| |
5.3 Two-Sided Defects and Bulky Things |
| |
5.3.1 Grain Boundaries |
 |
If I would follow the order put down in the beginning
of this chapter, one-dimensional defects or dislocations would be next. I won't
do that. I will skip the one-dimensional defects for the moment and go right to two-dimensional defects.
|
|
 |
Besides the two-dimensional defect "surface" (which is a bit trivial) we need to look at |
 |
Grain boundaries go first. They are easy to
conceive; we have a lot of them by definition if we look at a poly crystal. Whenever you look at crystals, chances are that you actually look at poly
crystals and thus at grain boundaries - even if you can't see them because your brain microscope is off.
|
|
 |
You don't see grains boundaries, even with you brain microscope on, whenever you look at single crystals.
There aren't all that many of those around, however. What we have are:
- Gemstones like your wife's diamonds and rubies.
- Some special turbine blades inside jet engines.
- Some very special optical components.
- Major semiconductors like silicon (Si), gallium arsenide (GaAs) or gallium nitride (GaN) found inside
electronic products like microchips or LED's.
The latter ones are the most perfect and ubiquitous ones. You must open up a chip if you want to see them. Go ahead,
do it. It is fun, even so you kill the device by doing it. You finally revenge yourself on all those chirping and beeping
gadgets that keep you from drinking beer. Also, it's less dangerous than fiddling around with your wife's diamonds. What
you are going to see is shown (in its original state) in this link |
 |
You have seen many grain boundaries without noticing. Mostly because they are
invisible to the naked eye, but also because you didn't know what you saw. |
|
 |
If you look at almost everything around you that is not glass or biology, you see poly crystals, which, by definition, contains plenty of grain boundaries.
You just don't see them because the surface typically doesn't show the internal crystal structure. Even if it does, chances
are that the grains are so small that without a microscope you can't see them.
Both points hold for your sword blade.
First, it is probably polished,
and that is just another way of saying that there is no visible structure on the surface. Second, the grains are so small
that you couldn't see them anyway, not to mention the boundaries that separate them. |
|
 |
There are exceptions, however. When a puddle freezes over in the more northern
countries you look at frozen water called ice. There you might see grain boundaries as the line separating different large
grains.
Grain boundaries are definitely visible if you look at a lamp post or any other "galvanized" metal object, i.e. metals with a thin coating of zinc (Zn). The zinc grains
can be very large and they are clearly visible, see below. The so-called "multi-crystalline" silicon used for
solar cells has huge grains, too, and also shows the grain structure rather nicely as shown below. |
| |
 |
 |
| Visible poly crystals and grain boundaries |
| Galvanized sheet metal with clearly visible grains of the zinc (Zn) coating. |
Slice of multi-crystalline silicon (Si) used for making solar cells with visible grains of silicon.
|
|
 |
Actually, what you really see are the grains.
You see them because different grains of zinc (Zn), silicon (Si), or whatever, with different crystal orientation, reflect
light differently. In between different grains there must be a grain boundary; look
at our old picture again if you can't quite "see"
it. So what does a grain boundary proper look like? |
|
 |
That's a rather difficult question. First we need to rephrase it a little:
What is the atomic structure of a grain boundary? How do the atoms stick together if
you cross from one grain to a another one? The answer leads into one of the more complex area of materials science with
rather heavy math and still unclear parts; this link to an advanced module
gives a little taste treat.
You don't quite see why this should be a difficult and complex part of Materials Science?
Then you should have no problem with a very simple exercise
You
don't want to know all that much about grain boundaries, you just want to see
some? OK— in this link are some pictures. |
 |
All you need to know about grain boundaries right now is that they cause massive
disturbances in a crystal. In other words, they introduce planes or "sheets" with rather high
energy in the crystal. There cannot be proper nirvana if grain boundaries are present; the disorder they produce
never balances the huge additional energy they carry with them. It is just as in the case
of extrinsic impurity atoms: You got 'em, you pretty much keep 'em! So let's note down in big letters: |
| |
Grain boundaries like all defects except
for intrinsic point defects prevent
nirvana conditions.
They just have too much energy.
|
|
 |
We call the energy coming with two-dimensional defects "grain boundary energy" or phase boundary
energy, or surface energy. We always give it as a specific
energy per cm2. The energy of two dimensional defects thus is their specific energy times
the area they occupy. For the nirvana state this is bad. The crystal doesn't want either of them.
Too bad that it is
very difficult for a poly crystal to get rid of its grain boundaries. Too bad for the
crystal, but good for us as we shall see. |
|
 |
The crystal certainly tries to get rid of its grain boundaries by trying to make the grains
larger and larger, until only one grain remains. Grains, however, will only grow at very high temperatures, and rarely enough
to produce a single crystal. If you are not inclined to wait at high temperatures until hell freezes over, your poly crystal
will still be a poly crystal, albeit with larger grains.
And all is well! For making good swords we need grain boundaries
for various reasons. One is shown right below: |
| |
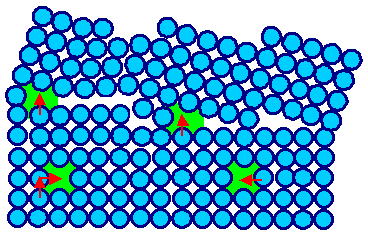 |
Let the atoms jump as indicated and the two green "vacancies" hiding in the grain boundary move
into the crystal. | Grain boundary emitting vacancies
|
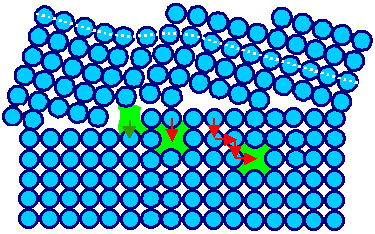 |
Let the atoms jump as indicated by the red arrows and the two green vacancies in the crystal
move into the grain boundary. The green arrow indicates an atom after the jump |
Grain boundary absorbing vacancies
|
|
 |
First I want you to appreciate that it is impossible to draw a grain boundary
"edge-on" in two dimensions without producing some "thinned-out" regions, with fewer atoms in a given
volume compared to the perfect crystal.
Now we can "see" why grain boundaries are high energy defects. Atoms
sitting in a grain boundary (or close by) just can't get as many partners as they want and where they want them. It takes
a lot of work or energy to produce such an arrangement and that's why grain boundaries are high-energy defects.
What
about their entropy or the amount of disorder they introduce into a crystal? Well—forget
it. Obviously they do produce disorder but the balance of energy vs. disorder is always very lopsided. There is no way to compensate for the heavy increase
in energy by gains in the TS product (remember?). For the
crystal, grain boundaries are therefore just as unwelcome as extrinsic point defects.
|
|
 |
From contemplating the figures, you may now have experienced a certain epiphany or enlightenment!
Grain boundaries (and other "big" defects) are the places where those "missing" atoms go when you make
vacancies!
Yes. That's correct. Now you know! It only remains to figure out where those missing single socks go but I will leave that task to you. |
 |
Next, I ask you to look at the figures above and imagine many vacancies going in (or coming out) of a grain boundary (there is a limit to my drawing skills).
What happens to the grain boundary in this case? You then realize that the only thing
that happens is that the grain boundary moves. |
|
 |
And now you also know why you need to heat your steel (or any crystal) to allow for grain
growth or to "anneal" or "normalize"
it. You simply need a lot of vacancies moving about to "heal" the crystal of all its other defects. Another "why"
question has found an answer. |
 |
One more big thought. A question
I asked before is how the crystal knows how many vacancies it needs to have for
nirvana? The answer was: it doesn't, and so on. |
|
 |
Now we see a bit better what happens. Grain boundaries emit and
absorb vacancies all the time in a completely random fashion. If on average they emit more vacancies per second than they
absorb, the vacancy concentration in the crystal goes up; the other way around it goes down. What happens depends on the
temperatures and maybe a few other things. But it just happens, no "thoughts" must be given to this business.
It's more or less like your bank account (or the state "bank account" called economy): it emits money at some
average rate to you (or to all of us). This is simply the money you withdraw or that is taken out of your account by others
like the IRS. Your account also absorbs money (whatever goes into your account). If the money concentration in your environment
goes up or down depends on the difference between the two rates. The rates change if the economic climate or temperature
changes; in crystal the rates change with temperature proper. And you don't really control all of this closely or think
much about it. You just react to general conditions. |
 |
Now let's generalize a bit: Grain boundaries are something like
garbage cans for all unwanted point defects. Self-interstitials and vacancies disappear
at grain boundary without any direct traces; all that happens is that the boundary has moved a bit. |
|
 |
Extrinsic point defects or impurity atoms cannot disappear
without a trace at a grain boundary; it's exactly like your garbage. It might be in the can—but it still exists. In
normal life, you probably like your garbage better in the garbage can than all over the place; your wife most certainly
does.
The crystal also likes to imprison his surplus impurity atoms
in grain boundaries just the same. The disturbance they cause in the perfect crystal is simply larger than the disturbance
they cause in the already disturbed grain boundary. However, grain boundaries are not very efficient garbage cans for impurity
atoms. They are useful if there are only a few impurity atoms around but if there are many, the crystal needs to find better
"prisons" for impurity atoms than grain boundaries.
If grain boundaries do manage to capture impurity atoms
and hold on to them, new problems might come up. For example, if your grain boundaries have sucked up all the sulfur atoms
that were roaming around in the grains, you now have clean grains and grain boundaries that consist of thin sheets of sulfur.
Heat up such a steel, and when the sulfur melts at just about 113 oC (235 oF), your grains will fall
apart. It's a bit more complex than that but that is essentially the mechanism that causes "red
shortnes". We will get to this later in more detail.
|
 |
Anyway, looking at grain boundaries, we learn a big truth. The crystal wants to
get rid of those defects but can't quite make it, it thus can never ever reach the true nirvana state. So what is it going
to do about that situation? Exactly what you and I would do: |
| |
|
|
 |
Lower the energy as much as you can. If you can't get rid of certain defects, mix and combine
them in the best possible way. The pain of having too much energy then is relieved to some extent, in particular at low
temperatures where the substantial disorder or entropy produced by all these defects hardly counts any more.
|
 |
It only remains to ask: Why do we almost unavoidably get poly crystals with grain
boundaries if that is not what the crystal likes? That's easy to see.
Whenever a solid crystal forms because a liquid
freezes, this doesn't happen in one fell swoop but gradually, starting from tiny nuclei (Latin
for seed or germ). That's so because the first step in going from being liquid to being solid, or if we generalize a bit,
from one phase to another one, is always a very difficult thing to do. |
|
 |
Phase changes (that's what freezing is, remember?)
almost always occur first at places where there is a little help to be had. For solidification or crystallization
the wall of the container helps a lot. It's colder there than in the inside of the liquid (the inside is always hotter than
the outside as you know if you ever ate a hot potato) and there might be convenient spots where an atom can get some rest
(they are running around in the liquid state, remember?)
Nucleation, the forming of the first tiny crystals of
the solid phase, then starts independently all over the place at "good" places
or nucleation sites at the container walls.
Then the initially formed nuclei grow—the crystallized parts become
larger—and eventually bump into each other. Since the crystal orientation usually is random, the crystal lattices do
not fit wherever they finally meet and a grain boundary must form, see below.
|
| |
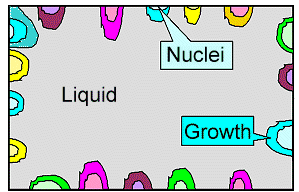 |
Solidification in progress
Nuclei
formed randomly and independently at some tiny irregularities on the container wall and grow into the remaining liquid.
The color symbolizes different orientations of the crystals / grains. |
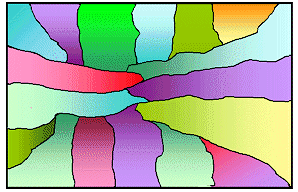 |
Solidification is finished
The independently formed crystals with random orientation grew together, forming the grains of a poly crystal. |
| Steps in uncontrolled crystallization of a meltVery schematic and far
too simple. I'll get back to this later. |
|
 |
While the schematic figures
above give an idea of what happens, this picture shows reality: |
| |
 | Grain structure in copper
Huge size | |
Source: Wikipedia / The Net at large |
|
| |
 |
Crystallization obviously proceeded from the outside to the inside. Whenever growing grains
met, a grain boundary formed. Also something needed to happen because there was not enough place for all grains to continue
growing. What happened is clearly visible: some grains stopped growing. |
 |
The figures and the picture are two-dimensional. Grain boundaries, however, enclose
a complete three-dimensional grain. They thus must curve around in three dimensions—but that is difficult to draw,
and not so easy to show in pictures either. It is not so difficult, however, to imagine the whole thing. Just think of the
foam on your beer, for example, imagining that
the thin membranes of the bubbles are rain boundaries, and you have an idea of what a three-dimensional grain boundary structure
could look like. |
|
 |
Here I always draw grain boundaries "edge on".
On rare occasions you also can see a real grain boundary exactly "edge on", indeed, in some thin slice of a material.
Then you might be able to take a HRTEM picture.
The picture below shows the atomic structure of an actual grain boundary
exactly edge-on in gold (Au), compare that to the figure above. |
| |
 |
Grain boundary in Gold (Au) standing "edge-on" and imaged
at atomic resolution.
|
| The insets show schematically the orientation of the cubic fcc elementary cell (not at the same scale as the picture).
|
|
 |
The picture once more illustrates nicely that grain boundaries are
good places to imprison impurity atoms in. There is just more room than between the atoms of a regular crystal. |
|
 |
Grain boundaries are also great places to nucleate
something. They are a kind of internal "wall" with plenty of distortions. So making a bit more distortion
by startingsomething new there—for example a precipitate—is
not as difficult as inside the perfect crystal. |
|
 |
Nucleationis a key word not only for sword
making but for making most everything from technical materials. It even governs your very existence. Did you ever wonder
why you (or a piece of wood, a bucket full of oil, ... ) do not spontaneously self-combust?
That would release a lot of energy and thus bring the collection of atoms that constitute you, a piece of wood, and so on,
much closer to nirvana.
I will come back to this a lot; it's a cornerstone in all of Materials Science! Meanwhile you
can ponder the question what kind of structure the collection of atoms that constitute you might assume for nirvana. You
might even want to jump ahead to the science module about nucleation. |
 |
Finally, let's take a quick look at why grain boundaries matter directly in sword making. A grain boundary, as the word implies, separates different grains of the same material. The grains are three-dimensional objects with a certain size. |
|
 |
If
the grains are enormous, (several millimeter (mm)) you can see them by just looking
(as in the pictures above). If they are just large
like several ten micrometers (µm), you can't see them with your unaided eye but easily with a standard optical microscope
or some fancier microscope as in the pictures below. If they are small and "nano" (far below 1 µm) you need an electron microscope.
|
|
|
 |
 |
| Grain structures in steel made visible by optical microscopy |
Source:
Top: Anne Feuerbach
Bottom: BAM;
Germany; with permission |
|
| |
 |
The top pictures shows the grain structure in a 5th - 6th century AD double-edged
sword from Kislovodsk. Black corrosion proceeds from the upper left, with cracks running down grain boundaries. This blade
was made from rather soft wrought iron. The black dots are slag inclusion
The bottom picture shows the grain grain structure
in hot-rolled modern steel. Fancy optical or electron microscopy provides for color. Grains with the same orientation have
the same color. Note the elongation of the grains by the rolling process. |
 |
Let's note right now that grain size
matters a lot for sword blades! If you somehow succeed to reduce the average grain size by a factor of 100 (from
50 µm to 0,5 µm, say), your blade will be about ten times harder (yes, there
is a square root law in this). The obvious "why" question going with this statement
I will tackle later. |
|
|
The message is clear:
We want small grains in sword blades!
|
|
 |
When you first crystallize a material from the liquid state, you almost always
end up with a poly crystal that has some average grain size. This may or may or may not be the grain size that you would
like it to have. In either case you must now do battle with the crystal: |
| |
You want the grains to be small.
The crystal,
for the usual nirvana reason, likes to have
its grains as
large as possible.
|
|
|
 |
A crystal is a tough opponent. It is not easy to make grains very small (say below micrometer)
and quite often you find grain sizes in the 10 micrometer range. For millennia we had to live with this (without knowing,
of course). There was just no way to make grains much smaller by trial-and-error.
The situation has changed. The present
"nano" madness in science and engineering is also
a bit about finding more an more ways to outsmart crystals and to make tiny "nano" grains. Not by trial-and-error
but by understanding and calculation before acting. |
 |
If we want to change the grain size in a given material, the grain boundary need
to move. That is a simple but far-reaching truth. If some grains gets larger, its grain boundary must move outwards, and
other smaller grains must get "eaten up" in the process. If you want to make the grains smaller, same thing in
reverse. |
|
 |
The obvious question coming up now is: how can a grain boundary move? Well, we have already
dealt with some aspects of the answer right above.
A bit more
generally the answer, like ever so often, is: |
| |
Grain boundaries generally move by
atoms jumping around or,
as we now call it, by diffusion of atoms.
|
|
 |
Here is the magic
word once more. Friendly vacancies need to come by, helping atoms on one side to move to a new position that now belongs
to the other side. |
|
 |
Vaguely recalling the relevant numbers, we realize
that appreciable movement of grain boundaries and thus grain growth will only happen
at rather high temperatures.
For the usually "achieve nirvana" reasons, the grains will invariably get larger
(and thus fewer) at high temperatures. There is no way you can make them smaller by just heating and waiting.
For making
grains smaller we need some other tricks, involving "dirt". I will give you more about that later. |
| |
|
© H. Föll (Iron, Steel and Swords script)









