| |
Producing "Nirvana" Silicon or Nearly Perfect Silicon
Single Crystals |
| |
Producing Semiconductor-Grade "Raw" Silicon |
|
Introductory Remarks
|
 |
It is written somewhere that in the beginning God created heaven and the earth.
It is not written what from. |
|
 |
We do not know for sure what the heaven is made of but we do know what the the
earth is made of - at least as far as the upper crust is concerned. Interestingly enough, he (or she) created mostly Silicon (Si) and Oxygen (O), with a bit of dirt (in the form of the other 90 elements)
thrown in for added value. |
|
 |
Indeed, the outer crust of this planet (lets say the first 100 km or so) consists of
all kinds of silicates - Si + O + something else - so there is no lack of Si as a raw material. Si,
in fact, accounts for about 26 % of the crust, while O weighs in at about 49 %. |
 |
However, it took a while to discover the element Si. Berzellius came up with some form of it in 1824 (probably amorphous), but it was Deville in 1854 who first obtained regular crystalline Si. |
|
 |
This is simply due to the very high chemical reactivity of Si. Pure Si (not
protected by a thin layer of very stable silicon dioxide (SiO2) as all Si crystals and wafers are)
will react with anything, and that creates one of the problems in making it and keeping
it clean.
It also holds on to its oxygen so fiercely that carbon cannot steal it, even at high temperatures. Silicon
thus could only be discovered in what I called the "electro-smelting of very difficult metals age". |
|
 |
Liquid
Si indeed does react with all substances known to man - it is an universal solvent. This makes crystal growth from
liquid Si somewhat tricky, because how do you contain your liquid Si? Fortunately, some materials - especially
SiO2 - dissolve only very slowly, so if you don't take too long in growing a crystal, they will do as
a vessel for the liquid Si. |
|
 |
But there will always be some dissolved SiO2 and therefore oxygen in your
liquid Si, and that makes it hard to produce Si crystals with very low oxygen concentrations. |
 |
What we need, of course, are Si
crystals - in the form of wafers - with
extreme degrees of perfection. |
|
 |
What we have are inexhaustible resources of Silicondioxide,
SiO2 that is fairly clean if obtained from the right sources. Since there is no other material with properties
so precisely matched to the needs of the semiconductor industry, the production process of Si wafers shall be covered
here in a cursory way. |
 |
It's cursory for my students. To you it might
look at bit involved. That's because the making of one of the most important "raw" materials of today, the silicon
wafer, is an extremely involved process. Only a few highly specialized companies are able to
do it. In case you wonder: Si wafers (to use the term a bit loosely) are the starting material for almost everything that
has to do with electronics or solar cells. If something runs on electricity (or makes electricity), it has almost certainly
(processed) Si wafer parts inside.
That's why I thought I will give you a cursory glance of what the making of Si wafers
involves. |
|
 |
And not to forget: What comes after that, what is done
with a Si wafer called "silicon technology", is a
hell of lot more complicated than the "simple" making of the wafer! |
| | |
|
|
Producing "Raw" Silicon |
 |
Fortunately, the steel industry needs Si, too. Silicon was already used
as a crucial alloying component of steel before
it started its career as the paradigmatic material of our times. |
|
 |
Most of the world production of raw Si still
goes to the steel industry and only a small part is diverted for the semiconductor trade. This is why this stuff
is commonly called "metallurgical grade" Si or MG-Si for short.
The world production in 2006 was around 4 Mio tons per year. |
|
 |
How is MG- Si (meaning poly crystalline material with a purity of about 99%)
made? Silicon belongs to the "electro-smelting of very difficult metals" category in my terminology. You must reduce the oxide in
a furnace by providing some reducing agent (usually carbon but aluminum also works), sufficient energy and
some electrical potential at rather high temperatures. |
 |
Like for most metals, the reducing agent is carbon
(in the form of coke (= very clean coal)). The necessary
energy is directly supplied by electricity. |
|
 |
Essentially, you have a huge furnace lined with graphite,
which will turn into very hard and inert silicon carbide (SiC) and with three big graphite electrodes inside (carrying a
few 10.000 A of current). The furnace is continuously filled with SiO2 (= quartz sand) and
carbon (= coke) in the right weight relation plus a few added secret ingredients like certain wood chips to avoid producing
SiC. Schematically it looks like this: |
|
| |
| |
 |
| Smelting silicon |
| Extremely simplified. The human gives an idea about size. |
|
| |
|
|
 |
The chemical reaction that you want to take place happens at about 2000 oC
and is simple in principle: |
|
| |
| |
| SiO2 + C + "electricity" Þ
Si + CO2 |
|
|
| |
|
|
 |
But there are plenty of other reactions that may occur simultaneously, e.g. Si + C Þ SiC. This will not only reduce your yield of Si, but clog up your furnace
because SiC, short for silicon carbide, is not liquid at the reaction temperature and extremely hard. If you make
SiC, your reactor ends up as a piece of junk! |
 |
Still, we do not have to worry about MG-Si - a little bit of what is made
for the steel industry will suffice for all of Si electronics applications. |
|
 |
What we do have to worry about is the purification of
the MG-Si. We must make it at least a billion times (109 fold) cleaner. |
 |
Making clean, "electronic grade" poly silicon is essentially done in
three steps: |
|
 |
First, Si is converted to trichlorosilane (SiHCl3)
in a "fluid bed" reactor via the reaction: |
|
| |
| |
|
| |
|
|
 |
This reaction (helped by a catalyst) takes place at around 300 oC. The resulting
Trichlorosilane is a liquid with a boiling point of 31.8 oC, already
much purer than the raw MG Si but still too dirty. |
|
 |
Second, the SiHCl3 is distilled (like
wodka), resulting in extremely pure Trichlorosilane. |
|
 |
Third, high-purity Si is produced
by the Siemens process or, to use its modern name, by a "Chemical Vapor Deposition" (CVD) process as detailed below |
| |
|
| |
Producing Doped Poly-Silicon |
 |
Typically,
we do not produce high-purity Si from SiHCl3 but "doped" polycrystalline solid silicon.
"Doping" is the name for putting well-defined
small amounts of special impurities into a crystal. We dope the poly-Si
(not to be confused with the poly-Si layers on chips) with either boron (B), phosphorous (P) or arsenic
(As), attempting doping atom concentrations of 1014 cm–3 - 1019 cm–3.
The doped poly-Si made this way will be used later to grow single
Si crystals. On occasion we do not dope the poly-Si because we want a Si crystal as perfect and clean as possible
- the "nirvana" silicon! |
|
 |
In principle, we have a vessel which can be evacuated and that contains an "U"
shaped arrangements of slim Si
rods that can be heated from an outside heating source. The Si rods become conductive as soon as the
temperature is high enough (roughly 1000 oC). By passing an electrical current through them, heating can
now be done electrically and the outside heating is switched off. That's important because we want only
the Si rods to be hot, not the rest. |
|
| |
| |
 | | Poly-Silicon reactor ("Siemens process") |
|
| |
|
|
 |
After the vessel has been evacuated and the Si rods are at reaction temperature, an
optimized mix of pure SiHCl3
(Trichlorosilane), H2 and doping gases like arsine AsH3
or phosphine PH3 are admitted into the reactor.
In order to keep the pressure constant (at a typical
value of some mbar), the reaction products (and unreacted gases) are pumped out at a suitable pace. |
|
 |
On hot surfaces - if everything is right this will only be the Si - a chemical reaction now takes place, reducing the SiHCl3
to Si and forming HCl (hydrochloric acid) as a new compound: |
| | |
| |
| SiHCl3 + H2 + "heat" Þ
Si + 3 HCl |
|
|
| |
|
|
 |
Similar reactions provide very small but precisely measured amounts of As,
P or B from the doping gases that will be incorporated into the growing polysilicon |
|
 |
The Si formed will adhere to the Si already present - the thin rods will grow
as fresh Si is produced. The incorporation of the dopants will produce doped polysilicon.
|
 |
As far as Materials Engineers are concerned, this is a simple process. It even
has a name: Siemens process because the process (and the use of silicon as semiconductor)
was pioneered by Siemens company.
Nowadays we call all those processes "Chemical
Vapor Deposition" (CVD) since similar processes are used a lot in microtechnology for depositing some material on a
substrate. CVD processes are also used to make titanium (Ti) and other elements.
It is not all that easy, however,
to work with CVD. Consider the complications just for silicon: |
|
 |
You have to keep the Si
ultrapure - all materials (including the gases) must be specially selected. |
|
 |
The chemistry is extremely dangerous. AsH3 and PH3, a
must for doping (no alternative), are among the most poisonous substances known to mankind. PH3 was actually
used as a toxic gas in world war II with disastrous effects. H2 and SiHCl3 are
easily combustible if not outright explosive, and hydrochloric acid HCl (in gaseous form) is even more dangerous
than the liquid acid and extremely corrosive. Handling these chemicals, including the safe and environmentally sound disposal,
is neither easy nor cheap. |
|
 |
Precise control is not easy either. While the flux of H2 may be in the 100
liter/min range, the dopant gases only require ml/min. All flow values must be precisely controlled and, moreover,
the mix must be homogeneous at the Si where the reaction takes place. |
|
 |
The process is slow (about 1 kg/hr) and therefore expensive.
You want to make sure that your hyperpure (and therefore expensive) gases are completely consumed in the reaction and not
wasted in the exhaust - but you also want high throughput and good homogeneity; essentially conflicting requirements. There
is a large amount of optimization required! |
|
 |
And from somewhere you need the slim rods - already with the right doping. |
 |
Still, it works and about 50.000 tons of poly-Si
are produced at present (2011), most of which now goes to the solar cell industry. |
|
 |
The electronic grade poly-Si that results is
not cheap, however, and will never become dirt cheap. With somewhat relaxed specifications for solar cell uses, it can be
produced relatively cheaply, though. |
|
 |
Here is an example for the poly-crystalline rods produced in the Siemens process: |
| |
| |
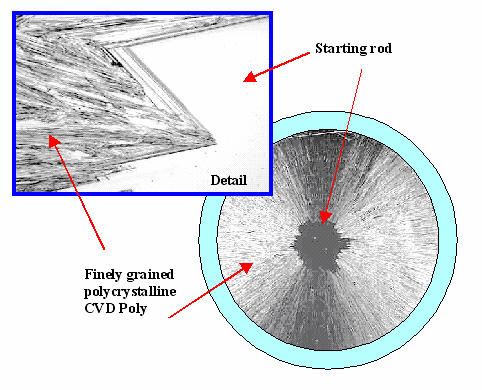 |
| A close view of the poly silicon produced in a "Siemens reactor" |
|
|
|
| |
Silicon Crystal Growth and Wafer Production |
| |
Single Crystal
Growth |
 |
We now have hyperpure poly-Si, already doped to the desired level, and
the next step must be to convert it to a single crystal. There are essentially two methods
for crystal growth used in this case: |
|
 |
Czochralski or crucible
grown crystals (CZ crystals). |
|
 |
Float zone or FZ crystals. |
 |
The latter method produces crystals with the highest purity, but is not easily
used at large diameters. 150 mm crystals are already quite difficult to make and nobody so far has made a 300
mm crystal this way. |
|
 |
Float zone crystal growth, while the main method at the beginning of the Si
age, is now only used for some specialities and therefore will not be discussed here.
The Czochralski method, invented by the Polish scientist J.
Czochralski in 1916, is the method of choice for high-volume production of Si single crystals
of exceptional quality. Below is a schematic drawing of a crystal growth apparatus (about three stories high) employing
the Czochralski method. The article gives an idea about the history and the present state of the art of Si crystal growth |
|
|
| |
|
|
|
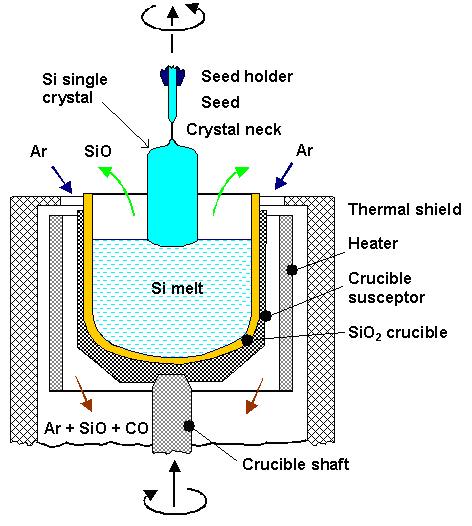 | | The Czochralski method for (silicon) crystal growth |
|
| |
|
|
 |
Essentially, a crystal is "pulled" out of a vessel containing liquid
Si by dipping a seed crystal into the liquid which is subsequently slowly withdrawn at a surface temperature
of the melt just above the melting point. A crucial step for making dislocation-free silicon is to produce a "neck",
i.e. a very thin region in the seed crystal before enlarging the diameter to the desired value. This can be seen nicely
in this module.
The pulling rate
(usually a few mm/min) and the temperature profile determines the crystal diameter.
The basic problem is to get rid of the heat of crystallization.
Everything else determines the quality and homogeneity
- crystal growing is still as much an art as a science! |
 |
Here we only look cursorily at one major point, the segregation coefficient
kseg
of impurity atoms. The full story can be found here. Segregation,
by he way, is also the key process for making wootz blades. |
|
 |
The segregation coefficient in thermodynamic equilibrium gives the relation between
the concentration of impurity atoms in the growing crystal and that of the melt. It is usually much lower than 1
because impurity atoms "prefer" to stay in the melt. This can be seen from the liquidus and solidus lines in the
respective phase diagrams.
In other words, the solubility of impurity atoms in the
melt is larger than in the solid.
"Equilibrium" refers to a growth speed of 0 mm/min or, more practically,
very low growth rates. For finite growth rates, kseg becomes a function of the growth rate (called kseff)
and approximates 1 for high growth rates (whatever comes to the rapidly moving interface gets incorporated). |
 |
This has a positive and a negative side to it: |
|
 |
On the positive side, the upper part of a crystal will be cleaner
than the liquid, crystal growing is simultaneously a purification method. Always provided that we discard the lower part
of the crystal, where all the impurities are now concentrated. After all, what was in the melt must be in the solid after
solidification - only the distribution may now be different.
This defines the negative side: The distribution of impurities - and
that includes the doping elements and oxygen - will change along the length of a crystal
- a homogeneous doping etc. is difficult to achieve.
That segregation can be a large effect with a sensitive dependence
on the growth rate is shown below for the possible doping elements; the segregation coefficients of the unwanted impurities
is given in a table. |
|
| |
| |
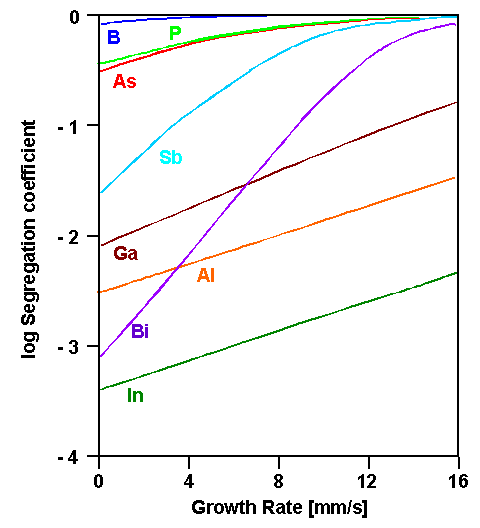 |
| Effective segregation coefficients as function of the growth velocity |
|
| | |
|
| | |
| Atom |
Cu |
Ag |
Au |
C |
Ge |
| kseg |
4 · 10–4 |
1 · 10–6 |
2,5 · 10–5 |
6 · 10–2 |
3,3 · 10–2 |
| Atom |
O |
S |
Mn |
Fe |
Co |
| kseg |
1,25 |
1 · 10–5 |
1 · 10–5 |
8 · 10–6 |
8 · 10–6 |
|
| | |
|
|
 |
We recognize one reason why practically only
As, P, and B is used for doping! Their effective segregation coefficient is still close to 1
which assures half-way homogeneous distribution during crystal growth. Achieving homogeneous doping with Bi, on the
other hand, would be exceedingly difficult or just impossible. |
 |
Present day single crystals of silicon are the most perfect objects on this side
of Pluto! Remember that perfection can be measured by using the second
law of thermodynamics, so this is not an empty statement! |
|
 |
What the finished crystal looks like can be seen in the link.
What we cannot see is that there is no other crystal of a different material that even comes close in size and perfection.
Our silicon crystal is even rather homogeneous. The concentration of the doping atoms (and possibly interstitial oxygen)
is radially and laterally rather constant, a feat not easily achieved. |
|
 |
Our crystal does not contain dislocations
- a unique feature that only could be matched by (really expensive) germanium (Ge) crystals at appreciable sizes (which
nobody grows or needs).
It also does not contain many impurity atoms. With the exception of the doping atoms (and possible
interstitial oxygen, which often is wanted in a concentration of about 30 ppm), substitutional and interstitial impurities
are well below ppb if not ppt level (except for relatively harmless carbon at about 1 ppm) - unmatched
by most other "high purity" materials. |
|
 |
Our crystal is as close to nirvana as a crystal could be. However, it still must
contain the point defects that must have been there in thermal equilibrium right after solidification because they had nowhere
to go during cooling down.
The story going with that is found in this link. |
| |
© H. Föll
© H. Föll (Iron, Steel and Swords script)




