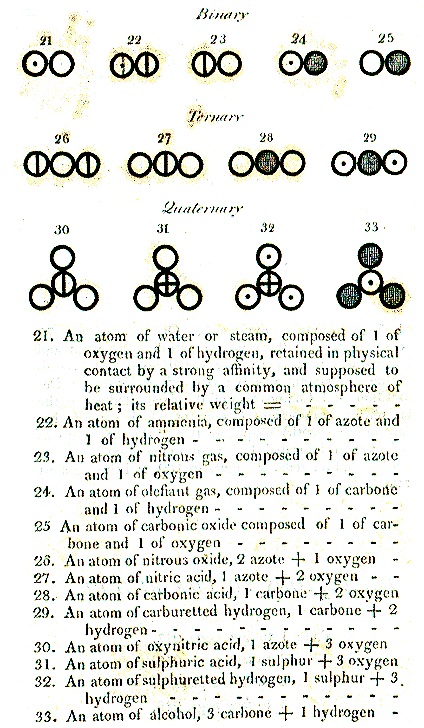|
Discovery of Atoms |
| |
A Long Time Ago... |
 |
...some ancient guy sat by the fire and started thinking. He wondered what would
happen if he took a piece of iron (or copper, or wood, or clay, or... ) and cut it into two equal parts. Well, that's hardly
something to wonder about. You get two pieces half as big as the starting piece.
The process only gets tricky when
you repeat it. Take one half, cut it again. And again. And again. And so on.
Of course, at some point he just couldn't
do it anymore with the tools he had at his disposal. Problem solved. He got a lot of small pieces. |
|
 |
That's when his wife cut in and did another cut. After all, women just can do
more delicate jobs than (most) men; just watch one putting on the war paint before she goes to some function. Our ancient
guy was fascinated and thought about his former girl friend, who, being particularly dexterous, could probably have cleaved
one of the tiny pieces in two once more.
Then he had a brain storm and realized that there is no real limit to cutting.
All you had to do was to imagine nimbler and nimbler cutters - small fairies, the very small fairies the fairies believed
in, and so on.
In other words: he invented the brain cutter, an early relative of
your brain microscope. |
|
 |
Wondering what you would get after applying the brain cutter many times to an object quickly
led to irrevocable brain warpage, however, forcing the cutter to become a professor. This was not a good career move, considering
that universities hadn't been invented yet.
Luckily, most everybody around then didn't get that far because after a
while of brain cutting, parting or cleaving, the cutter got wobbly and developed powerful urges to invade Gaul, invent beer, or, looking at cleavage elsewhere, do a bit of parting the wife down the middle
for a change and see what that would lead to. |
 |
Just one ancient Greek, probably a wifeless teetotaler who couldn't hold a sword,
cut all the way to the end and pronounced that at some point, whatever remained, could not be cut in half anymore. No way.
No matter how small the fairy or whoever else was conjured up to do the cutting. |
|
 |
That was good old Leucippus,
who lived around 450 BC - 370 BC. He claimed to be a philosopher so he didn't have to do real work. He wrote down what he
thought about the cutting business but his manuscripts got lost (they were probably used to kindle the fire needed for brewing
beer). We know of Leucippus only because Aristotle (384 BC
– 322 BC), another philosopher (the one who got everything
wrong) mentioned Leucippus' thoughts later in order to make fun of them. |
|
 |
Like most cranks, Leucippus had followers (often called disciples), and one of them was Democritus. He was born around 460 BC in Abdera in Thrace. Democritus
was rich, didn't need to work, and thus had time to write a lot of books (72!) about all kind of stuff and what he thought
about it. Unfortunately (or luckily), all we have today is a list of his oevre. The books themselves are gone. Aristotle,
of course, made fun of him, too. |
|
 |
Enter Galen of Pergamon, born around
130 AD and well-known for his medical interests and for writing an unbelievably large amount of stuff that has survived
to a considerable degree. He commented on the writings of Aristotle and in this context refers to the "atomists"
in some detail, in particular to Epicurus (341 BC – 270
BC; another atomist but more famous for promoting a happy, tranquil life style) and to Democritus and Leucippus. That's
why we know that Democritus and Leucippus held that everything is composed of "atoms", (atomos = indivisible)
and that empty space lies between atoms. The rest of their (science) philosophy
is pretty much BS, as we know now. |
| |
| |
| |
 |
 |
| Pergamon and people in Pergamon today, quite bored after 2.000 years. |
|
| |
| |
 |
Of course, a lot more people all over the world pondered the "cutting"
question or similar conundrums by heavy thinking and arrived at all kinds of answers. We just don't know about this because
they either didn't write it down or their writings are lost for good. |
|
 |
What some people know today (and communicate via Wiki) is that in India "schools of atomism"
may even date back to the 6th century BC. |
 |
My point is that just by "thinking", one might hit the basic concept
of atoms - but just as well all kinds of concepts that are simply wrong. Aristotle, for example, enlarged upon the "four
element" theory (fire, air, earth, and water) and claimed that those entities were not made of atoms, but were continuous.
He was also violently opposed to the idea of an "empty space"; a vacuum or "void" in other words, on
strictly philosophical reasons - and was utterly wrong on all counts! |
|
 |
Only with scientific methods, always involving experiments,
a definite and true answer to the "cutting" question (or any other "scientific" question) could be found.
Mankind thus first needed to "invent" science before progress could be expected. Galileo
Galilei (sometimes referred to as "father of science) and his buddies must be mentioned in this context - and now
we moved on almost 2.000 years and are in the 17th century. |
| | |
|
| |
After Galileo and Before Einstein |
 |
Aristotle's wrong views of matter won not only the day, but the better of 2.000
years. Why? People were weird or worse, religious. Take for example Thomas Aquinas (1225 - 1274; promoted to sainthood in
1323). He was the foremost proponent of "natural theology" within the system of scholasticism,
a method of "critical" thought dealing, among other things, with questions like: how many angels can dance on
the head of a pin? He was instrumental in promoting Aristotle to the all-time philosopher King, and thus assured that science
as we know it had to wait for a few hundred more years to escape from of the combined might of Aristotle + Church. |
|
 |
When Galileo defied
the church in 1633 (to his detriment and our advantage), the light of science began to shine but was still far too feeble
to solve the "let's cut things" question once and forever. However, a physical "theory" called corpuscularianism
emerged, with proponents like Rene Descartes or Robert Boyle. The great Isaac Newton also subscribed to corpuscularianism
and applied it to his theory of light (where he went wrong to some extent, however).
The names above are all heroes of science, still well known to all with a halfway decent education. Nevertheless, those guys were also frequently wrong. Newton for
example, while certainly a prime contender for the title "greatest scientist ever",
held rather weird views on many things; he believed in alchemy,
for example. Corpuscularianism, while promoting the (correct) view that all matter is composed of tiny particles or "corpuscles",
was flawed in many other respects and did not really introduce atoms proper. That had to wait for the emergence of chemistry
from the ashes of alchemy. |
 |
Enter Antoine Lavoisier,
the "father of modern chemistry". In 1789 he supplied the first list of elements
in his "Traité élémentaire de chimie (Elementary Treatise on Chemistry) defining an element to
be a basic substance that could not be further broken down by the methods of chemistry. But Lavoisier's elements weren't
proper atoms yet, not to mention that some of Lavosier's elements were "wrong".
More to that in this link. |
 |
In the context of the discovery of real atoms
(as opposed to philosophical constructs), the name John Dalton
(1766 – 1844) must now appear. |
| | |
|
|
|
 |
John Dalton
Portrait by Charles Turner (1773-1857) after James Lonsdale (1777-1839). Mezzotint. |
| Source: Wikicommons / United States Library of Congress's Prints and Photographs division
|
|
| |
| |
|
 |
In 1805, as a secretary of the Manchester Literary and Philosophical Society, he introduced
the concept of atoms to explain why elements always react in ratios of small whole numbers
(the law of multiple proportions), and a few other things. The law of multiple proportions is easy to understand. Imagine
a party with a number of boys and girls but 7 or 11 or 15 or m more boys (symbol B) than girls (symbol
G). After a while of mixing, couples form until all girls are paired. The reaction equation to this is 1B + 1G
= B1G1; the index "1" is "the small whole number" going with the reactions.
At a more exotic party at a more exotic place (paradise or hell) where every boy pairs up with exactly three girls, we would
have 1B + 3G = B1G3.
In the first case we would have m boys left over, in
the second case we'd have a riot. |
|
 |
And that is exactly what happens when chemical reactions take place. The numbers involved
are always small and always integers, and some original component is usually left over. Easy to explain with atoms; entities
that come in integer numbers and pair up in integer numbers. |
| | |
|
|
|
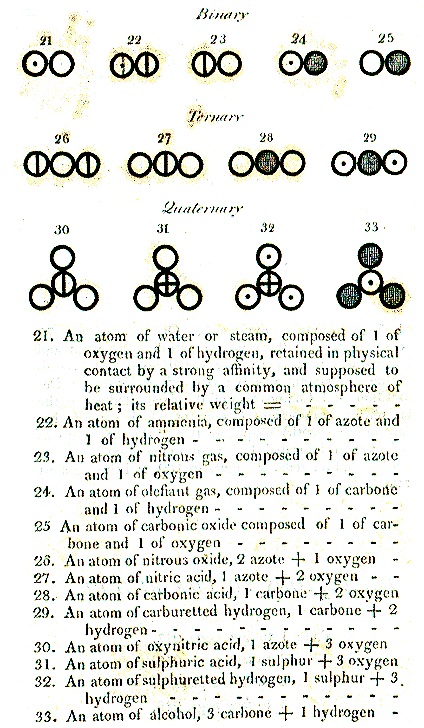 |
| From John Dalton's 1808 book: A New System of Chemical Philosophy that contains his atomic
theory based on scientific experimentation |
| Source: Public domain in the United States |
|
| |
| |
|
 |
That looks rather like modern (symbolic) pictures of molecules in chemistry book. Except that
modern books would not contain all the mistakes you can find in the picture above.
Dalton got plenty of things wrong
in detail. He couldn't help it, he simply couldn't know better (in contrast to philosophers lilke Aristotle). While various
deities have made many pronouncements about this or that via prophets, saints or offspring, none of them has ever bothered
to reveal something useful, like the second law, how to make
beer (plus a refrigerator) or, in Dalton's case, that a water molecule consists of one oxygen and two
hydrogen atoms. |
 |
Of course, not everybody subscribed to this view. Beer, for example,
usually also comes in integer numbers. You get 5 hogsheads or
13 bottles, and you pay in integer dollars and cents. That doesn't mean that beer itself
needs to be an "integer" quantity, it only means that a "book-keeping" system that is based on "atoms"
of the stuff might be the most simple way to deal with certain aspects of beeriness. Many early physicists believed just
that about atoms. Nice for book-keeping but without substance. |
|
 |
Dalton did could not know all there is to know about atoms. He had no idea,
for example, how large they are, except that they must be rather small. The first answer to that rather important question
came from Johann Josef Lohschmidt, an Austrian scientist,
who determined the size of the molecules in air rather well in1865 by arguments based on measurements.
The "Loschmidt constant" (or
Loschmidt's number) is named after him. Sometimes, particularly in Germany, people refer to that number (incorrectly) as
Avogadro constant. Lohschmidt was an Austrian, after all. |
|
 |
Loschmidt befriended his younger university colleague Ludwig Boltzmann. We might safely assume that both scientists where firmly convinced of the
existence of atoms. Boltzmann, after all, dedicated his scientific life to "reducing" thermodynamics to a branch
of mechanics by looking at the (average) behavior of a large number of atoms / molecules in matter". Lohschmidt's criticism
of his early efforts finally lead him to his statistical concept
of entropy, and thus to the discovery of the basics of the second law. |
| |
| |
| |
 |
| Ludwig Boltzmann (1844 – 1906) |
|
| |
| |
 |
This could have been it. Around 1880, let's say, the reality of atoms was rather
firmly established even so nobody had ever "seen" one. However, not everybody was convinced and big fights developed,
essentially because of "philosophical" problems. |
|
 |
We will never know if the rejection of his marvellous work by other titans of science was
instrumental for Boltzmann's suicide in 1906. |
| | |
|
| |
Are Chemists Smarter than Physicists? |
 |
Well - emphatically no! (I'm a physicist). Looking back one might get this (slanderous
and completely irrelevant) impression, however. The chemists didn't doubt the existence of atoms long before the physicists.
In 1860 they had a big congress in Karlsruhe, Germany, and the "atom" won in a big way. Of course, there are always
a few who don't give up opposition to anything new, but after Karlsruhe no chemist listened to them anymore. |
 |
Famous physicists begged to differ. A particular outspoken opponent was not just
a physicist but the founder, if you can attach such a label to a single person, of physical
chemistry: Wilhelm Ostwald
- a titan of science who we meet many times in this Hyperscript. |
|
 |
What one needs to be aware of is that chemistry was not yet that part of physics
that "deals with simple solutions to the Schrödinger equation" as many physicists see it nowadays, but a
science in its own right and somewhat mysterious to physicists. The intersection between physics and chemistry thus was
a particular difficult field to plow, and Ostwald is and will be remembered as one of the heroes in this field. |
|
 |
Ernst Mach was another outspoken opponent
to the atomic hypothesis, but so were other famous physicists, including no less a celebrity than Max Planck, the "father" of quantum theory. With quantum theory proper, coming
into its own around 1920, one could calculate the properties of atoms and molecules in fine detail and explain everything
there is to explain about atoms and how they react with each other, making chemistry a well-understood branch of physics
(in principle). |
 |
Why did all these guys, who were not exactly stupid people, oppose the "obvious"
so much? Because, in contrast to the chemists who did essentially the "book keeping"
of things that happened on a small scale in space and time, these guys were worried about the big picture. If there are
just atoms, following the immutable laws of (Newtons's) mechanics as Boltzmann taught, the universe as we seemed to know
it was in danger. In fact, it was doubtful if it could even exist. |
|
 |
The laws of mechanics do not change if you reverse the direction of time. Without old-fashioned
entropy defined as some entity in its own right that could only grow larger as a law
of nature, there was no direction of time anymore. There should be no difference between the future and the past, and that
was a bit hard to swallow.
Moreover, if some atoms in some ensemble would happen to be in the same state of motion as
they had been at some other time before, they would be forced to do exactly what they
did before; the whole system would be locked in a repetitive circle.
And that simply didn't make any sense. |
|
 |
These (and many more) objections to "statistical thermodynamics" that followed from the existence of atoms were (and are) irrefutable
serious arguments. The discovery of quantum mechanics solved these conundrums to some extent, but remnants of these questions
are still loitering in the dirtier rooms of the physics basement, waiting for that final answer. |
|
 |
So Ostwald, Mach, Planck and many others weren't stupid - they were doing science. There is
no such thing as a perfect scientist, who always pronounces the truth and nothing but the truth, and recognizes on first
sight if theorems of others are true or false. There are only imperfect and fallible humans, that eventually arrive at the
truth together, by thinking and experimenting independently, and refuting and fighting
each other. It's a messy process, bouncing back-and-forth, getting stuck in blind alleys
for a while, appearing beautiful for some and ugly to others - but in the end the truth will prevail.
All these guys
who were violently opposing Boltzmann and the other "atomists" eventually turned around and acknowledged the reality
of atoms because they didn't just believe but were open to arguments. In contrast, we are still waiting for a Pope to become
a Muslim or at least a Protestant (or vice verse). |
 |
Why did "the atoms" win in the end? Because of Albert Einstein. Einstein is not just a contender to the title "greatest scientist ever"
like Newton but actually won the (informal) election for this title in 1999. He is, of
course, well-known to all and sundry because of his theory of relativity and his hairdo (not necessarily in that order).
His only Nobel prize (he should have obtained at least six), however, he obtained for the explanation of the photoelectric
effect in 1922. |
|
 |
Einstein established the photon, the light "particle"
(or "light atom") with his Nobel prize work, and this simply made no sense at all without regular atoms. Long
before that, in 1902–1903, he had published two papers attempting to interpret atomic
phenomena from a statistical point of view. These papers were the foundation for the seminal 1905 paper on Brownian
motion, the incessant jittering of small things that you could see with a
good microscope. He showed that Brownian movement can be construed as firm evidence that molecules exist. Going on from
there, he also showed that the well-known phenomena of diffusion
emerged straight from having atoms jump around. |
|
 |
Everybody familiar with all these phenomena and capable to follow the math (i.e. all physicists),
had no choice but to acknowledge the reality of atoms. And that's what they all did.
Nowadays we can see and manipulate single atoms and calculate all there is is to know about them. |
 |
Yet the objections raised against the mechanical view of the universe that is
inextricably tied to the concept of atoms have not gone away completely. Some have been resolved, to be sure, but others
are still lurking in the background, possibly a bit modified. |
|
 |
Roger Penrose, for example, figured
out that the universe as we seem to know it, must have started - at the "Big Bang" - in a state with extremely
small entropy. That's essentially the reason why entropy always increases. However, starting an universe that way is an
extremely unlikely thing to happen.
Nobody has an answer to this (and many more "universal" questions) that
are tied to the old questions of the anti-atomists. Only time (a consequence of the second
law) will tell. |
| |
|
© H. Föll (Iron, Steel and Swords script)