| |
5.2.1 Point Defects |
| | 5.2.1 The Gang of Four
|
 |
We start with zero-dimensional defects;
the smallest denizens of the defect zoo. Here they are - all four of them: |
 |
| The gang of four. |
|
 |
It's pretty simple. More than those four "point
defects" we cannot make by playing with atoms inside an elemental crystal,
meaning a crystal made from just one of the elements like iron or silicon.
The question we need to answer now is "why" and "how many"?
Why should a crystal contain those defects and if there is some reason, how
many should it be?
|
 |
Well, we already know that any crystal can and will make
vacancies because it needs to produce disorder to achieve nirvana. If you glimpse
ahead a little bit, you can see how the crystal makes vacancies. |
|
 |
For exactly the same reasons —creating increasing disorder with increasing
temperature—it also can and will make "self-interstitials",
atoms of its own kind that squeeze themselves into the interstices or interstitial positions
in the lattice. |
 |
The first question thus finds an easy answer for two of the four point defects.
We are left with figuring out how many vacancies and self-interstitials we must have
at some temperature. |
|
 |
Disorder is more important at high temperatures. This can only mean that the
number or the concentration of both vacancies and self-interstitials must
increase with increasing temperature. If you bothered to look at the science
module about vacancies, you know that the concentration increases exponentially.
If you didn't, you may want to check the "science" module to get some idea what "exponential
growth" means.
Just a hint: If you hate exponentials and logarithms but like money, you should reconsider.
It's those dreaded math objects that will tell you how your money will go down the drain with the coming inflation (or through
the roof if you invest it wisely). | |
|
|
 |
OK—for those of you who can't overcome their fear of math: Exponential growth
of the vacancy concentration with temperature means that the concentration goes up rapidly
with temperature. Here is a calculated curve showing the approximate vacancy concentration in iron as a function of temperature: |
| |
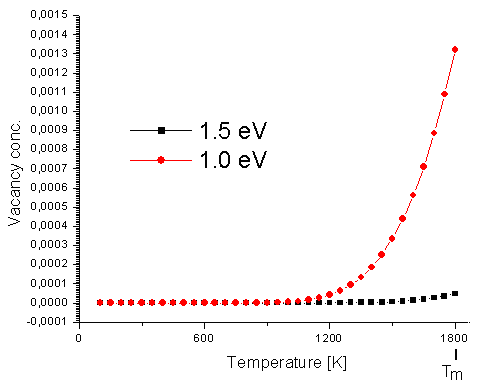 |
Approximate vacancy concentration in iron as function of
(absolute) temperature. Tm=melting point. |
In order to calculate curves like this one needs to know a parameter called "vacancy formation energy" that is not all that well known for iron. That's
why there are two curves for the two values shown. The "real" curve is probably somewhere in between.
0.01
would be 1 %; 0.0001 is 1/100 of 1% or 100 ppm |
|
|
 |
So, yes, the concentration is indistinguishable from zero over most of the temperature
regime. Only at rather high temperatures it goes up rapidly. That doesn't mean that there is nothing at lower temperature,
you just can't see it in this plot. If you plot the total earnings in your country versus time, your salary of 2.654 €
(or of whatever you currency might be) this July will simply not show either—even so it was very
meaningful to you.
In the science module you can look up
how to make better plots of vacancy concentration. |
 |
OK—now you know more about vacancies than you ever fancied in your wildest
dreams. How about self-interstitials? |
|
 |
It is exactly the same thing. Self-interstitials do the same thing as vacancies:
they introduce disorder and thus raise the entropy. They just don't do it quite as well as vacancies in almost all common
crystals.
Most crystals, including all metals, therefore prefer vacancies to self-interstitials
by far, and only make a tiny amount of self-interstitials that we can simply forget
about.
Iron self–interstitials thus are completely unimportant to sword making and we won't mention them ever
again (except in the science module). |
|
 |
The big exception, by the way, is Silicon
(Si). In silicon crystals the self-interstitial is of major importance. How that was found out and drummed into the heads
of the non-believers, would be a long story of its own. I'd love to tell it because I had a hand in it. I will restrain
myself here—but not in this link. |
 |
The vacancy and the self-interstitial are the two zero-dimensional defects that
we subsume under "intrinsic point defects" for obvious reasons. "Intrinsic" is one of those fancy Latin words we scientists like to use in order to show
off. It means "innate", or "incarnate" in the sense that the crystal needs nothing from the outside
for making them—in contrast to the extrinsic (extraneous, foreign) point
defects shown on the right in the figure above.
So let's look at the two extrinsic
guys now, some foreign atom in an interstitial position, or some foreign
atom at an substitutional position.
We also call them impurity
or dirt atoms, and sometimes doping atoms or alloying elements, I'll come back to that. |
|
 |
I know. Those names suck. If you can come up with better ones, let me know. And
while you are at it, find better names for other outlandish expressions like: democracy, republicans, maitre'de, caucus,
anonymous, dermatologists or ophthalmologist, too. Or simply accept that it's not worth the bother to rename things that
already have good working names, however weird. All our intrinsic / extrinsic / interstitial / substitutional stuff is reasonably
clear and you got to have some names, after all.
If you could live with things having names like "George Walker
Bush", you shouldn't have a problem with dirt going by the name "substitutional foreign atom" either. |
| |
As far as dirt is concerned, foremost on our mind is of course Berlusco
carbon in iron. Carbon, it turns out, lives as an extrinsic interstitial
in between the iron atoms as long as it is "dissolved", meaning incorporated
as a single atom.
In silicon, carbon would
be a substitutional foreign atom, replacing ("substituting") one of the silicon
atoms, so it depends on the host crystal which of the two possibilities a given foreign atom will assume.
Let's summarize: |
| |
|
 |
When we talk about carbon in iron, we usually consider concentrations between
0 % and 1,5 %. Those are always
weight percent (written wt % if you want to be
precise) if not otherwise stated (remember?). |
|
 |
This makes a lot of sense in material engineering
because you can easily make desired mixes by weighing the ingredients.
In materials
science, however, we look at atoms. In order to
assess foreign atoms in a host crystal, we need their concentration in
atom percent (at %). In other words, we want to know how many percent
of all the atoms present are carbon atoms?
There is no easy way to switch from one to the other; consult the link for
how it's done. | |
|
| |
 |
If we take, for example, the "magical"
6.7 wt % of carbon and express it in atom percent, we get 6.7 wt %=25 at %. A quarter
of all atoms then are carbon atoms. Why 6.7 wt % of carbon of carbon is "magical" we will see
later—but you are welcome to make a guess, now that you know it corresponds to a mix of iron : carbon=3 : 1 in
terms of atoms. Hint: one could write that Fe3C. | |
 |
You are looking at a virtual cut through the crystal, of course, and at any plane
inside the crystal it would look like this, just with the carbon atoms distributed at random at interstitial positions.
We also call this "the carbon is dissolved" or it is in solid
solution.
Now why should that be virtual? I stated some time ago that there are special microscopes
that allow you to look at the surface with atomic resolution. Yes—but:
If we look at a real
surface of almost anything you wouldn't see much because a real surface of almost all crystals in air is oxidized.
With an iron sample you always would look at "rust", even if to your eye it looks bright an shiny. An iron oxide
layer just a few atoms thick, totally invisible to the eye, is all it takes to completely obscure a surface from atomic
"view". |
|
 |
Even if we use the common (and expensive) technique of putting our sample in ultra-high
vacuum (UHV) where
it can't oxidize, and "somehow" take off the original rust layer in there, we would still have a hard time seeing
anything! Why? Because iron is magnetic!
That tends to mess up many microscopes, including the STM we would need to employ here. I have therefore no iron surface
pictures taken at atomic resolution that looks remotely like the schematic picture above. The best I can give you are pictures of point defects in a gallium arsenide (GaAs) surface. |
 |
Note that despite the rather small weight percent
concentrations of about 0.3 wt% or 1 wt%, respectively, in the figures above, it's actually quite
a bit of carbon if you think about it. Nevertheless, most "bonding springs" still connect iron atoms
and thus Young's modulus is hardly changed
relative to that of pure iron.
Note that this not a theoretical prediction open
to some doubt. This is as hard a fact as you like them to come. |
 |
Now that we have played around a bit with an extrinsic
defect like carbon in iron, the same question as above for the intrinsic defects comes
up:
Why should a crystal contain those extrinsic defects,
and if there is some reason, how many should it be? |
|
 |
In an ideal
crystal world the answers to those questions would be exactly the same as for
intrinsic point defects:
- Extrinsic defects increase disorder, and a certain number of all the other 90 or
so elements should be part of any nirvana-seeking crystal.
- The proper "nirvana" number of extrinsic defects depends, among other
things, strongly on the temperature. Generally, the concentration should increase with increasing temperature.
|
|
 |
In an ideal world you
would have the proper amount of money necessary for achieving your personal nirvana conditions. But neither you nor the
crystal exist in an ideal world. A crystal cannot make foreign atoms at will, just as
(most of) us cannot print money all that easily. |
|
 |
So in the real
crystal world, the answer to these questions is different from the one describing the ideal
word. It is extremely simple but will have formidable consequences: |
| |
|
|
 |
Tough luck. The number of any atoms forming
extrinsic point defects—interstitial carbon in iron, substitutional carbon in silicon, substitutional phosphorous in
iron, ..., (the list here would contain about (90 · 90)=8.100 entries; can you see why?)—is whatever
it is. Mostly that number simply comes from the original process used for making the
material.
The crystal can neither make more foreign atoms or impurities
if it wants too, nor can it easily get rid of the ones it has. It needs a little bit
of help from friends like you for this task
The consequences are dramatic: |
| |
If a crystal contains impurities in any
form, it can no longer achieve
absolute nirvana. Never. Ever.
|
|
 |
Our steel blade or crystal with 0,5 wt% carbon in it, or just any crystal containing
impurities, is stuck now. It wants to have a certain precise number for nirvana but chances are extremely high that the
number it actually has is far off that nirvana number.
The crystal is stuck. What is it going to do? |
|
 |
Exactly what you would do if your 9 children are running all
about the place, producing far more disorder than needed, and keeping you from achieving you preferred state of being (easy
chair, peace and quiet, good wine, a good book like this one). You round 'em up and lock them into a room somewhere else.
Crystals do the same thing; they imprison their impurity atoms or, as it is properly called, precipitate
them. We will see how that works shortly. But before we do that I want to generalize a bit. |
 |
It should be clear that as far as extrinsic point defects go, we are not restricted
to carbon. I can take whatever else the periodic table has to offer (about 90 elements) and put it into my iron. I know
how to do it in reality, for you it's an easy thing to do in your brain lab.
Now let's look at how these impurities
have settled in the iron. The good news is that the foreign atoms can assume only three
different configurations: |
|
|
- They could be dissolved as interstitial impurity
atom. Then they are somehow wedged in between the regular atoms.
- They could be dissolved as a substitutional
impurity atoms. Then they "substitute", i.e. replace an iron atom in the crystal lattice.
- Only a few are dissolved in one of the two possible ways and and the rest is securely put away in a precipitate (I'll come to that shortly).
|
|
 |
Let's look at few examples
- Carbon (C), nitrogen (N), and hydrogen (H) are always dissolved as interstitial impurity atoms in iron.
- Phosphorus (P), Sulfur (S), Manganese (Mn), Nickel (Ni), Chromium (Cr), Vanadium (V) and most other atoms are always
dissolved as substitutional impurity atoms in iron.
- Oxygen (O) and boron (B) can be dissolved in both ways.
- All of them will be imprisoned in precipitates if they exceed a certain specific concentration.
|
| |
 |
Which elements we have inside the iron crystal,
how many, and in what kind of configuration determines
most of the important properties of your steel. |
 |
So, without a little bit of help, the concentration of extrinsic
point defects—impurities— in crystals won't change, in contrast to the intrinsic
point defects that the crystals can make and dispose of on its own.
This is great because that's where you and I come
in. If you know how to do it, you can change the impurity concentration of a given crystal, no matter if the crystal likes
it or not. That's where technology starts. Let's look at two examples: |
|
 |
Building a silicon microprocessor or most anything else from silicon
(Si) needs to start with a very clean and perfect silicon crystal, as close to nirvana as it can be. This involves producing
extremely clean liquid silicon first. In a second step an almost perfect crystal is grown by rather involved methods.
Then we mess up the crystal by putting substitutional impurities like phosphorous (P),
arsenic (As), or boron (B) inside well defined parts of it.
So the concentration of extrinsic point defects does change:
it goes up. The crystal doesn't like it, of course, because that takes it rudely off his near-nirvana state of being. It
fights back in many ways, and it fights dirty! That is one of the reasons why microelectronics technology is not a simple enterprise.
| |
|
|
 |
When we make steel in modern times, we start with rather dirty liquid iron, it
is actually cast-iron. We remove most of the unwanted
dirt already in the liquid state and we add some wanted dirt. Crystallization is not controlled on a microscale so we get
(huge) polycrystals. Certain processes in the solid crystalline state (like "case hardening") will change the
concentration of some impurities (mostly carbon) to some extent. |
 |
One last word to impurities in crystals. Without going into more details, one
thing becomes clear: it is far easier to mess up a crystal then to keep it clean or to clean it up if its messy. It's not
so difficult to get impurities inside. Crystals, after all, like to have some at high temperatures. It is just far more
difficult or well-nigh impossible to get them out again. |
|
 |
It is just the reverse of opening a can of worms, or squeezing tooth paste or
your heir out of a tube. The problem there is to get the stuff back in. |
| | |
|
© H. Föll (Iron, Steel and Swords script)