| History of Carbon |
|
Introductory Remarks |
 |
Carbon compounds are rather prominent on this planet. They are called organic compounds and are found in abundance in what's called "biology". For example, the
following three simple and well-liked molecules contain plenty of carbon (C):
- C2H5OH. Chemical short-hand for the (ethylene) alcohol found
in beer, red wine and other important liquids.
- C6H12O6. Known as starch and found, for example,
in pasta and other necessities of life.
- C6H10O5. Sweetening life because it's (glucose) sugar.
|
|
 |
There are a hell of a lot (million, billion, trillion, whatever you like) more organic molecules
around, including DNA and other rather complex stuff. Pretty much all of them could be used to make carbon.
We also
have carbon compounds in gases like the carbon dioxide (CO2) that provides the fizz in soda and changes the climate,
or carbon monoxide (CO) that kills you and allows (carbon) smelting of metal ores. |
|
 |
Elemental carbon, however, is not so easy to find. Ancient
humans pretty much only encountered it in the cinder, soot, and charcoal leftovers found after some tree was hit by lightning
and burned down, or after they started to make fire themselves on a regular base (some 300,000 to 400,000 years ago; possibly
even more than 1 Mio years). Whatever black stuff remains after a careless burning of
organic material is very dirty carbon. The greyish stuff is ash, consisting of inorganic "salts". |
|
 |
The name carbon comes from the the French "charbone",
which in turn came from Latin "carbo", meaning "charcoal". In German the name is "Kohlenstoff"
which literally means "coal-stuff". |
 |
Looking around today, we can find more or
less elemental or natural carbon in the following modifications:
- Residues of (incomplete) burning as outlined above. The best you could do for producing pure carbon was to go for charcoal or soot deposited on some surface.
- Coal; coming in various grades from lignite with about 60 % - 75 % carbon content to anthracite with > 91.5 % carbon
content.
- Graphite, the hexagonal form of crystallized carbon.
- Diamond, the cubic metastable form of crystallized carbon.
|
|
 |
Synthesized or man-made carbon adds a few more versions;
use this link for details. |
 |
The History of Carbon thus has several main
and subsidiary branches:
- The history of all its modifications before it was known that they all are just
different expressions of the same element.
- The history of figuring out what the element carbon can do after its nature became clear.
- The history of the relation of carbon and iron.
- The history of organic chemistry.
- .....
|
|
 |
Let's start with the first branch. |
| | |
|
| |
History of Carbon Modifications |
|
Charcoal |
 |
Charcoal results when (dry) wood is heated to at least 275 oC (527
oF) in an oxygen-free environment. The wood then can't burn or oxidize with the basic reaction C + O2
® CO2 but pyrolyzes to carbon (in the form of charcoal)
and various gases.
Pyrolysis is the fancy word for "thermochemical decomposition of stuff" or
simply the falling apart of chemical compounds when you heat the stuff but don't supply oxygen for a proper burning. "Pyr"
is Greek for "fire" and the root of the English word "pyre";
"lysis" is Greek for "separating" as in "electrolysis". |
|
 |
Chemically speaking, wood is mostly a mixture of cellulose (40% –
50%), hemicellulose (15% – 25%) and lignin (15% – 30%). Chemical formulae for that kind of stuff are, for example,
(C6H10O5)n for polysaccharide cellulose, or C10H12O3
for a lignin variant.
In other words: besides carbon (C) wood contains quite a bit of oxygen (O) and hydrogen (H).
It is thus not surprising that the gases formed during pyrolysis are mostly carbon monoxide (CO), hydrogen (H2),
methane (CH4) and carbon dioxide (CO2). Note that the first three gases burn readily - if there is
oxygen available. |
 |
Pyrolysis of pretty much all organic stuff (including you) thus can be done for
several purposes:
- Making charcoal needed for old-fashioned smelting or modern barbecuing from wood.
- Making coke for modern smelting from coal.
- Making combustible gases needed, e.g., for driving a car if no gasoline is around, from coal, wood, plants, waste, ....
- Cleaning up organic waste including toxic and smelly stuff by turning it into coal and gases.
- Turning old tires into coal / coke, diesel oil and combustible gases (then used for firing the plant).
|
| |
 |
The first two processes we will encounter more often in this Hyperscript, they relate directly
to iron and sword making. The last two items are hot topics in our modern waste-producing American way-of-life world.
Number 3 is now out of style but, believe it or not, was quite prominent in WW II Germany. Gasoline was scarce and cars
and trucks had a "wood carburetor", or more precisely, an unwieldy gas generator for pyrolyzing wood, a feed tube
for the gases, and a carburetor etc. adopted to feeding the gas to the engine. The picture below shows it all.
It wasn't
much fun to run a wood-fueled car. But that was still far superior to not running a car at all. I guess that the urge for
getting away on wheels is second only to drinking
beer, which comes right after urge No.1. |
| |
| |
| |
 |
| German car around 1940, running on wood as fuel |
| Source: Bundesarchiv_Bild_183-V00670A,_Berlin,_Auto_mit_Holzgasantrieb.jpg |
|
| | |
|
 |
Having decent charcoal around was important for millennia. Charcoal was decisive
for: - Smelting of metals from the ores.
- Melting of the smelted metals for casting.
- Forging of non-meltable iron and steel.
- Making glass and advanced pottery.This coul also be done
with dry wood, hoever.
If that list confuses you, turn to this module.
Charcoal making is relatively easy and was done in pretty much the same way for thousands of years. Wood is all that is
needed. Northern Europe thus had no problem in this respect for quite a while, but ancient Egypt, for example, had not much
wood at its disposal and was thus at a certain disadvantage concerning the charcoal-enabled technologies given above. On
the other hand, they didn't need wood as urgently as my forebears to keep warm during the 9 months when it is cold. |
|
 |
Charcoal in the time-honored fashion is produced by pyrolyzing a pile of wood that is contained
in an (almost) airtight enclosure made from mud bricks or stuff like that. Shoveling earth on the pile might already do
the trick. Of course, the ignorant charburners and the important well-educated or at
least rich people didn't call it "pyrolyzing" for the first few thousand years but "burning" because
some (small) part of the wood is really burnt. For that you needed to have a few vents in the mud cover, and the initial
burning proper produced the heat to get the pyrolysis going in the larger part of the pile.
The pictures below show
what a charcoal pile looked like since about 1.400 AD. Before that, the wood was simply thrown into a pit in the earth,
set on fire and then covered to prevent all-out burning. |
|
| |
| |
 |
| Charcoal making 1910. Next, the pile will be covered with earth. |
| Source: Photographed in the Museum "Hochofenanlage Neue Hütte",
Schmalkalden, Germany in 2012 |
|
| | |
|
|
|
 |
| The internal structure of a charcoal pile |
| Source: Wikipedia |
|
| |
|
|
 |
As soon as the process has been started around 275 oC (527 oF), it
is self-supporting since pyrolysis generates heat of its own and thus raises the temperature. The process continues until
all the wood has been converted. As a rule of thumb, 5 tons of wood make 1 ton of charcoal.
Of course, since wood contains
more stuff besides the major ingredients listed above, all the non-burnable stuff (called "ashes"
in burning proper) is now contained as dirt in the charcoal. Have you ever wondered why flour (mostly starch) in Germany
has always a number on it like "type 405" or "type 1050"? This number just gives the weight of the remaining
ash in milligram (mg) if you burn 100 grams (g) of the flour. The more ash, the more minerals are in there. Ash generally
accounts for 1 % or less of the weight of (dry) wood. For some odd reason my wife thinks that ash is good for you when she
uses flour for baking.
Wood ashes consist mainly of oxides of calcium (Ca), potassium (K) and magnesium (Mg). These
oxides account for roughly 80 % of the ash; next come oxides of aluminum (Al), phosphorous (P), sulfur (S) and a few more.
Charcoal retains the original cell structure of the wood and thus is very porous or, in other words, has a large surface-to-volume
ratio. That's why it burns far better and hotter than wood or regular coal. There is simply more surface area where the
process can take place; see below: |
| |
| |
| | |
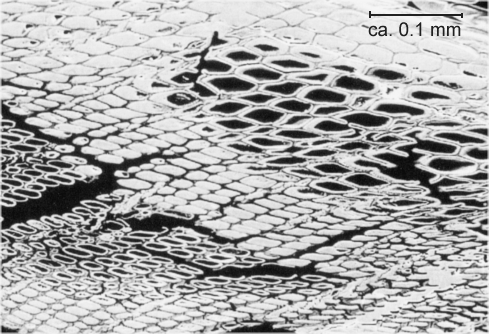 |
| Charcoal (probably from pine) conserved in slag during early iron smelting |
| Source: Vagn
Buchwald's wonderful book |
|
| | |
|
|
 |
Of course, ever-present "sympathetic magic"
was invoked to relate hard iron to charcoal made from hard
wood and soft iron to charcoal from soft wood, etc.
It is true to some extent that charcoal made from hard wood might make your iron hader,
i.e having a higher carbon content - but no because of magic but for good
scientific reasons. It was a good idea to be very concerned about the charoal that you used in your smelter, and smelter
operators had a strong tendency to never chane a "winning team". |
 |
When metal smelting became a major industry, let's say ever since 500 BC, de-forestation
became a problem. Wood or charcoal had to be transported over ever increasing distances, and there is some speculation that
the depletion of wood resources lead to the decline of whole empires, e.g. in Africa, (for example the famous Kingdom of
Kush), where iron was smelted as early as 900 BC. |
|
 |
Charcoal burners or Charburners were a-plenty in ancient metal-working society but their profession
usually had a somewhat seedy reputation. Of course, throughout most of history, the useless but powerful nobility looked
down on manual labor anyway, and the appearance of always black and dirty charburners did not particularly recommend them
as good companions. |
| |
|
|
Diamond |
 |
In a few areas around the globe raw diamonds could be found just so; in particular,
it appears, in deposits of some Indian rivers. Diamonds have been known in India for at least 3,000 years, maybe even 6.000
years. What exactly people did with them is unclear. They were used as (uncut) gemstones,
religious icons (whatever that might be) and as tools for scratching and working hard things. |
|
 |
The name diamond (German: Diamant), you
guessed it, derives from the ancient Greek "adámas", meaning "proper", "unalterable",
"unbreakable", and so on. I would guess that it is the same root as "atomos", meaning indivisible or
uncuttable, which gave us the "atom" - but it is all Greek to me. |
 |
Diamonds have a simple cubic
face-centered (fcc) structure like silicon (Si) or germanium (Ge) as shown below. |
| | |
| |
 | | Diamond crystal structure. |
|
| |
|
|
 |
Blue and green spheres mark the position of carbon atoms, the blue spheres mark also the position
of lattice points. Red lines show the bonds between atoms. The black lines are meaningless,
they just guide the eye to identify the cubic structure. |
 |
A Chinese work from the 3rd century BC refers to diamonds as amulets of foreigners
(warding off evil influences) while the Chinese themselves used (imported) diamonds as tools for working jade. While diamonds for millennia came mostly from India, Brazil (major
finds in 1725) and South Africa (major finds 1867 in Kimberley) eventually took over. |
|
 |
Small numbers of diamonds began appearing in European jewelry in the 13th century; they were
used as "accent points" among pearls set
in gold. Louis IX of France (13th century) reserved diamonds for the king by law, demonstrating that this piece of carbon
was now seen as extremely valuable.
The big days of diamond,
lasting until today, started when the facetted cut that we have today as a matter of course was invented by Jules Cardinal Mazarin in the 17th century. |
 |
Diamonds are a metastable phase of carbon; the stable phase is hexagonal graphite. The formation of diamonds therefore requires
very specific conditions: rather high pressure (around 5 GPa) and high but not excessively high temperatures (around 1.000
oC). That makes it rather difficult to grow big single diamond crystals in the laboratory. On the other hand,
making small diamonds or coating all kinds of materials with a thin layer of diamond is now commercial routine. |
|
 |
The black stuff left in your oven, after doing your turkey or whatever, contains what one
could call "amorphous diamond" with a lot of strong diamond-like bonds. It is thus not surprising that this annoying
matter is very hard and durable and almost impossible to scratch off or to dissolve in chemicals that don't tend to kill
you on the side. |
| | |
|
|
Coal and Coke |
 |
Coal is the black stuff we dig out of the ground in
ever increasing quantities. The table below lists the 8 biggest producers; there are of course many more. |
| | |
| |
| Country | 2009 | 2011 |
| | Mio tons |
| China | 2.971 | 3.162 |
| USA | 985 | 997 |
| India | 571 | 571 |
| Australia | 399 | 420 |
| Indonesia | 301 | 336 |
| Russia | 297 | 324 |
| South Africa | 247 | 255 |
| Poland | 134 | 135 |
| Total | 5.905 |
6.200 |
|
| |
|
|
 |
Most of that coal ends up as carbon dioxide (CO2). Frightening, isn't it? A lot
of this is used to produce the - roughly - 1.200 Mio tons of steel per year. |
 |
In some areas of the world coal seams came out of the ground, and coal then was
just dug out and used on a small scale, e.g. by the Romans. Wikipedia writes: "In Roman Britain,
the Romans were exploiting all major coalfields (save those of North and South Staffordshire) by the late 2nd century AD.
While much of its use remained local, a lively trade developed along the North Sea coast supplying coal to Yorkshire and
London. This also extended to the continental Rhineland, where bituminous coal was already used for the smelting of iron
ore."
I can't quite believe that. Smelting iron ore with bituminous coal, or any coal for that matter, would
generate very inferior steel because of the sulfur problem. My guess is that smelting and melting have been confused once more.
Major use of coal started
when Great Britain had finally cut down most of its forests in the 16th century. The use of coal as domestic fuel rapidly
expanded, as did the diseases caused by the smoke. The industrial revolution in the 19th century finally led to an explosive
growth of coal mining that has essentially continued to this very day. |
| | |
|
|
|
 | | Bitumineous coal seam exposed at the beach |
| Source: wikipedia |
|
| |
|
|
 |
Coal, mind you, is not a well defined substance and it
is not carbon as already pointed out above. In the
best case (anthracite) it contains about 10 % foreign matter, in the worst case (lignite),
dirt accounts for around 40 %. The non-coal stuff contains mostly oxygen and hydrogen. But all
grades of coal, unfortunately, contain about 1 % of sulfur, and that is rather bad for steel production, not to mention
the environment (look up "acid rain").
The figure below gives a schematic view of the composition of coal. |
| |
|
|
|
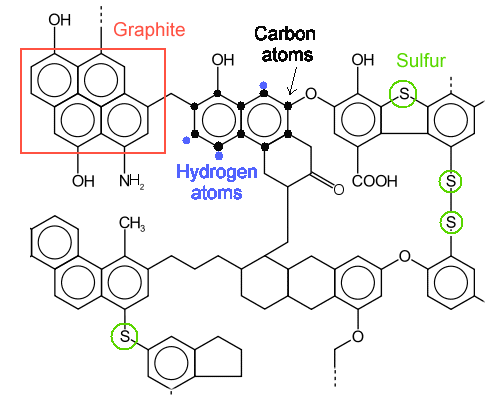
|
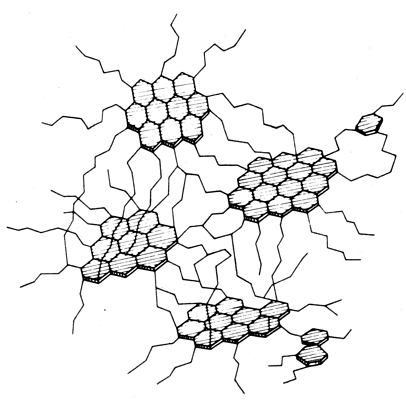 |
| Composition of coal - very schematic and three-dimensional |
|
Source: Adopted from wikipedia; Obscure old Russian text book from A. I. Kitaigorodsky |
|
| | |
|
| |
 |
If there would only be ordered arrays of those hexagons, it would be pure graphite. Wherever
two lines meet in the upper picture sits a carbon atom and a hydrogen atom (mostly not
drawn). Where three lines meet is only a carbon atom. |
 |
Coke (short for “coal-cake”), is to coal what
wine is to grapes: a highly refined and superior product in comparison to the raw material it was made from. One could also
say that it relates to coal the same way charcoal relates to wood. Coke here, to be perfectly clear, is not
something you snort up your nose. |
|
 |
In a nutshell: coke results when you pyrolyze coal.
Rather pure carbon is left over, and as a by-product coal-gas, and coal-tar are produced in
large quantities. These by-products created their own industries. Coal gas (also called town gas
and illumination gas) was the primary source of gaseous fuel for lighting, cooking and heating in many cities in the 18th
and 19th century. Coal tar was used for making all kinds of early organic compounds like creosote.
In the western world,
Sir Hugh Plat (1552–1608), an English writer and inventor,
first suggested the process in 1603. Nobody heeded his idea, of course, until the need to have something that could replace
charcoal became a matter of life and death. Charcoal was becoming expensive in merry old England in the 16th century because
the forests were mostly cut down by then, and that meant that metal smelting became expensive. Well, yes, but who cares?
Far worse was that beer
brewing and thus beer became expensive, because you need a good fuel to roast
the malt needed for making beer! You just can't run a decent civilization that smelts metals and is bent on conquering the world (or at least the French) without large quantities
of good and affordable beer, as I have ascertained before.
Using coal for roasting the malt impairs a foul taste to the beer because of the sulfur in the coal. Making coke became
imperative, and beer brewing with coke started 1642 in Derbyshire for good.
Of course, after the bulk supply of beer
was ensured, coke could then also be used to smelt metals, make swords and later guns, and all the other hardware needed
for conquering the world. It took a while, however. It was not before 1710 that one Abraham Darby (1678 – 1717) developed a method of producing cast iron in a blast furnace fuelled
by coke rather than charcoal. It took even longer before that caught on - only around
1800 pretty much all blast furnaces were run on coke. Some charcoal fanatics, however, where not convinced even then and
kept their smelters on charcoal well into the 20th century |
|
 |
The Chinese did it the wrong
way around. They started to make coke already in the 9th century AD but didn't use it for making first beer and then iron.
They somehow got confused and started smelting iron with coke right away. In the 11th century they had a major iron industry
running that was based on coke and not just charcoal. That kept them so busy that they never got around to making decent
beer.
Poor suckers, it was downhill ever since. They could have conquered the world quite easily in the 15th century,
long before the Spaniards and Portuguese made their bid, because they had superior hardware and ships, and many other advanced
things like live-in concubines. Fortunately (for us), they didn't have the balls beer needed for some conquering.
Now look at the British and the Germans. They focussed on beer for quite a while - and the British eventually did conquer
most of the world and they still feel good about that! We Germans weren't quite that successful but at least we tried. The
Americans today have some success, but their conquering-the-world efforts get rather mixed reviews. I blame it on the quality of their beer.
|
| | |
|
|
Graphite |
 |
Graphite is the stable phase of carbon with a hexagonal lattice. It is not a simple hexagonal close-packed structure but a bit more complicated as shown below. |
| |
|
|
|
 | | Graphite Crystal structure |
|
| | |
|
|
 |
The bonds in the hexagonal plane are very strong just
like in diamond, while the bonds between the planes are very weak. That's why it is
very easy to deform graphite in directions parallel to the hexagonal planes and very difficult in directions perpendicular
to it. That allows applications that are breathtakingly different:
- Use poly-crystalline graphite. Whenever you press or pull on it, some areas shift easily and stick to the contact material.
This is great for making pencils or lubricants.
- Use monocrystalline graphite in long fibres oriented in the hexagonal plane. When you pull at the fibres, you are trying
to break diamond bonds and that is tough to do. Now protect your fibres from forces at right angles by encasing them in
some epoxy. Bingo! You have made carbon-fiber-reinforced polymer or plastic (CFRP or CRP) with a strength-to-weight ratio
that exceeds the best steels by far.
|
 |
The name "graphite" was coined
by one Abraham Gottlob Werner in 1789 from (what else?) ancient
Greek: "grapho"=to draw, to write, because it was used in "lead" pencils. This already gives a hint that there was some confusion as to the nature of the stuff in pencils.
People thought for a long time that natural graphite was some lead mineral. |
|
 |
Graphite is quite essential to modern technology. It is not only good for pencils, as a lubricant,
or for CFRP; it is an electrical conductor that can take enormous temperatures (it doesn't melt but becomes directly gaseous
around 3.750 oC (6780 oF) and is chemically very stable. That's
why it is used in the high-temperature "electro" smelting of difficult elements like silicon. |
 |
Graphite may be considered as the highest grade of coal (above anthracite) and therefore is found in small quantities wherever coal is found. It was used pretty
much throughout history as "black paint", it seems, e.g. on pottery. Personally, I'm not sure if the graphite
paint found on old pottery resulted from using "true" graphite or just soot. After firing the pots, the result
could be about the same. |
|
 |
Graphite proper came into its own after a huge deposit of extremely pure
and soft stuff was discovered in 1565 (or possibly somewhat earlier) in the Borrowdale parish, Cumbria, England. The local
yokels used it for marking sheep and probably didn't worry much about what that soft black stuff actually could be. A somewhat
more advanced use coming up a bit later was to line the molds for cannon balls with this graphite, resulting in rounder,
smoother balls that could be fired farther.
The military guys did wonder about what that useful black stuff could be,
and promptly confused it with lead or some of the more common lead ores like galena.
That is why graphite was known for a long time as "lead" or "plumbago"
(based on the Latin "plumbum" for lead). This error survived to some extent up to the present day. In German,
a pencil is still called "Bleistift", literally "lead pen". Archeologists also confused
lead and lead ore. Granted that graphite, lead and galena look similar, one could
at least distinguish graphite from the two others easily because the difference in (specific) weight is rather obvious,
you might think. Yes, but to everybody before - roughly - 1750, the notion of chemical elements was unknow. Things that
were similar were thought to be about the same. The differences were assigned to the presence or absence of "vital
juices", "spirits" or "priciples" of this or that. Graphite in many aspects is far more similar
to lead or lead ore then to diamond, or soot. I'm quite sure that even today it would be far easier to persuade most people
that graphite is related to lead and not to diamond. |
| |
| |
|
Soot and Carbon Black |
 |
Soot
is that fine black stuff that remains in the air from burning something, and that the chimney sweep takes out of your chimney
on a regular base. You only can avoid it in very "clean" fires. It results from the "incomplete combustion
of a hydrocarbon", for example when a candle burns wax. Put a glass plate over a candle flame and you catch the soot
in the air. It is part of what we call "smoke" and accounts for a lot of sick people, especially in countries
where open fires are the standard for cooking. |
|
 |
Soot consists of rather small (below 100 nm) particles of carbon (plus some dirt).
This particles might agglomerate to some extent, forming chains and God knows what, and at least parts of them consist of
amorphous carbon. I'm sure, however, that you will find all other forms of carbon too, if you search long enough. |
| | |
|
| |
|
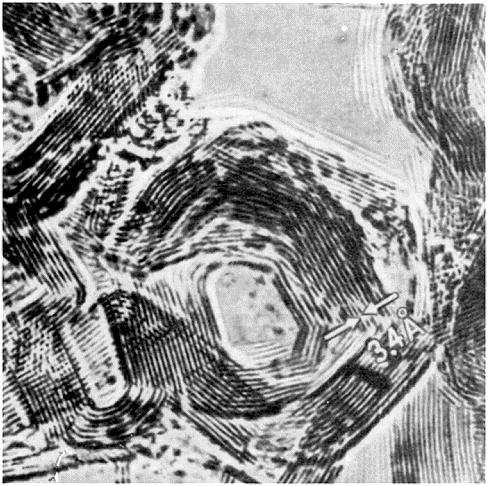 |
Atomic structure of graphitized carbon black; HRTEM picture
The parallel lines
are small graphite (nano)crystals; you look at the hexagonal planes "edge-on". |
| Source: Obscure old Russian text book from A. I. Kitaigorodsky; actual source not identified. |
|
| | |
|
 |
Soot, made unintentionally by you via burning
something, should not be confused with carbon black that is made intentionally
(by burning something). Carbon
black is rather pure carbon that serves as raw material for important carbon-based products. It is, for example, used
as pigment in your toner cartridge, and it is what makes care tires black. World production is around 10 Mio. tons a year.
Make it very hot and it graphitizes as shown above.
And don't confuse "carbon black" with Black
Carbon - look it up yourself! |
|
 |
Ancient man used soot for painting himself, for tattooing, painting caves, whatever.
It was not High-Tech and thus is not very interesting to us.
Modern man like me and my colleagues used very pure carbon
black for a while in experiments designed to make very pure silicon via "electro-smelting
of difficult elements". That also needed very clean silicon dioxide (SiO2). It didn't really work but
that is another story. Working with the stuff makes everything (including you) quite black, too. I have never done anything
quite that dirty again during my career, and that includes convincing my kids that the shortcuts I proposed on major hikes
would bring us home pronto, not to mention running a major university faculty as Dean. |
| | |
|
| |
History of Putting Things Together |
|
It's Carbon! |
 |
You must admit that anybody not familiar with the basics of chemistry and the
periodic table would declare you to be completely nuts if you
would propose that all the stuff described above is one and the same basic substance. Before about 1700, "anybody not
familiar" and so on would simply have been everybody minus a handful of fledgling scientists. |
|
 |
It was Robert Boyle
who suspected in 1661 that there were more than just the four classical elements that the ancients had postulated. He endorsed
the view of elements as the undecomposable constituents of material bodies and made a distinction between mixtures and compounds.
Nevertheless, he was also an alchemist (and a racist) and believed in the transmutation of metals - making gold from lead,
in other words. |
|
 |
Antoine Lavoisier,
the "father of modern chemistry" whom we have encountered
before, supplied the first list of elements in his "Traité élémentaire de chimie (Elementary
Treatise on Chemistry) in 1789. Oxygen, nitrogen, hydrogen, phosphorus, mercury, zinc, and sulfur were (correctly) identified
as elements - but also light and "caloric" (heat stuff), which
he incorrectly believed to be a material substance. In 1775 he also recognized soot as being the element carbon, and more
importantly, established that diamond was also carbon by doing the amazing experiment described below.
While Lavoisier's
work was daring and pioneering, we need to be aware that Lavoisier could not really prove beyond any doubt if some substance
was an element or a compound. His inclusion of carbon, while correct, could also be seen as a lucky guess. It was not clear,
for example, which manifestations of the element were really carbon. Some experiment by one Guyton de Morveau much later
in 1799 lead others to believe that only diamond would be pure carbon, while graphite would be an oxide of the "1st
degree", charcoal of the 2nd degree, and carbonic acid, finally, would be the "complete oxide". Pepys and
Allen corrected that in 1807, which helped Dalton in formulating his "law of multiple proportions" in 1808, paving
the road to atoms and the periodic table.
Of course, Lavoisier was neither alone in his enterprise nor did everybody
believe him right away (or later, considering that there are mistakes; see below). The eminent Priestley,
for example, also credited with "discovering" oxygen, never believed him at all.
It goes wothout saying that
the people actually making and working with iron and steel couldn't have cared less for quite a while. Major insights were
fine around then but usually not all that helpful for practitioners. |
| |
|
|
|
 | | Elements according to A. Lavoisier |
| Source: Wertheim's
book. Obviously from an early translation to English. |
|
| | |
|
 |
Some time in 1772, Lavoisiser and some of his buddies pooled their funds, purchased
a diamond, put it into a closed glass jar, and focussed the rays of the sun on it with a big lens, supplying the ultimate
"clean" heat. The diamond disappeared and since the weight of the glass
jar was unchanged, the unavoidable conclusion was that the diamond had turned into a gas that could only be carbon dioxide,
proving that a diamond was pure carbon.
The experiment was not as simple it as it appears nowadays, as this contemporary
illustration proves: |
| |
|
|
|
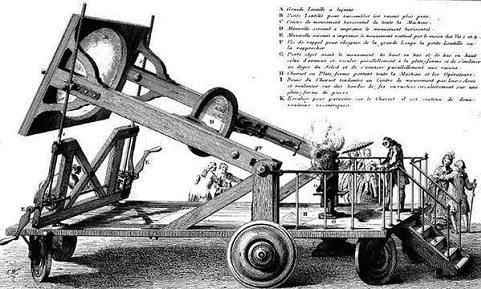 | | Burning diamonds in style |
| Source: http://historyofscience.free.fr/Comite-Lavoisier/f_galerie_sur_lavoisier.htm
|
|
| | |
|
|
 |
I don't know how Lavoisier's insight was received by the public but
will bet that almost nobody believed him (and that his wife was mad at him for destroying that diamond). The judge who had
him beheaded in 1794 said: "La République n'a pas besoin de savants ni de chimistes ; le cours de la justice
ne peut être suspendu" (The Republic needs neither scientists nor chemists; the course of justice cannot
be delayed).
Well, Lavoisier also supported the metric system,
and fought for the rights of a number of foreign-born scientists, including mathematician Joseph
Louis Lagrange, during the Reign of Terror, so he had it coming for himself. |
 |
In essence, the 18th century was when "chemistry" was born. This was
a difficult delivery because it required not only to do away with the "four element theory" (plus "aether"
or "quintessence", added by Aristotle, being wrong as ususal, as the fifth element) but also to get rid of the more modern "phlogiston"
theory. Boyle and Lavoisier, mentioned above, were decisive in this enterprise, but also a lot of other fledgling scientists. |
|
 |
Carl Wilhelm Scheele
noticed in 1799, four year after Lavosisier recognized soot to be carbon, that graphite was carbon, too. Scheele also proved (as far as was possible) the elemental character of:
Barium (Ba), Chlorine (Cl), Fluorine
(F), Manganese (Mn), Molybdenum (Mo), Phosphorous (P), Oxygen (O) Nitrogen (N), and Tungsten (W).
Of course now you
wonder. Just above I wrote that Lavoisier had oxygen, nitrogen and so on listed as elements - and that was earlier. Well,
as far as oxygen is concerned, I must also mention Joseph Priestley
who described a special gas - we call it oxygen - in 1774, even before Lavosisier. I guess the discovery of oxygen and nitrogen
was in the air, so to speak. As far as priorities go, Scheele probably made his discoveries in 1773, even before Priestley,
but published it later than Lavoisier. Whatever, Priestley and Lavosier were English and French and thus not trustworthy;
look what they did with Lavoisier. Scheele was German and now you could believe it. Scheele won ever-lasting fame by being
instrumental in overturning the phlogiston theory. |
|
 |
Smithson Tennant,
an English chemist, not only discovered the elements iridium (Ir) and osmium (Os) but proved in 1797 once more, but now
also beyond reasonable doubt for the imbecile, lawyers and politicians, that diamond is indeed a phase of carbon. He did
that together with his assistant William Hyde Wollaston, another
well-known name in scientific circles, by reacting one part (=mol)
of carbon with two parts of oxygen, obtaining nothing but carbon dioxide (CO2). |
 |
So at the beginning of the 19th century it was clear that soot, graphite, diamond,
coal, coke and charcoal were just different manifestations of the element carbon, containing more or less "dirt"
on the side. |
|
 |
In the following 150 years the understanding of carbon manifestations was refined
and developed in great detail but nothing really new was added to the list above until about 1950 when (small) diamonds
could be synthesized to some extent. Somewhat later, around 1970, the new and exciting carbon manifestations known under
names like "Bucky balls", fullerenes, carbon nanotubes and graphene, started to appear, causing major scientific
orgasms. The party is still on. This is described in another module. |
|
|
| | |
|
|
It's Carbon
That Makes Steel! |
 |
Everybody involved in the early iron and steel industry knew that the there were
pronounced differences between (wrought) iron, steel and cast-iron and that one could change the properties of these materials
to a large extent. One could even make steel from both wrough iron and cast-iron by some proper processing. Some of the
practitioners of old must have given some thought to the question of what is responsible for the differences. Unfortunately,
none of them wrote it down, or if somebody did, it was lost.
Most people thinking about that probably followed Aristotle and considered steel to be a more
refined form of iron and thus were completely wrong. The question thus is: When did it become clear that steel is actually
"dirty" iron, and that the most important dirt in this context was carbon? The answer is: In the 17th / 18th century
- but details are a bit muddled. It can't be otherwise. You cannot possibly figure out that carbon is the decisive element
for steel making if you lack both: a valid concept of elements and an idea of what is carbon. |
|
 |
We do know, however, that René Antoine Ferchault Réaumur (1683-1757) figured out that steel is "dirty" iron. He may not
have been the first one to entertain this notion but he did clever experiments and wrote about it at length. It's the same
Réaumur, by the way, to whom we owe the Reaumur
scale for measuring temperatures. He is famous for a lot of other things but he seems to be the first (proto)scientist
who conducted a systematic study into the production of steel. That wasn't just for fun. In the early 18th century the French
iron and steel industry was seriously backward, and the French, intend on conquering the world even so they lacked decent beer, needed decent hardware for fighting and shooting.
Réaumur was able to obtain documentation
concerning the iron and steel industries of foreign countries and his direct boss, Philippe II, duke of Orleans, subsidized
Réaumur’s research by granting him a pension of 12,000 livres (presumably a lot) to further his studies concerning
iron and steel. | |
 | | Steel (!) engraving of Réaumur |
|
 |
In Réaumur's times the basic product of the ferrous industry was "pig
iron" (=cast-iron) from early blast furnaces, from which wrought iron was made. Steel was produced by "cementation",
i.e. by mixing iron and charcoal and heating the mix for weeks, until the iron was carburized to what was called "blister steel". France somehow missed the advent
of this new technology but Réaumur knew about it and thus didn't have to think too hard to get the idea that steel
was iron plus something. It's however, not quite as obvious as it looks to us. Not knowing
that wrought iron is rather pure iron, the cementation process could just as well have sucked something out of the wrought
iron and thus turned it into steel. |
|
 |
Réaumur went at it systematically and did what a true scientist would do:
He charged lots of identical small crucibles with wrought iron plus all kinds of stuff that could supply the "cement"
needed for making steel, and heated the mix under "rigidly" controlled conditions. Essentially he tried to change
just one variable from experiment to experiment, exactly the right thing to do even in modern science.
After a large
number of experiments Réaumur concluded that the best mixture for making steel out of iron was to use a specific
combination of chimney soot, charcoal, ashes, and common salt.
More important, his general conclusion was that steel
was in fact impure iron, somehow intermixed with “sulfurous and saline particles.”
More than that, he also concluded - correctly - that cast-iron was even more impure than steel.
But sulfur?
Looks like he didn't recognize that the essential stuff needed to make steel was actually carbon?
Well - carbon hadn't been "invented" yet, look above. In the early eighteenth century chemistry, “sulfurous”
was the general term for inflammable stuff or for the "oily principles" contained in combustible substances, such
as charcoal, soot, or whatever else could be lit up. It didn't just mean the element
sulfur (S). Thus Réaumur was rather close to answering both essential questions from above in 1722, when he published
his studies entitles "The art of converting wrought iron into steel" and "The art of making cast iron malleable"
(of course the original titles were in Latin). |
|
 |
Réaumur actually did far more. He investigated the structure of fractured
iron and steel pieces and tried to explain a lot of things from the differences he observed (iron - fibrous; steel - lamellar;
cast-iron - granular; not too bad). Lacking a microscope and proper preparation methods, not to mention the concept of atoms,
crystals, grains, and so on, he could not possibly arrive at the truth but got much closer than his contemporaries. For
example, he maintained that quenching changed the structure of the metal without putting some "vital juices" from
the water into the steel. He also put an end to the mythology about quenching
agents (water, oil, blood,...) by pointing out that it is only their different ability to cool (we would call it
different thermal conductivity) that was important.
Most remarkable, perhaps, he reasoned that the infusion of his “sulfurous
and saline particles” made not only hard steel out of soft iron, but also hard and brittle cast-iron depending on how
much of the stuff was infused. That is essentially correct. Cast-iron was very important for making cannons but also very
dangerous because you tended to kill about as much of your own soldiers by exploding cannons made from the brittle stuff
as enemy soldiers by getting the cannon ball over there. Malleable cast-iron, that actually could be cast, (in contrast
to steel), would have been of utmost importance - but didn't exist.
Réaumur figured that one could make malleable
cast-iron by taking some of the "sulfurous and saline particles" out of the ubiquitous brittle cast iron. He thus
could be seen as the grandfather of the first "designer" steel. He even found a method to do so but (cover the
cast-iron with some hard-to-get mysterious substance then know as "saffron of Mars") but (wrongly) considered
that not to be practical, due to the rarity of the stuff. Much later in the 19th century, the “Réaumur process”
was actually used on a large commercial scale because the mysterious reddish stuff turned out to be simple iron oxide or
iron ore. |
 |
The next guy who must be mentioned in this context, is Torben
Olof Bergman (1735-1784), a Swedish subject. Sweden was a major producer of iron and steel
and used to be a superpower that liked to invade Germany, Denmark, Finland, Russia, and so on, until it was reduced to roughly
what it is today in 1721.
Bergman, from Sweden's Uppsala University, published a book called "Dissertatio Chemica
de Analysi Ferri" in 1781 (together with his Ph.D. student Gadolin; the book was essentially his thesis, it appears)
in which he sought explanations for the different types of iron and steel in terms of the metal's chemical composition.
He reasoned, not quite correctly, that only elements commonly found in the ore could be responsible for the changes in the
metal: sulfur, plumbago, arsenic, zinc, and manganese. Note that "chemistry"
in the modern sense was around the corner but that Lavoisier's seminal book was to appear 8 years later.
| |

| | Torben Bergmann |
|
| |
 |
In contrast to Réaumur, who arrived at this conclusions by synthesis, by
making steel, Bergmann's approach was analytical: look what is in there. That's why Bergmann is sometimes called the father
of analytical chemistry. |
|
 |
Bergmann is sometime given credit for discovering that it is carbon
that makes steel out of iron. To understand that we need to remember that "plumbago", while meaning lead (Pb)
in principle, also was the name for graphite. In 1781 Bergman reported
he had found that cast iron contained up to 3.3% of 'plumbago', steel contained up to 0.8% and wrought iron less than 0.2%.
You can't do much better that that.
One of Bergmann's student was Carl Wilhelm Scheele
mentioned above and by now the proto-science of iron and steel was well on its way. |
 |
Finally, in 1786, Gaspard Monge
(1746 -1818), Charles Augustin Vandermonde (1727–1762)
Claude Louis Berthollet (1748-1822) published major work that
finally used the word carbon (actually "charbone") instead of "plumbago" or "sulfurous particles"
and thus put an end to the basic question of what makes steel. |
|
 |
Well - not quite. While it became clear to the initiated that you
could make steel by putting some carbon in otherwise pure iron, you could make steel in the sense of "hard iron plus
something" also by many other means, e.g. with phosphorous.
And hardness was only one important property. Just as important (and rather unclear) were properties like malleability,
"cold shortness" and "red shortness", and just as much or even more work was spend on the questions
coming up in this context. |
 |
Since even the late 17th /early 18th century scientists knew more
about that than we you do at this stage; I'll stop now. Rest assured that the matter will come up again
further down this Hyperscript. |
| |
| |
 With frame
With frame

 Glossary
Glossary
 Periodic Table of the Elements
Periodic Table of the Elements
 Books and Other Major Sources
Books and Other Major Sources
 Invention or Discovery?
Invention or Discovery?
 4.2.2 Being Iron
4.2.2 Being Iron
 Steel Guys
Steel Guys
 Beer
Beer
 10.1.3 Smelting, Melting, Casting and Alloying Copper - The First
10.1.3 Smelting, Melting, Casting and Alloying Copper - The First
 Temperature Measurements
Temperature Measurements
 Phlogiston
Phlogiston
 Discovery of Atoms
Discovery of Atoms
 Early Metal Technology - 2. Silver and Lead
Early Metal Technology - 2. Silver and Lead
 10.5. Iron and Steel in "Modern" Europe. 10.5.1 From Bloomeries via Stückofen and Catalan Forge to the Blast Furnace
10.5. Iron and Steel in "Modern" Europe. 10.5.1 From Bloomeries via Stückofen and Catalan Forge to the Blast Furnace
 The Ages
The Ages
 9.1.1 Things are Complicated
9.1.1 Things are Complicated
 Bravais Lattices and Crystals
Bravais Lattices and Crystals
 Wagner's "The Ring of the Nibelung"
Wagner's "The Ring of the Nibelung"
 11.2.1 Background to Celtic Swords
11.2.1 Background to Celtic Swords
 8.4.1 Martensite
8.4.1 Martensite
 Jominy Test and Hardness Depth
Jominy Test and Hardness Depth
 Early Copper Sites
Early Copper Sites
 10.5.3 Making Steel after 1870
10.5.3 Making Steel after 1870
 Smelting Science - 1. Furnaces
Smelting Science - 1. Furnaces
 Some Old Names Around Steel and Iron
Some Old Names Around Steel and Iron
 10.2.3 Smelting Wrought Iron, Steel and Cast Iron
10.2.3 Smelting Wrought Iron, Steel and Cast Iron
 Steel Revolution
Steel Revolution
 Early Pyrotechnolgy - Pottery
Early Pyrotechnolgy - Pottery
 Early Pyrotechnolgy - 2. First Technical Uses
Early Pyrotechnolgy - 2. First Technical Uses
 Smelting Science - 5. Smelting Details 2
Smelting Science - 5. Smelting Details 2
 Diamond, and other Carbon Specialities
Diamond, and other Carbon Specialities
 9.1.2 Problems with Alloying
9.1.2 Problems with Alloying
 Medieval and Modern Texts Concerning Crucible Steel
Medieval and Modern Texts Concerning Crucible Steel
 Iron in China
Iron in China
 10.4. Crucible Steel 10.4.1 The Making of Crucible Steel in Antiquity
10.4. Crucible Steel 10.4.1 The Making of Crucible Steel in Antiquity
 Units of Length, Area, and Volume
Units of Length, Area, and Volume
 4.4.1 Perfect Crystals and the Second Law
4.4.1 Perfect Crystals and the Second Law
 Lattice and Crystal
Lattice and Crystal
 Crystal Models
Crystal Models
 10.1.2 Copper
10.1.2 Copper
 6.2.1 Creamy or Chunky?
6.2.1 Creamy or Chunky?
 Smelting Science - 2. Charcoal Technology
Smelting Science - 2. Charcoal Technology
 Iron in Africa
Iron in Africa
 Gemstones
Gemstones
 Beer and Conquering The World
Beer and Conquering The World
 Last Charcoal Smelter in Germany
Last Charcoal Smelter in Germany
 Entropy
Entropy
 Sword Places
Sword Places
 Producing "Nirvana" Silicon or Nearly Perfect Silicon Single Crystals
Producing "Nirvana" Silicon or Nearly Perfect Silicon Single Crystals
 Overview of Major Steels: 1. Classifying Steels
Overview of Major Steels: 1. Classifying Steels
 Hardware Around the Making of Metals and Their Proper Names
Hardware Around the Making of Metals and Their Proper Names
 4. Mercury
4. Mercury
© H. Föll (Iron, Steel and Swords script)













