 |
Vacancies are extremely important
since moving atoms is a big deal, just wait. The major reason why we heat up our iron and steel a lot during sword making
(or Si for making microchips, or most everything else for making most everything else; excluding babies), is the need to make vacancies so we can move atoms around.
|
|
 |
It's a good bet than whenever a (crystalline) material is heated up to a temperature
higher than, let's say, half of its melting point (as measured in Kelvin), you do it because you need vacancies. By heating
you produce vacancies. It's unavoidable, the second law will see to that.
My buddies
and I, by the way, know exactly how many vacancies you will produce in a given material
at a given temperature.
It's quite easy to calculate (with a bit of calculus, logarithms and combinatorics, involving
factorials), the link shows how. Even so the math may look a bit involved, it is about the most simple calculation you can
do when invoking the second law with Boltzmann's super-famous formula.
Calculating vacancy concentrations is a kind of paradigm for statistical thermodynamics. |
|
|
| |
Please look it up! Like Wagner's
music1), it is better than it appears. |
 |
Now let's deal with that big "why"
question you have on your mind: |
|
|
Why do I need to move atoms around
in order to make a sword?
(or a microprocessor?)
|
|
 |
Because whatever you do to make something, you must
change the way the atoms are arranged in your material. You must rearrange the atoms in your crystal somehow, and for that
you need to move atoms around.
If you don't move atoms around when you try to do
something to your material, it is exactly what it was before you did something to it—so
why bother? That's so important that I will repeat it in huge letters: |
| |
|
|
 |
If you think about that for a moment, you will realize that there aren't so many
ways to move atoms around in a crystal without invoking vacancies. Let's look at some options that may occur to you:
- You can scrape some atoms off the surface of your steel (with a file, for example). The atoms inside then won't even
notice that something has happened. Well - yes, you can do that! And you don't need to invoke vacancies for that. But you
only changed the shape of a material by reducing
its volume in this way, but not its properties.
- You can bang your material with a hammer. The material will crack it if it is brittle or changes its shape without loosing volume if it is ductile.
Changing the shape of a crystal without changing
the number of its atoms can only happen if some atoms are now somewhere else. So plastic deformation
is indeed the other major way to move atoms. We will see presently how that is done without vacancies.
- You might corrode your iron by throwing it in acid or just by letting nature takes its course. It will certainly change.
But that's once more doing things at the surfaces like in item 1, and we already covered that.
- Then you could — Yes? Any suggestions? I bet you are at the end of your tether now because I
am (almost). If you find another way to move atoms around without using vacancies, let
me know. I'm really curious. I will present your idea as my own and get the Nobel prize.
|
 |
I'm going to assert that insight with a big green box: |
| |
Moving atoms in a crystal randomly
is done via vacancies !
|
|
|
 |
Note the "randomly" in the statement
above. Moving atoms via plastic deformation (No. 2 above), from the viewpoint of the afflicted atoms, is not a jolly, free-spirited,
hopping-around-at-random thing but an orderly and forceful process - and that's why we are going to call it "military
movement" later on. |
 |
It is easy to see how the vacancy mechanism of atom movement works. One of the
atoms bordering a vacancy jumps into the empty space that is the vacancy, see the figure above, or the more schematic figure
below. As a result, one atom has moved and is now somewhere else, and so has the vacancy.
|
|
 |
Which one of the neighboring atoms will jump
into the vacancy, and when it's going to do this, is a purely statistical or random event. Randomness always means that you can simulate what is going on by casting dies
and following some simple rules. For example, get a 12-sided die, make the rules like: 1 = atom on top moves, 2 = atoms
on the upper left moves, …) and you now can cast a die to determine which one of the 12
neighbors to a vacancy in an fcc or hcp crystal is going to jump. For a bcc element crystal you need a 8-sided die.
Of course it is far easier and faster to your program your computer to simulate those dies. Just utilize its "random
number" generator. |
|
 |
Again, even so it gets a bit repetitive: we can calculate precisely what will
happen on average. It's not called "statistical
thermodynamics" just for fun; there is an awful lot of chance, randomness, and statistics involved. But "stochastics", to give it its proper name, is a precise mathematical discipline and we
know precisely how often, on average, atoms jump around in their crystal or, same thing,
vacancies jump around in their crystal. |
|
 |
For example, in pure iron
at 900 oC (1652 oF), just below the temperature where it turns into a bcc
crystal, a vacancy will jump about 100 million times per second, while at room temperature
it essentially sits still. That means that at the high temperature about 100 million
atoms per vacancy in there will be somewhere else after just one second. |
 |
The whole process of atoms moving around randomly is called "diffusion".
Diffusion happens naturally all the time in liquids and gases because there the atoms move randomly by definition. It is
the reason why you might smell your Beloved before you see him—the smell "molecules" just move or diffuse
faster than he moves. But diffusion of gases or liquids is not of much interest to us here.
Diffusion in crystals via vacancies is at the root of changing properties of solids,
and that's what we are actually discussing, remember? |
|
 |
Let's look at the importance of diffusion via vacancies in the light of modern
technology. Let's make a microchip and not a sword for a change.
The basic task is to
take a perfect silicon (Si) crystal and to replace a few of its atoms (one out of 10 million, let's say) by Arsenic (As)
atoms in a certain region. That's called "doping" the silicon, and doping is the process at the very heart of microelectronics.
How are you going to do this?
You only can "go in" via the surface, so let's put some arsenic atoms on the surface of the silicon crystal. This
is shown schematically on the right-hand side in the figure below; the arsenic atoms are colored red. |
| |
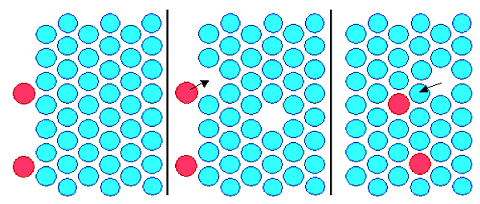 |
| Diffusion and the making of a chip |
|
 |
If you have some arsenic on the surface at room temperature, you're not getting
anywhere. The arsenic atoms would be sitting on the silicon surface forever, give or take a small eternity. |
|
 |
What you are going to do now is to produce some mobile
vacancies as shown in the middle picture. For that you must heat your silicon
to a sufficiently large temperature (at least 800 oC (1472 o). In the picture we have generated two
vacancies. The vacancies move around at random because silicon atoms are jumping into them all the time. Eventually, as
shown in the middle figure, one vacancy happened to move next to an arsenic surface atom, giving it a chance to make a first
jump into the silicon. And so on.
The picture on the right shows the two arsenic atoms now inside the crystal. Most
of the time they are just sitting there, waiting for vacancies to come by. If that happens, they might make another jump,
always provided if they can beat the silicon atoms that are competing for a jump into the vacancy. If they jump, they might
make it deeper into the crystal or back to the surface; it's all equally likely, after all. |
 |
This looks like some truly awkward process for going places. Well, it is—but
it works. It works quite nicely, in fact. |
|
 |
Look into your silicon microprocessor with your brain microscope. The structures
you will find inside the silicon are far more complex then you could imagine in your
wildest dreams - but they were produced mainly by this vacancy diffusion process.
The vacancy diffusion process also
works for iron. It is the cornerstone of iron and steel technology, and of any other "crystal changing" technology. |
 |
By the way, how fast do you think those atoms oscillate? What's your guess?
Back-and-force a few hundred times per second like a tuning
fork? A few thousand, million, billion, … times per second?
Well, you either know or you don't. You can't really
guess that number intelligently. If you don't know it, you must do some calculations.
Since we don't do calculations here, I will tell you. |
|
 |
Those atoms are small and nimble; they actually oscillate with
a frequency of about 1013 Hertz (Hz) or 1013 times
back and forth per second. In other words: 10 billion times per second in Europe, or 10 trillion
times in the USA. By the way, that's about 1 % of the frequency of light if you consider it to be an electromagnetic
wave.
What that means is that all this jumping around can be quite fast. An atom next to a vacancy "tries"
10 trillion times per second to move over to the vacancy position. So even if the probabilities for making that big jump
are very low, it still could happen rather often per second.
All in all, atoms can move around quite
swiftly at high temperatures despite single jumps being very small (about 1 nm; remember?).
|
 |
Let's summarize: |
| |
- A perfect element crystal is an ordered assembly of one kind of atom (for example iron). Its lattice type (bcc for iron
at room temperature) is the one belonging to its "nirvana" state.
- The atoms oscillate around their basic lattice position with a rather high frequency. The energy contained in these
oscillations we call "temperature". When the temperature increases, the amplitude
of the oscillations increases.
- Depending on temperature, a certain number of vacancies
must also be present and their concentration increases sharply (a better word is: exponentially) with increasing temperature.
- Oscillating atoms and vacancies exist because the second law commands with absolute
authority that by increasing temperature, things must become more disorderly; the entropy
must increase. And that is good! Being perfect all the time is boring.
- The vacancies can move around and that provides for movement of the atoms, something we call diffusion.
Diffusion is the key to processing crystalline materials.
|
|
 |
Now read out loud to yourself: |
| |
If my steel changed - from soft to hard or the other way around, whatever - the internal atomic structure
must have changed.
This means: atoms must have moved.
Diffusion via vacancies provides one of the two ways to move atoms in a crystal.
|
|
 |
So far, so good. But we still have a major why
question open that has no obvious relation to vacancies and diffusion: |
| |
Why does pure iron change its
phase from a bcc
crystal to an fcc crystal
at precisely 911 oC (1.672 oF)?
|
|
 |
This question applies equally well to all
elements that change their crystal structure upon heating (or cooling), for example like Cobalt (Co), Strontium (Sr) and
carbon (C) – look up the table with the list to find more. |
|
 |
We might ask just as well why elements like
Ag (silver), Au (gold), Al (aluminum), Cu (copper), Si (silicon), and so on, do not
change their structure at some temperature? |
 |
The answer is simple in principle. In practice it will provide work for generations
of materials scientists to come. Here it is: A crystal of any material can be considered to be an extremely powerful analog
computer that at any instant in time assesses the following items:
- The present situation, including, for example, the actual temperature, pressure and the availability of foreign atoms.
- Its actual structure, including the actual crystal structure, the number of vacancies and other defects we will encounter later.
- The balance between energy and entropy for the present situation and the
actual structure.
- The balance between energy and entropy for the present situation and any conceivable
possible structure.
|
|
 |
If one of the conceivable states is nirvanawise better than the actual state,
the crystal implements all the measures at its disposal to change its present state to that more desirable state. And that
may be a different crystal structure at some changed temperature. |
 |
Of course, the crystal does not really compute. It is mathematically
just as challenged as your average banker or politician; it might just be a little bit ahead when it comes to large numbers.
|
|
 |
The crystal just does whatever it does because every atom simply does what comes naturally, like an ant in an anthill. All the possible structures
are continuously tried out on a very small scale. Some atoms just assume some state, purely at random, and if it "feels
good", it spreads. All possible states always "compete" with each other, and the winner takes all.
Only
if you look at the big picture with regard to crystals or ant colonies, something meaningful
emerges that is not visible on the atom or single ant level. |
|
 |
It's only us curious humans who have to go through an algorithm (in the form of
a computer program) as outlined above to figure out what is going on. The four-step procedure above is simple enough in
principle but outrageously difficult in detail. What are the possible potential structures?
If challenged, you probably could come up with a few—but rest assured that there are far more than you could imagine
in your wildest (Material Science) dreams. The link gives an example
of what previously unimaginable wonders we have already encountered. |
 |
So, to answer the question, it just so happens that in iron, cobalt, and few other
metals it is necessary to change the crystal structure at some temperature for achieving nirvana, while for aluminum gold,
and so on it's not. |
|
 |
Simply speaking, the crystal has one more trump card up his sleeve for balancing
energy and entropy than just adjusting atomic vibration amplitudes and vacancy concentrations. It can also change its basic
crystal lattice structure. Doing that also changes vibrations and vacancy numbers. That makes it a bit difficult for us
to compute if changing the basic structure, meaning to initiate a phase
transition, would be a good move.
Now for the good news. Being engineers, we don't have to compute all that!
The crystal, after all, can and will do that for us. We just have to measure what it
is doing by designing some suitable experiments. |
|
 |
What we will note already here is that the fcc phase that iron assumes at high
temperatures is actually only a little bit closer to nirvana than the bcc phase, while
at low temperatures the bcc phase is a lot better than the fcc phase. |
 |
Come to think of it, we actually know about this already. Pretty much all crystals change their basic
structure at a very special temperature that we call melting point.
This particular phase transition
just looks more dramatic compared to changing from one crystal lattice structure to another one, but in essence it is the
same thing. |
|
 |
If an element crystal undergoes one or more phase transitions before the final
big one called melting, and at what temperature this happens, we know from experiments.
But not only did we measure it in great detail, we can actually (almost) calculate it. Essentially we know how to calculate
it (see above), and we get good approximate numbers. Getting precise numbers is still beyond our ken (but not for much longer)
because you need to do extremely tricky math and that's not only tough but needs more computing power than we command right
now. |
 |
Not many elemental crystals undergo a phase change in the solid state, and very
few do it more than once like iron. Now you have a first indication why iron is just about the most complicated crystal
among the major metals. Its bag of tricks for achieving nirvana is rather larger than that of most other crystals. |
|
 |
Iron is also magnetic in its bcc state—but
not in its fcc state. This provides for a few more tricky ways to play with energy and entropy. Being (ferro) magnetic is
a rare treat among elements. Only nickel (Ni) and cobalt (Co) are ferromagnetic, too! I won't go into this here because
it is not very important for sword making.
There is a science module about magnetism, however.
All things considered,
iron is just a rather special material, already in its ideal form as perfect "brain" crystal. |
|
|
 |
In reality, however, there is no such thing as a perfect
iron crystal in a state of nirvana, utterly at peace with itself and the world. |
|
 |
There are only real iron crystals out there,
not brain crystals, and none of them can ever achieve perfect nirvana, just like you and me. They only can get close. Their
desire to get as close as possible is what makes things happen. |
|
 |
So we now need to look inside a real iron
crystal and see what happens there. |
| | |
|
© H. Föll (Iron, Steel and Swords script)

