium is exceedingly
difficult to extract from its various ores (like the other elements in this group). Aluminum was more valuable than gold
before the late 1880s, when electricity became available on a large scale and production started in earnest.
The apex
of the pyramid on top of the Washington monument in Washington, D.C., finished in 1884, weights 100 ounces (2.85 kg) and
represented the largest single piece of aluminum cast at that time, when aluminum commanded a price comparable to silver.
Similar stories apply to the other "difficult" metals. |
 |
This" age" is of no interest to us here, so I won't go on. |
| | |
|
| 1) |
Oleg D. Sherby and Jeffrey Wadsworth: "Ancient blacksmiths, the Iron Age,
Damascus steels, and modern metallurgy" J. Mat. Processing Technology, Vol. 117 (2001) p. 347-353 |
| 2) |
Colin Renfrew in his article "Varna and the emergence of wealth in prehistoric
Europe" (in the book: "The Social Life of Things" Cambridge Uni. Press), or
Ch. J. Raub: "The Metallurgy
of Gold and Silver in Prehistoric Times", NATO ASI Series, 1995. |
 With frame
With frame

 Critical Museum Guide
Critical Museum Guide
 General Remarks to Literature and Sources
General Remarks to Literature and Sources
 Books and Other Major Sources
Books and Other Major Sources
 Beer
Beer
 History of Carbon
History of Carbon
 10.1.3 Smelting, Melting, Casting and Alloying Copper - The First
10.1.3 Smelting, Melting, Casting and Alloying Copper - The First
 Group 1 / IA; Alkali Group
Group 1 / IA; Alkali Group
 Group 2 / IIA; Alkaline Earth Metals Group
Group 2 / IIA; Alkaline Earth Metals Group
 Group 12 / IIB; Titanium Group
Group 12 / IIB; Titanium Group
 Group 11 / IB; Copper Group
Group 11 / IB; Copper Group
 Group 13 / IIIA;
Group 13 / IIIA;
 Part 1 Basics about Scythians and Their Akinakai
Part 1 Basics about Scythians and Their Akinakai
 Early Metal Technology - 2. Silver and Lead
Early Metal Technology - 2. Silver and Lead
 Critical Museum Guide: Metropolitan Museum, NYC
Critical Museum Guide: Metropolitan Museum, NYC
 Critical Museum Guide: Roemer- und Pelizaeus-Museum, Hildesheim, Germany
Critical Museum Guide: Roemer- und Pelizaeus-Museum, Hildesheim, Germany
 Critical Museum Guide: Archaeological Museum in Heraklion (Crete)
Critical Museum Guide: Archaeological Museum in Heraklion (Crete)
 Landesmuseum für Vorgeschichte (Halle)
Landesmuseum für Vorgeschichte (Halle)
 10.2 Making Iron 10.2.1 Early Iron
10.2 Making Iron 10.2.1 Early Iron
 Leda and the Swan
Leda and the Swan
 10.1.1 Discovering Metals and Smelting
10.1.1 Discovering Metals and Smelting
 9.1.1 Things are Complicated
9.1.1 Things are Complicated
 Magnetism
Magnetism
 Old Suebian Things
Old Suebian Things
 Radiocarbon (C14) Dating
Radiocarbon (C14) Dating
 Polishing Crystals
Polishing Crystals
 Large Format Pictures
Large Format Pictures
 The Al - Cu System
The Al - Cu System
 Smelting Science
Smelting Science
 Meteoritic Iron
Meteoritic Iron
 Early Pyrotechnolgy
Early Pyrotechnolgy
 Old Sagas, Heroes and Swords
Old Sagas, Heroes and Swords
 Old Iron Things
Old Iron Things
 Phallus Symbols
Phallus Symbols
 2.3.2 What Makes Steel so Special?
2.3.2 What Makes Steel so Special?
 4.4.1 Perfect Crystals and the Second Law
4.4.1 Perfect Crystals and the Second Law
 Riveting, Soldering, Liquid Welding Plus Gluing and Screwing
Riveting, Soldering, Liquid Welding Plus Gluing and Screwing
 Cast Iron; 9.5.1 General Remarks
Cast Iron; 9.5.1 General Remarks
 10.1.2 Copper
10.1.2 Copper
 Producing "Nirvana" Silicon or Nearly Perfect Silicon Single Crystals
Producing "Nirvana" Silicon or Nearly Perfect Silicon Single Crystals
 Hardware Around the Making of Metals and Their Proper Names
Hardware Around the Making of Metals and Their Proper Names
 Powder Metallurgy
Powder Metallurgy
 Copper Ores
Copper Ores
 10.1.5 Copper Final
10.1.5 Copper Final
 The Goldilocks Principle
The Goldilocks Principle
 Early Metal Technology - 1. Gold
Early Metal Technology - 1. Gold
 Old Copper Complex of the Western Great Lake
Old Copper Complex of the Western Great Lake
 Free Enthalpy of Reduction Processes
Free Enthalpy of Reduction Processes
 Large Pictures I
Large Pictures I
 Rosh Horesha, Shanidar Cave
Rosh Horesha, Shanidar Cave
 Smelting Science - 4. Smelting Details 1
Smelting Science - 4. Smelting Details 1
 Justitiae
Justitiae
 Speak Softly and Carry a Big Sword
Speak Softly and Carry a Big Sword
 Venus Figurines
Venus Figurines
 How Sagas Develop. The Gospels as Examples
How Sagas Develop. The Gospels as Examples
 Can Hasan
Can Hasan
 Das Grab im Busento
Das Grab im Busento
© H. Föll (Iron, Steel and Swords script)