|
Making Vacancies |
| |
Calculating the Equilibrium Vacancy Concentration from
Scratch |
 |
How many vacancies
does a real crystal need for achieving nirvana?— excuse me, here in the science section we call it
thermodynamic equilibrium. It is about the most simple thing one can calculate
"from scratch" by using the second law. |
|
 |
First we need to determine the free energy—excuse me once
more. Among purists it's called free enthalpy but that is just a different word for the
same thing. What matters is the mathematical definition:
G(N, n, T) is the free enthalpy of a crystal with the variables
N
= number of atoms forming the crystal.
n = number of vacancies in the crystal.
T = (absolute, of course) temperature of the crystal.
I will give you the
full function G(N, n, T) first and then discuss it. What we have is |
| |
|
|
|
| G(N, n, T) |
= |
G(N, T) |
+ |
n · EF |
– |
TS(N, n) |
| | | 1stterm |
| 2ndterm | |
3rdterm |
|
|
| | |
|
|
 |
The first term, G(N, T)
, simply describes the total free enthalpy of a perfect crystal with N
atoms at the temperature T and no vacancies. It contains the energy Ecryst of the
crystal, essentially contained in the vibrations of the atoms, and the entropy Svib resulting from
that.
If you like you can write it out as G(N, T) = Ecryst(N, T)
– TSvib(N, T) but that will just make the equation longer.
For any number
of atoms N and temperature T, the function G(N, T) would produce
some number if we put our mind to it. We won't do that, however, since it will turn out that we don't really need G(N,
T) |
|
 |
The second term acknowledges that we expect to have n
vacancies in our crystal.
Making a vacancy requires that we sever the bonds between the atoms an and rip out the cut-off
atom somehow. That will take some effort or energy.
The energy needed to make one vacancy in a given crystal is always the same, no matter how "we" make the vacancy.
It is aptly named formation energy of a vacancy and abbreviated with
EF.
We want to make n vacancies so we need to add
n · EF to the energy contained in the otherwise perfect crystal. |
|
 |
The third term acknowledges that having vacancies increases
the disorder and thus the entropy S of the crystal. This is called the "entropy
of mixing" since it comes exclusively from mixing up "things" and not from the "things"
itself.
We thus must subtract TS from the total energy of the situation. The entropy depends only on the number of atoms N and the number of vacancies n so we
can write it S(N, n). |
 |
The third term with the entropy S(N, n) is the decisive
term. Boltzmann's famous formula for the entropy, S = k · lnP,
applies. We have |
| | |
|
| |
| S(N, n) |
= |
k · ln {P(N, n)} |
|
|
| | |
|
|
 |
P(N, n) is the number of possibilities to arrange n
vacancies in a crystal consisting of N atoms; k = Boltzmann's constant. And of course, you know what
"ln", the symbol for a natural logarithm means
|
 |
How large is P(N, n)? You might also ask: In how many
ways can I distribute 7 socks in my bedroom, assuming I have 42 places (in the actual sock drawer, in any
of the 11 other drawers, under the bed, on the bed, ....). |
|
 |
Question like that are answered by combinatorics,
a much-hated branch of stochastic math. Another combinatorial question (but not mathematically
equivalent) could be "How many different car license plates can you make by using a combination of two letters and
three numbers" or "How many ways to dress differently with two pairs of pants, three shirts, five pairs of socks",
or "how many different hands are there at poker", or... You get the idea. |
|
 |
The answer to our question is so elementary (and simple) that the combinatoric folks came
up with a special mathematical symbol, the binomial coefficient, to abbreviate
equation writing. We have |
| |
| |
| |
| P(N, n) |
= |
æ
è |
N
n |
ö
ø |
= | N!
n! · (N – n)! |
æ
è |
N
n |
ö
ø |
= Binomial coefficient of N over n |
|
|
| | |
|
|
 |
In case you forgot: An exclamation mark behind a mathematical symbol or number means the factorial of that number.
A factorial is an extremely simple thing: n!
= 1 · 2 · 3 · 4 · 5 · ..... · n. Take 5! as example. We have 5!
= 1 · 2 · 3 · 4 · 5 = 96. Nothing to it? Just wait. |
 |
Now we are done. The full expression for the free enthalpy G(N,
n, T) of a crystal consisting of N atoms that contains n vacancies at the
temperature T is |
|
|
|
|
| G(N, n, T) |
= |
Ecryst(N) – TSvib(N) |
+ |
n · EF |
– |
kT · ln |
N!
n! · (N – n)! |
|
|
| | |
|
 |
We are seeking nirvana for a crystal of given size (i.e. given and constant N)
at some given and constant temperature T and that means we are seeking the minimum value of that function.
The only variable left is n, the number of vacancies. We now need to determine
the minimum of the function G(N, n, T) with respect
to the number of vacancies n. |
|
 |
Obviously we have to differentiate G(N, n, T) with respect
to n and set it to zero: dG(N, n, T)/dn = 0. The first constant
term disappears if we do that (that's why we didn't need to worry about it) and we get |
| | |
|
|
|
dG(N, n, T)
dn |
= |
EF |
– |
kT |
d
dn |
æ
ç
è
| ln |
N!
n! · (N – n)! |
ö
÷
ø |
= 0 |
|
|
| | |
 |
Oh Sh...!
If you had an extremely good
high school education in the US, or an average one in Germany, you know how to differentiate
a natural logarithm:
y = lnx; dy/dx = 1/x.
But only with an extremely good high school education
in Germany you know how to differentiate a factorial function:
y = x!; dy/dx = can't be done!
It can't be done! It's not a smooth
function. |
|
 |
Thank God for Stirling's
formula that makes an approximate smooth function out of factorials for large
x: |
| |
| |
| |
|
| | |
|
|
|
Since we have a hell of a lot of atoms (if you can see a crystal it has more than about 1015
atoms) and therefore also a lot of vacancies, Stirling's formula is extremely precise for our undertaking.
We now replace
all the factorials by Stirling's expression, do the differentiation (lengthy but simple), use the fact that n
<< N, i.e. we have way fewer vacancies than atoms, and and the extremely simple
final result emerges: |
| |
| |
| |
n
N |
= |
concentration
of vacancies |
= |
exp – |
EF
kT |
|
|
| | |
|
 |
To be honest, it is a bit too simple. In full generality our basic equation for the equilibrium or nirvana concentration
cPD of all point defects we have: |
| |
|
|
|
|
| |
|
|
 |
The only difference is that we now have a pre-exponential
factor
c0 that takes into account that there is a bit more to the entropy than just the entropy of mixing atoms and vacancies. For example, a single vacancy, or just about any point
defect, also carries some direct entropy around, since it disturbs the arrangement and
in particular the vibration spectra of its neighbors and thus increases the local disorder.
The pre-exponential factor
c0 carries a lot of information about what is going on in detail. However, its numerical value
is typically around 1 - 5, which means it doesn't matter much as long as we just look at the basics of what is going
on. |
 |
Puhhh! Quite a bit of work. And I didn't even run through the details. |
|
 |
The result, however, is of supreme beauty. We have derived the most important mathematical
expression in sword making.
No, it's not the vacancy concentration, it is the exp–(E/kT)
term known as Boltzmann factor. The link takes you there. Here
we look at bit more closely at what we can learn from our vacancy concentration formula. |
| | |
|
Discussion and Generalization |
 |
The first thing to realize is that we calculated far more than just the vacancy
equilibrium concentration. We can use this basic equation also to calculate, for example: |
|
 |
The self-interstitial
concentration. Everything is pretty much the same, except that the quantity EF now would be the
formation energy of one self-interstitial. In most cases this energy is somewhat larger than the formation energy of vacancies,
meaning that we have a lot less interstitials than vacancies in equilibrium. That's what exponential
functions will do. |
|
 |
The di-vacancy concentration, i.e. the concentration of vacancy
pairs. EF is now the formation energy of a di-vacancy, which we can safely
assume to be twice the formation energy of a single vacancy minus some binding energy.
|
|
 |
The equilibrium concentration of foreign
atoms - interstitial or substitutional. Same procedure once more (eschewing some details that really don't matter much).
The energy in the exponent then is called "solution energy" and tells you
how much a crystal likes or hates to have a foreign atom sitting around. If the solution energy would be zero, the crystal
doesn't care - the foreign atom is just as good as one of the own kind. Positive values mean the crystal does not like the
foreign atom, negative values (possible in this case) that it actually prefers a bit of change.
You realize, of course,
that the equilibrium concentration value of foreign atoms depends exponentially on the
temperature, while the real value is more or less constant. You can't easily change
a given concentration of foreign atoms, after all. |
|
 |
In essence the major difference between point defects is only how much energy E
- formation energy, solution energy, whatever - they carry around with them. |
 |
The basic equation for the equilibrium concentration of all those point defects
always contains the "Boltzmann factor" exp–(E/kt).
The link will show that the vacancy concentration equation derived here is just a special case of a much more general principle
around the Boltzmann factor. |
 |
We now need a number for EF so we can evaluate our basic
equations for the concentration of point defects. So are we now going to calculate formation or solution energies for all
kinds of point defects? |
|
 |
No, we won't. It simply can't be done with pencil and paper. Just about now, it might be possible
to get reliable numbers with the biggest computers around. It's not that we don't know how to do it; it's just too much
number crunching. Since EF comes up in an exponent, we need a rather precise number in order to
avoid big uncertainties.
So far we actually do experiments and measure those numbers,
as far as possible (which is not very far). It's not easy and we don't have as many reliable numbers as we would like.
Here are some examples: |
| |
| |
| |
| Crystal |
HF(V) [eV] |
HF(i) [eV] |
| Ag | 1,09 |
Sufficiently larger than HF to make concentrations negligible
| | Al | 0,66 |
| Au | 0,94 |
| Cu | 1,17 |
| Fe | 1.4 |
| Ni | 1.7 |
| Mo | 3.0 |
| Pb | 0.49 |
| Pt | 1.49 |
| W | 4.0 |
| Si |
? 2,0 - 4,5
Still debated |
? Small enough to make concentrations count. |
| Data adopted from: Helmut Mehrer, "Diffusion in Solids", Springer, 2007.
|
|
| |
| |
|
 |
As a rule of thumb, "normal" metals have vacancy formation energies HF(V)
roughly around 1 eV, and self-interstitial formation energies HF(i) a bit larger, say 1,5
eV. No reliable numbers are known for self-interstitials. However, if their formation energies are just a few tenth
of an eV larger than those of vacancies, the interstitial concentrations will be far smaller (factor 100 or more)
than the vacancy concentration. In this case some interstitials will be around but are neither of any importance for diffusion
nor experimentally accessible.
Another rule of thumb asserts that the maximum vacancy concentration close to the melting
point is around 10–4 |
 |
Now let's look at some graphs, representing the point defect concentration. |
| |
|
|
|
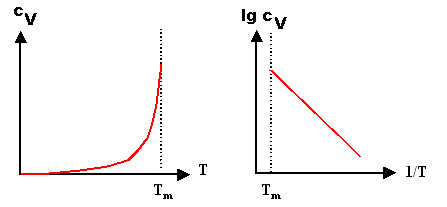 | Direct representation
cv = exp–(EF/kT) |
Arrhenius representation
lg(cv) = –(EF/k) · 1/T |
|
| |
|
|
 |
A direct plot is not very helpful. Except for temperatures close to the melting point, the
curve is indistinguishable from the T-axis. That's why we mostly plot equations with exponentials
in a so-called Arrhenius
plot. We simply take as new parameter the logarithm
of the concentration, lg(cV), and the reciprocal value of the absolute temperature, 1/T.
If we call them "y" and "x", respectively, the equation reads y
= –(EF/k) · x and that is the equation of a straight line with slope –(EF/k). |
 |
Now we interpret the equation as giving the equilibrium concentration
for some foreign atom in some crystal, e.g. carbon in bcc iron. Then we typically plot
it directly, but with the axes' switched as shown below: |
| |
| |
| |
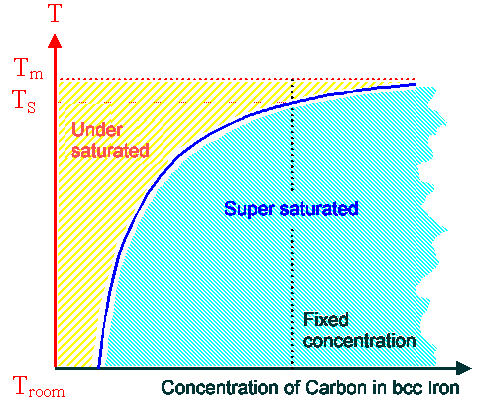 |
| Phase diagram plot of foreign atom concentration |
|
| | |
|
|
 |
The dotted line indicates some fixed concentration. Only concentrations on
the blue line are the proper nirvana or equilibrium concentrations for the extrinsic point defect "carbon in iron" at the temperature T. Since the concentration
of foreign atoms typically can't change but has some fixed value, any point in the yellow region signifies that the crystal
is undersaturated, i.e. has less foreign atoms dissolved than necessary for nirvana.
There is absolutely nothing the crystal can do about undersaturations of extrinsic point defects, in contrast to the
intrinsic point defects that it can
make (or remove) "at will".
In the blue region, the extrinsic point defect is supersaturated;
there are too many. The crystal can and will do something about that: it can imprison the surplus atoms in a precipitate
and thus remove them. What exactly a crystal does about supersaturations of extrinsic point defects is at the very core
of steel technology (and about any other material-based technology). |
 |
Hey, I'm just about to invent phase diagrams
and thus get ahead of myself. So let's stop here. You might come back after you went through chapter
6. |
| | |
|
© H. Föll (Iron, Steel and Swords script)

