|
10.3 Iron and Steel in Early Europe |
|
10.3.1 Technology Transfer |
| |
Spreading the Technology |
|
Way back I asked: Who smelted iron
for the first time? When and where? And how, exactly?
I didn't really answer these questions because neither I nor
anybody else knows for sure. If we discount the more or less accidental production of some of iron during copper smelting
and the few if spectacular objects from early times, the best we can say is that the systematic production of iron in bloomeries
started around 1200 BC somewhere in Anatolia. To credit the Hittites with the "invention" of iron, as archeologists
were inclined to do in the past, is most likely overstating the case. True, the Hittites left a lot of written documents where iron is mentioned but practically no iron artifacts to speak
of.
If we believe some ancient Greeks like Strabo (65 BC
- 25 AD), we should look for the Chalybians, who possessed the secret of steel making. Hence Chalyb=steel
in Greek (and Latin), in contrast to "sideros"=iron. Besides the Greek references we do not know much about these
guys but there are strong hints that they lived in the general Colchis area at the Black Sea coast as shown in the map below.
The term "Chalyb" might come from the Hittite "Khaly-wa", meaning land of "land of Halys",
which is also shown in the map below. The Halys river (now know as Kizilirmak) starts around there.
Note that the Greeks
wrote their stuff about 1000 years after iron became generally known and used. Their differentiation between iron and steel
might indicate that it took that long before metal workers could select and manipulate the stuff from the bloomeries in
ways where this distinction became meaningful, including proper forging and quenching for getting hardened steel. Whatever.
We must treat all these not-so-old writings like rumors and not as literal truth.
In this module I will give a very
cursory look at the spreading of the technology and the trade with iron and steel "halfware" in mostly Europe.
The "Eastern" technology of crucible steel will have its own module.
This chapter will be very patchy because the knowledge about these topics is patchy and because I want to keep this short.
It goes without saying that most of what follows contains a lot of educated guessing. |
|
 |
What we do know is that iron technology spread rather quickly
in the Mediterrean after 1200 BC. The source seems to be present-day Turkey. One might assume that the collapse of the Hittite empire around 1200 BC displaced a lot of people, including skilled iron engineers,
who were now free (or forced by circumstances) to move about and offer their skills to all and sundry.
The map below,
drawn after the map in Buchwalds's book, is rather
trivial. What one sees is that the knowledge about iron spread by "water" - along major rivers or by seafaring.
Small wonder - that's how people did get around for long distances. After you walked from Kiel in North Germany to East
Anatolia once (and survived), you tended to take a boat for getting back.
The map also shows a few major places for
iron production that I will discuss here. |
| |
|
 |
Spreading
iron technology means that people in some area learned how to smelt iron
and how to forge it into something useful. Essentially all you need is charcoal, iron ore and some clay for building the
bloomery, plus a hearth, an anvil and tools for forging. If you can procure those things, that odd traveler who came by
boat from far away (on his own or as slave) will show you how to smelt and forge iron in exchange for some money or the
opportunity to stay alive. |
|
 |
Chances are he won't succeed on his first try. As we have seen in all those modules before,
you must do your smelting just right. Your gangue is "wrong",
your bellows are leaky or especially efficient, your charcoal is too reactive, and so on, and and all you produce is slag,
useless cast iron, or small useless blooms.
Eventually, after optimizing local processes, you will be able to produce
some iron. But some areas / people will produce better stuff than others, not to mention that some produce their iron more
cheaply than others. If for a given product there are differences in quality and / or price you have all that is needed
to start some trading.
In other words: the spreading of the technology did not preclude
trading. You might make your own iron but you will still import some from "specialists" who make a particular
good (or cheap) variety. If conditions were right, you may even have given up your own production in favor of imported stuff.
The exporters of iron / steel owed their success not just to secret procedures or some magic. More likely they had
better ingredients, a better organization, and a more motivated and skilled workforce. It is simply easier to make good
iron with nice "clean" siderite than with dirty bog iron,
for example. It is thus no great surprise that certain areas specialized in iron working quite early, and that their iron
was traded wide and far. |
 |
What follows are a few remarks about some places of iron production. Taken together,
one can get a feeling for what has happened. |
| | |
| |
The Etruscans - Populonia and Elba |
 |
I have mentioned Populonia and Elba
before in the context of slag production during metal smelting. The whole
area - Elba and the Italian coast region just across - was an ancient metal center, sort of an early Pittsburg, run by the
Etruscans and later, as the Etruscans became absorbed in the Roman Empire, by the Romans. |
|
 |
Ancient writers remarked on it, for example the Greek Diodorus (60 BC - 30 BC):
"Off the city of Tyrrhenia known as Populonium
there is an island which men call Aethaleia (=Elba). It is about one
hundred stades distant from the coast and received the name it bears from the smoke (aithalos) which lies so thick about
it. For the island possesses a great amount of iron - rock, which they quarry in order to melt and cast (philosophers
never get it right) and thus to secure the iron, and they possess a great abundance of this ore. For
those who are engaged in the working of this ore crush the rock and burn the lumps which have thus been broken in certain
ingenious furnaces; and these they smelt the lumps by means of a great fire and form them into pieces of moderate size which
are iron their appearance like large sponges. These are purchased by merchants in exchange either for money of for goods
and are then taken to Dicaerchia (=Puteoli, Campania) or other trading-stations, where there
are men who purchase such cargoes and who, with the aid of a multitude of artisans in metal whom they have collected, work
it further and manufacture iron objects of every description." |
 |
The area, in particular Elba, had copper ore and huge amounts of iron ore. The
Etruscans already had a large copper smelting operation going there and smoothly adopted to iron at the latest around 600
BC, it appears. I have already stated that 2 Mio tons of slag have been produced in about 500 years, some of which could
have been from copper smelting since this also produced iron-rich slag. This number is actually debated and might be considerably
smaller but that doesn't matter for the three points I like to make:
- I could not find a single picture of an iron object or of the remains of a smelter from that area. This was simply not
something one particularly looked for when the area was "cleaned" of its slag in 1915 - 1943.
- This kind of negligence used to be the case for about all metal-producing regions.
In 1980 or so, almost no old smelters / furnaces have been found and dug up, even so millions must have be around in Europe
alone.
- This has changed dramatically in the last 20 years or so. An increasing number of archeologists, archeometallurgists,
lay-people, and guys like me are looking into the issue.
|
|
 |
The Etruscan graves found under the slag deposits attracted far more attention than the slag
for obvious reasons. Fortunately, a few iron objects were found in these graves and thus preserved and described (but rarely
analyzed). Of particular interest here are the "spieti" (Italian) or "obelsikoi"
or "oboli" (Greek), looking like thin (barbecue) spits of iron or bronze. They can
be seen as a kind of currency, and that will not be the last time we encounter iron pieces as a kind of money. |
| |
|
| |
 |
We can be reasonably sure that the oboli and any other iron products from this region consisted
of inhomogeneous forged iron, with varying concentrations of carbon and other elements (like phosphorous) and always a lot
of slag inclusions, typically elongated from smithing. Oboli, according to general theory, were a kind of currency, sort
of very elongated coins, that you could use as money or directly as raw material of a certain value. You could forge a knife
from am oboli, or you could use them as spits for roasting your chicken on the fire. Some modern archeometallurgists even
subscribe to a heretical point of view: Oboli were nothing but spits for cooking on an open fire, as was the custom in those
days. What else but iron would you use for a spit?
Below is what real spits looked like - they could easily have been
made from oboli. |
|
|

| | Iron spits for roasting meat |
| Source: Photographed in the Heraklion
Museum, Crete |
|
| |
Ferrum Noricum |
 |
The Roman empire was rather large compared to the average distance people, goods
and news could travel per day. It's influence on present day Germans (Italians, French, ....) can still be seen and felt
if you have sensitive and trained receptors. The Romans were not only highly organized but used the specialities of the
conquered territories to their best advantage. They also left a lot of written stuff and that's how we know about the "famous"
Ferrum Noricum; the superior iron (actually steel) from the province Noricum. |
|
 |
Noricum used to be an independent kingdom (we believe) that became (peacefully)
incorporated into the Roman empire in 16 BC. It coincided with much of what now is Austria. Ancient Noricum included Hallstatt, a place that gave the name to a whole culture (Hallstatt culture, 800 BC
- 600 BC or Early European Iron Age). It also contained a place (still) called Erzberg
I ("ore mountain") close to the present town of Hüttenberg
(cottage mountain or possibly smelter (smelter=Hütte) mountain). That's where Ferrum Noricum was produced during the
Roman times and somewhat earlier during the (late) La Tène period (following
the Hallstatt culture; 450 BC - the Roman conquest in the 1st century BC). There is another place called Erzberg (II) in
Noricum that is also known as an iron producing place - but not before early medieval times. Iron production around Erzberg
II went on until recently; it should not be confused with Erzberg I.
The Romans run a kind of large-scale well-organized
iron industry out of (present -day) Magdalenenberg or Magdalensberg, the "city"
close to Erzberg / Hüttenberg. It is all in the map below: |
| |
|
|
 |
If you are now a bit confused about all these folks and cultures (Romans, Austrians,
Celts, Hallstatt, La Tène,...) that's as it should be. Let's simplify and just look at two cultures: The Celts, having
their heydays from about 600 BC - 0 AD, and the Romans who absorbed and replaced the Celts around 50 BC and later. The Hallstatt
and the La Tène culture (and others) are just expressions of Celtic "super" structures.
The Celts are
supposed to have been the first "iron masters" in Northern Europe; the link goes a bit deeper into the Celtic
Culture.
You might be inclined to associate Celtic culture with Ireland, Wales and Scotland in more recent times because
people there speak Gaelic, a kind of left-over Celtic tongue. Historians are not so sure. Celts and Celtic culture did survive
to some extent in these places after it was all but extinguished on the continent, but it did not come from there. |
|
|
 |
The Celts may have discovered and explored the Hüttenberg ore deposit in
the first century BC. They might have had a cultural center on Magdalenenberg, working and trading the iron from there and
thus making it known to the Romans. The Romans eventually - more or less peacefully, it appears - took over and lots of
artisans settled there, making Magdalenenberg at major hub for iron making and trading for centuries to come. It is possible
that the fame of Ferrum Noricum owes just as much (or more) to the organizational skills of the Roman and to the cunning
of their smiths than to the quality of ore and iron.
We have known for quite some time that Ferrum Noricum was famous
in antiquity because the Romans wroteabout it. Quite a lot, actually. However, besides
the descriptions in the literature not much was known about this iron from direct evidence. That has changed only recently
and I will get to that. First, however, I like to give a few quotes from ancient references to Ferrum Noricum. That is easier
said then done because all sources on Ferrum Noricum mention that there are numerous
references to its outstanding quality in ancient literature but very few actually supply quotes. Here is what I found (sometimes translated by me):
|
|
 |
Ovid (43 BC – 17/18
AD), was a Roman poet who is still well-known in educated circles.
He wrote: "...durior
[...] ferro quod noricus excoquit ignis..." (...harder than iron tempered by Noric fire...).
One might
conclude that he distinguished steel from iron and knows about "tempering".
That word, however, has changed its meaning in the course of time from "quenching" to heating up again. So what
Ovid meant depends on what tempering means. |
|
 |
Titus Petronius(14
AD - 66 AD), the author of the famous "Satyricon", has one of his figures, Trimalchio, praise his cook in strong
terms, finishing with: "... and since he is so gifted, I presented him knifes made from Ferrum
Noricum that I brought back from Rome...". The knifes are fetched and admired for their sharpness.
There
is more to this quote then meets the innocent eye. Trimalchio, a somewhat vulgar "noveau rich", shows his money
by presenting his cook with a gift of not just one but several very expensive knifes, normally far too good for regular
kitchen work. It's like you giving your cleaning Lady a Porsche instead of a subway token so she can get to work.
|
|
 |
Galen of Pergamon (129
AD – 200/216 AD) we have met before. He was a
prominent Greek-speaking Roman physician, surgeon and philosopher; arguably the most accomplished of all medical researchers
of antiquity. He developed fine surgical instruments that needed to be made from the best iron "like
Ferrum Noricum" if they should be serviceable. |
|
 |
Marcus Valerius Martialis
(known in English as Martial; 40 AD – 102 /104 AD), was a Spanish poet who published short, witty and satirical poems
in Rome. He was born in Bilbilis and praises this town as the place were first-rate steel blades originated from, "better than the iron of Chalyb and Noricum". In his
times the iron from Chalyb was already a myth and not real.
Martial also refers to the use of hunting spears with tips
of Ferrum Noricum that were used by the emperor Domitian just for for playing around with! Once more a show of using the
best and most expensive for inferior purposes, just to show off! |
|
 |
Pliny the Elder (23
AD– 79 AD) or Gaius Plinius Secundus, famous Roman author, naturalist, philosopher, naval and army commander (those
guys were good at multi-tasking) writes:
"...the high quality of iron goods is ensured by the ore as in the Noricum, by the working as
in Sulmo, or by the water as in the places given..."
One of the "water" places is the Martial's home town Bilbilis from above.
Tremendous confusion ensued
from that statement. Can you make great iron / steel from inferior stuff by working
it in special ways? (Yes, but only up to a point). Is it enough to have good ore for making good iron / steel? (Not really).
Is the result of quenching, polishing, etc., dependent on the quality of the water you use (definitely not! - at least as
long it is half-way clean water).
The water myth comes up even today on occasion, e.g. if a Spanish merchant tries to
convince a tourist that they have "special water" there, making for swperior swords when used for quenching. |
 |
The 20th century literature about Ferrum Noricum is rather murky, mostly because
it was based on discussing old literature. If we want to shed some light on the topic, it is illuminating to ask a few pointed
questions:
- The Ferrum Noricum of the Romans made its literature debut around 0 BC / AD. But the people in the Noricum were smelting
iron long before they became Romans. We must ask: Who? How? How early? How much? How well?
- Could it be that there were two kinds of Ferrum Noricum? Regular and superior? After all, it appears
that there was a major iron industry in the Noricum, making a hell of a lot of weapons for a large part of the huge Roman
army plus plenty of everyday objects. All that iron could not have been extremely precious and costly. But then we also
have the expensive prestige stuff mentioned by the literati. A case like Fiats and Ferrarris from Northern Italy? 2)
- Why was it somehow easier to make good iron in the Noricum then elsewhere?
- What do we know about Ferrum Noricum from archeological digs plus metallurgical analysis of artifacts?
There is the stuff for an involved and lengthy PhD thesis. Applications from suitable candidates who are able to pay
all expenses (including those of the Prof. in charge) are welcome. Until we get that research done, I will give you just
short answers. |
|
 |
1. The people in Norica were Celts before they became
Romans. They did produce iron in Norica but not necessarily only in Hüttenberg. There are indications that the Celts
discovered the Hüttenberg "iron hat" (gossan)
in the second century BC and then exploited that source of minerals. If the Celtic iron / steel from the Noricum was better
than Celtic iron /steel from elsewhere is an open question, it seems.
The iron industry in Hüttenberg went strong
ever since the Celts started it until about the fall of the (Western) Roman empire in 454 AD. After a 500 year break (the "dark ages"), operations
resumed in the 11th century and were kept going until 1978. |
|
 |
2. I owe this question to Gerhard Dobesch3), and it makes some sense to me. The metallurgical information to date does not unambiguously
support these claims but one might speculate that:
- Particular good ore (see below) plus special skills in smelting and working the bloom produced better than average but
"cheap" regular Noric iron in bulk.
- Highly specialized smiths could produce superior and very costly stuff from the regular Noric iron. They knew how to
pick different grades of iron or steel from the supply, and how to use theses materials for forging special layered composite
structures (as opposed to fire-welding random pieces). They even knew how to carburize and case harden the outer layers.
Some more recent metallurgical investigations of Noric iron (see below) point in this direction but so far the "two
kinds of Ferrum Noricum" hypothesis is just speculation. |
|
 |
3. The ore found in Hüttenberg is essentially
siderite or iron carbide (FeCO3). Not the richest
of ores but particularly easy to smelt as people find out right now once more. Why? I shall leave that questions open for
the time being (meaning I don't know the answer yet). An analysis of what one finds in roasted
ore from the area as given in Buchwald's book yields:
- 50 % - 54 % Fe
- 6 % CaO (quicklime)
- 5 % MnO
- less than 0.0 8 % P and S.
This is not a bad mix for making "good" iron. It is often assumed that the relatively high manganese (Mn)
concentration makes that ore so special because some manganese in the iron is always beneficial. Quicklime might be good for making slag. It's not that easy, however. Manganese oxide is
not so easy to reduce and does not ensure sizeable manganese concentrations in the smelted iron as we shell see somewhat
below. Nevertheless, siderite ore is easier to smelt in a bloomery and produced better iron than, for example, bog iron. |
|
 |
4. So far there aren't many analyzed Noric iron
pieces but their number is going up as I'm writing this. This is due to the ever increasing interest in ancient metallurgy
and the many digs that have been started in recent years. New archaeological excavations at Hüttenberg started in 2003
under the direction of Brigitte Cech. Earlier results are well published in Harald
Straube's book |
 |
Let's have a quick look at the pertinent results of analyzed Ferrum Noricum. I
will only use two sources. There are undoubtedly more but as far as I'm aware of them nothing really new is added to what
follows. |
|
 |
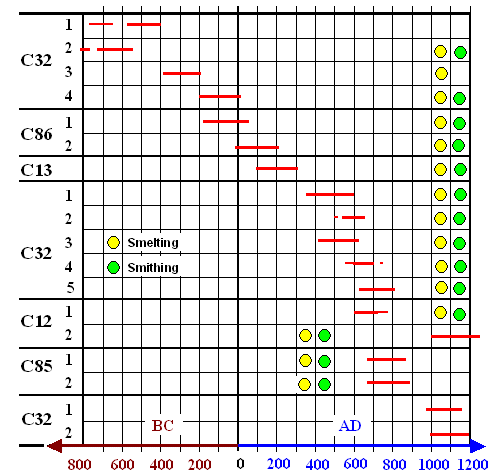
First we look at three swords
likely made from Ferrum Noricum. All we need to do is to look at an old picture,
augmented with some new data: |
|
|
 |
| Carbon (C) and phosphorous (P) concentrations in Celtic swords from about 500 BC - 250
BC. Three swords are (probably) made from Ferrum Noricum |
| Source: Data are from Buchwald's
book; p. 116 |
|
|
 |
Vagn Buchwald has reasons to assume that the three Celtic swords marked were made
from Ferrum Noricum. His conclusion is based on a thorough analysis of the composition
of the slag inclusions. If he is right, the three swords do have a relatively high carbon
concentration and a low phosphorous concentration, a possible mark of high quality.
But that is also true for some other swords. From the data given Ferrum Noricum is not
outstanding.
Buchwald's final words are of some interest: |
| | |
The general impression of Celtic swords,
here covering a period from roughly 650 to 100 BC, is that the blade was normally manufactured from a single
iron bar of no particularly good quality. .... No deliberate attempt at carburizing - or decarburizing - can
be observed. It happens that a pearlitic hard steel is located along the edge or at the point, but just as often the soft
ferritic zones are located here. The laminated textures are due to the original heterogeneous nature of the bloom and bars,
and not to some deliberate piling of material with known carbon or phosphorous content ....
Common to all the Celtic
swords is the extensive cold working that has taken place." |
 |
Second we look at results
from two kinds of early "Austrian" steel, published by H. Preßlinger and colleagues 4). What they investigated in some detail were:
- About 30 specimen from around 100 BC and found North of the Danube In Gründberg,
part of the present Linz (indicated on this
map). The objects found in 4 depots were mostly "utility" things like hooks or hammers and not made from Ferrum
Noricum.
- About 10 objects from 100 BC - 100 AD found in Magdalenenberg, right in
the heart of the Ferrum Noricum region.
|
| |
 |
Major results for the Gründberg specimen
were:
- Hardness values (Vickers) ranging from 80 (quite soft) to 680 (extremely hard) and were not always correlated to the
observed structure.
- Structures are typically ferritic (soft) or ferritic-pearlitic (medium hard). Occasionally grain-boundary ferrites (Widmannstätten structure) were observed, and martensite in
one case .
- Manganese (Mn), sulfur (S) and copper (Cu) concentrations were typically below 0,01 % and thus of no consequence to
properties.
- Phosphorous (P) concentrations between < 0.01 % and 0.7 % were found, with very strong variations inside one specimen.
Preßlinger concludes that the objects were made by fire welding of many thin layers with conscientiously
picked grades of steel, in particular phosphorous-rich steels for the intended hard parts.
It is not for me too put some doubts on the conclusion - but compare to what Buchwald has to say about
"piling" right above. One particular interesting result, as far as I'm concerned, are measurements that offer
a solution to the puzzle contained in these data: ferritic or ferritic-pearlitic steel - but very large hardness? How is
that possible? The picture below gives the answer. |
|
|
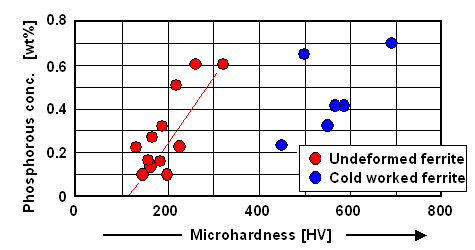 |
| Phosphorous concentration and hardness achievable by cold working, |
| Source: Redrawn (and augmented) after H. Preßlinger
et al. |
|
|
 |
It's the phosphorous, of course. The red and blue dots are measured
values, the red line gives (roughly) the expected relation
due to a basic solution hardening mechanism for undeformed iron. Cold working phosphorous steel increases the hardness values
rather dramatically. That is conceived as being good.
However, the steel might now be rather brittle and
given to "cold shortening". Nothing seems to
be known about that. |
|
 |
Major results for the Magdalenenberg specimen
(knifes, axes, hammer) were:
- Structures span the range from ferritic to martensitic. No hardness values are given.
- Phosphorous (P), Manganese (Mn), copper (Cu) (and presumably sulfur (S)) concentrations
are very small and supposed not to influence properties.
- Carbon concentrations vary substantially from specimen to specimen (between 0.03 % and 1.5 %) and within one specimen
(e.g. 0.04 % - 0.16 % on the low side, or 0.52 % - 1.5 % on the high side) with about half of the specimen found on the
large side.
- Some specimen have large amounts of slag inclusions; not a sign of high quality.
- All specimen were supposedly made by fire welding several pieces.
- "Case hardening" was probably
used.
|
 |
What do we learn? First of all, to be careful
with interpretations. There is some disparity between Buchwald's and Preßlinger's results. Of course, both could
be right - the number of specimens investigated is far too small to allow full generalizations.
What we might be able
to conclude is that Ferrum Noricum was generally low in phosphorous but also in manganese.
The manganese oxide contained in its ore thus does not make for better iron because appreciable amounts of beneficial manganese
end up in the iron. It quite likely makes for better smelting, however, maybe by somehow helping to produce "better"
slag? It is an open question
at present.
"Classical" wrought iron with a low carbon and phosphorous
concentration might be inferior to iron with an appreciable phosphorous concentration, i.e. phosphorous steel. Wrought iron
or more or less pure ferrite, in other words, is simply not as hard as phosphorous steel. The latter cannot be hardened
in the "usual" way but can become quite hard by cold working. Phosphorous steel is reported to be difficult to forge or easy to forge,
take your pick. A pure carbon steel with proper care to carbon concentration and hardening would be superior, though - provided
it does not contain too many "bad" slag inclusions. |
|
 |
In other words: It is possible that very
good smiths on occasion could make superior steel objects from very
good Ferrum Noricum, while only normally good steel resulted from normal Ferrum Noricum
(a very good smith never makes anything bad). It is, however, hard to judge the fighting quality
of an old sword by a metallurgical analysis of just a small portion that usually does not include the edge. "The harder
the better" is certainly not generally correct. |
 |
I haven't found other comparisons between Ferrum
Noricum and "Ferrum normalium", the iron / steel produced elsewhere. At present we must admit that
we do not really know what exactly made Ferrum Noricum so special. Maybe the bulk of it wasn't all that great after all,
and only some very expensive objects, made by special smelters and smiths, made it into the literary fame that started the
whole craze.
I have no problem to subscribe to the "two kinds of Ferrum Noricum"
hypothesis but will not commit myself before more results are in. |
| | |
|
| |
Snowdonia |
 |
The Snowdonia National Park in Northern Wales does contain a few early smelting
places (plus some from far later times) - just like a few thousand other areas in Europe. The only thing remarkable about
Snowdonia is Peter Crew and his crew (couldn't resist). Peter is
employed by the Snowdonia National Park Centre; here is the address (couldn't resist once more): Snowdonia National Park
Centre, Plas Tan y Bwlch, Maentwrog, Blaenau Ffestiniog, Gwynedd, LL41 3YU, UK.
Peter decided to do some smelting experiments:
"From 1983 to 2007 a series of nearly one hundred ironworking experiments were carried out, primarily
to provide data for the characterization and quantification of the slag from the excavations". He also decided
to eschew learning about the science of smelting first and thus to go about it like the local yokels 2500 years or so ago:
"It is very easy, but potentially misleading, to use technological tricks from later processes
or other cultures". Use the materials you find locally, built smelters with it, following the "blue-prints"
of what you dug up instead of learning it from the master, do not use modern equipment of any kind, and learn by trial and
error. The result is: "Our early smelts were a disaster". |
|
 |
Several highly readable articles appeared, and in 2010 Peter Crew published "Twenty-five
years of bloomery experiments: perspectives and prospects" 5); the quotes
above are from this paper. Peter also became quite scientific in the meantime, witness one of his newer
papers.
Of interest for us here is to learn that 25 years of experimenting did produce a lot of interesting results
- some of which are shown below - and definitely helped our understanding of early iron technology. However, Peter Crew
did not yet reproduce exactly what he has dug up. In essence, Peter and his co-workers learned a lot from all their experiments,
in particular that the Golidlock Principle strictly applies.
Everything needs to be just right if you want to do a high quality smelt. |
|
|
| |
|
Peter Crew investigated the influence of a lot of parameters on smelting.
Besides playing around with the smelter geometry and air blowing parameters, they looked in particular on different kinds
and grades of ore, flux and charcoal. Some quotes concerning results can be found in the "Odds and End" link. |
 |
Moreover, Peter Crew also looked into processing
the bloom, from refining to making billets, bars, and finally artifacts like knife blades. He needed the help of
blacksmiths for doing that and learned that present-day blacksmiths display various degrees of skills when faced with iron
blooms. My feeling is that none of them would be up to the standards of an ancient smith. But then, how could they? They
have to re-invent all the trade secrets that were passed on from master to apprentice for millennia before they became lost
in more modern times. |
|
 |
Peter Crew and colleagues produced a wealth of insights and data
from their many experiments and from checking with written accounts. I will give you just two pictures out of many. First,
let's look at the average size of blooms as a function of time. Bloom size was limited for two reasons. First, in the beginning
of iron technology you were happy to get a bloom at all. In small furnaces, run without much experience, you tend to get
a small bloom. Second, blooms too large cannot be refined by hitting them with muscle-powered hammers.
Now let's look
how bloom size developed (in England) between about 500 BC and 1600 AD: |
| |
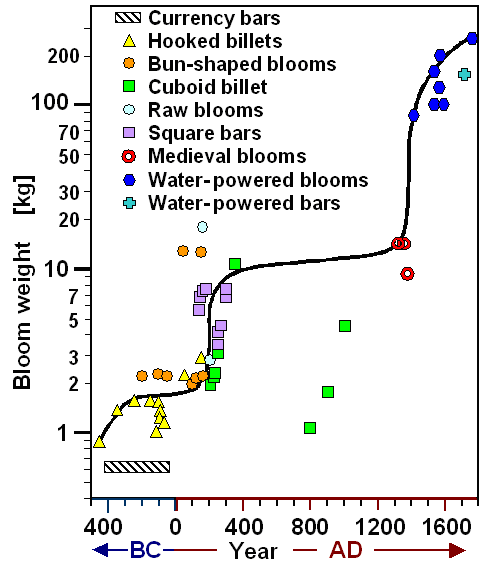 |
| Bloom size (logarithmic scale!) vs. time as deduced from products |
| SourcePeter Crew |
|
|
 |
Around 500 BC people in England and Northern Europe started smelting iron in relevant
quantities, Bloom sizes tended to be rather small, probably limited by technology. With growing experience, bloom size settled
around 2 kg for quite some time. That is an easy-to-work small size but large enough for making a few small object or a
knife blade or two. Iron itself was mostly traded in the form of billets,
bars, currency bars,
the latter with weights around 500 g.
Around 250 AD a "quantum leap" occurred. Bloom size increased about
5 fold to around 10 kg. New ways of forging must have been used. What we see is probably the Roman experience with running
a well-organized iron industry. This allows, for example, to split a 12 kg bloom into 2 or 3 pieces while still hot. With
enough smiths and hearth places around and in working conditions, the pieces of a large bloom can now be tackled.
A huge leap forward occurred around 1500 AD. Huge blooms of 100 kg and more appear routinely. The innovation
in this case is the water-powered hammer that could deal with blooms this large. Look at the following picture to get an
idea of what that means: |
| |
|
 |
| Refining a big bloom with a "water hammer". |
| Source Peter Crew |
|
|
 |
This is a bloom weighing in at at around 40 kg. You could not impress it very
much (in both meanings of the word) with an arm-wielded 2 kg hammer. It will, however, yield to that monster hammer that
is powered by a substantial water-wheel. |
 |
Now let's look at some data describing the efficiency of smelting. The curves
below show how much iron you get in various smelting experiments (A - E), and how much of that is left over after various
forging steps, relative to the amount of charcoal used |
| |
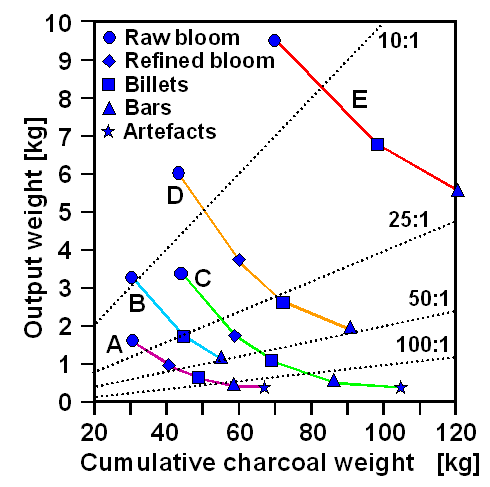 |
The relation of charcoal to iron after the various steps
needed to make an iron object.
Reference lines for selected charcoal to iron ratios are also given. |
- A: High P bog-ores, 8kg smelts in 25cm diameter furnace, without slag tapping.
- B: Bog-ores, with low P and moderate Mn, 10kg smelts in 25cm diameter furnace, without slag tapping.
- C: Sideritic ores, 16kg smelts in 30cm diameter furnace, with slag tapping.
- D: Sideritic ores, 16kg smelts in 30cm diameter furnace, with slag tapping, hot-bloom removal and immediate refining,
with re-heating in the furnace.
-
E: Bog-ores, with low P and high Mn, 32kg smelts in 40cm diameter furnace, blown with an air rate of about 1000 litres
per minute, with slag tapping, hot-bloom removal and immediate refining to a smithed bloom; cut to three pieces for refining to bars.
|
| Source Peter Crew |
|
|
 |
If we look at curve C, for example, we see that this experiment yielded a bloom
of about 3.4 kg for about 45 kg charcoal burned; a relation of 13 :1.
After refining the bloom, about 1.8 kg of iron
are left, decreasing to 1 kg after forging a billet, 500 g for the bar, and just about 300 g left in the "artifact",
a knife blade in this case.
Each heating and forging step looses iron because you cannot avoid losses by oxidation
and flaking off. More experienced smiths might be able to keep losses low but your are definitely loosing more than 50 %
of your original bloom weight to the necessary follow-up processes. |
|
 |
Even for curve E representing the most efficient smelting, about 120 kg of chacoal
were needed to produce a final product weighing 6 kg! |
| |
|
|
What Did We Learn? |
 |
From all the above some insights emerge:
- It's a lot of work to make an iron object. Producing a 10 kg bloom keeps about 10 - 15 people busy (including the charcoal
maker, the ore suppliers, smiths, and so on) for days, and you can't produce more than a handful of blooms a week. It follows:
- Iron objects were still special and not cheap before the "industrialization" of the process around 250 AD.
- Whatever you made, it wasn't wrought iron. You had a mix of carbon-rich and carbon-lean iron, often with substantial
amounts of phosphorous (and possibly some other stuff) in just one bloom. You might also find large variations in "steel
quality" from boom to bloom.
- But you also had experienced smiths who could judge the quality of a piece of iron / steel by a number
of ways (some probably lost) and work "good" pieces into rather satisfying products.
- Most iron products were made by haphazardly banging together whatever grade of iron / steel you had in your shop. That's
why we find knifes with hard cores and soft edges and many objects where the carbon and / or phosphorous concentration changes
unsystematically from here to there.
- However, some guys somewhere and sometime knew exactly what they did. In fact, smithing skills around 400 AD allowed
to produce wonderful pattern-welded
blades and were never surpassed. Some smiths even had a working knowledge of case hardening and used that conscientiously.
There must have been some frustration, however, for sometimes it worked,and sometimes it did not.
Great. But the headline was: Technology Transfer - Spreading the Technology".
I bet you forgot. What did we learn about technology transfer from looking a the five places enumerated above? Not all that
much, I do admit that. Nevertheless, I will dare to draw a few conclusions! |
|
 |
1. It did take quite some time for iron technology to get out
of Anatolia / Cyprus / Palestine (and possibly Iran). In most of Italy, Spain, and Egypt, plus pretty much all of Northern
Europe, iron did not come into frequent use before about 800 BC if not a few centuries later. In Greek there are signs that
iron was used around 1100 / 1000 BC but then "disappeare" for a few centuries, re-emerging again around 700 BC.
Anything you could call "technology transfer" can't take that long. So what happened? Well, for technology transfer
you need three ingredients: 1. A society that has the technology, 2. A society ready to receive the technology, and 3. Ways
/ people for transferring the technology. Sounds simple but the emphasize is on "A society
ready to receive the technology". A "society" simply demands a state of organization far more
complex than that needed for running a tribe or clan, and "ready" means just
that. Look a than example from today. Germany does contain a highly organized society that can make microelectronic products
or solar cells. I, personally, would be willing and capable to transfer the technology (for a small consideration, of course).
Well-defined societies in (pick about any country / society in Africa / Arabia) do exist and might be eager to receive the
technology. They are not ready, however.
I don't think it is an accident that "local"
iron ages are associated with the rise of large-scale societies like the Etruscans, Celts or Romans. |
|
 |
2. Emulating a smelting process that worked in Anatolia, or
in some other place like Elba or the Noricum, most likely wouldn't work all that well. As we have seen in all cases, everything
must be just right - for your local conditions. While you can learn about the basic process of smelting iron from listening
to what some traveller has to say, you must optimize the process for your local conditions by trial and error. That is a
tedious and costly process without any guarantee that you will make it. |
|
 |
3. Learning the art of forging from hearsay - veything from
working your bloom to actually making a knife blade - is like learning to knit a sweater with a complex pattern or to build
a Stradivari from hearsay. It takes years of learning and practicing under the supervision of a master before you can call
yourself a blacksmith. And an experienced blacksmith who had made it from Cyprus to England in 1000 BC, say, would not have
found it easy to practice his craft: no iron, no infrastructure for all the things he needs.
Transferring just knowledge,
even in the form of experienced people, is not good enough. You need to transfer a whole infrastructure including a certain
mind set of the people involved.
I was lying above. I could not transfer the technology
of making solar cells to the Congo, say. Why, the people there don't even need me. They just need to buy a few books that
contain all there is to know about making solar cells. You could buy those books and
start to make solar cell. I bet you wouldn't be sucessful. OK, out of the goodness of my heart, I send you some PhD students
who have deep hands-on experience in the business. You know what? You, together with these knowledgable foreigners, still
won't be making solar cells. I'm quite sure of that.
Technology transfer is just not all that simple in other words. |
 |
Assuming that some technology exists somewhere, technology transfer
only happens as soon as some area / society is ready and connected to the rest of the world. Then it will spread in a self-organized
way and possibly even progress rapidly beyond the level of the society where it first evolved. Witness the advent of metallurgy
in China and the Roman empire after these cultures were ready. Due to their then unmatched
organizational skills, they quickly turned smelting and metal working from isolated artisan procedures to industrial activities.
So far so good. The problem we face now is |
|
|
Who discovered steel technology
for the first time?
When and Where?
|
|
|
 |
With steel technology I mean:
- Realizing that there are different "grades of iron" called steel.
- Finding a way to sort these different steels from one or more blooms into at least three groups (too soft, too hard,
just right).
- Producing halfway uniform pieces from the members of one group by fire-welding.
- Producing knife or sword blades by fire-welding pieces of different steels with the soft / hard parts in the right places.
- Going through the proper processes for case hardening, including quenching from high temperatures and and possibly subsequent
tempering at medium temperatures.
|
 |
This is a tall order. It includes paraphernalia like recognizing
phosphorous steel and knowing that it can't be hardened by quenching, being able to do all the forging without too much
loss of material and carburizing / de-carburizing the outer layers, and being able to produce a longish object like a sword
blade in a way that makes its structure rather uniform throughout the length even so you can only work a small part of it
at a time. Nobody usually has a hearth more than a meter long, after all. |
|
 |
Note that finding some martensite in some old steel does prove that the smith
has thrown the red-hot object into cold water, indeed. It does not prove, however, that this smith knew a thing about steel
technology. He might have thrown all his finished products into cold water, just to safe time, not caring if he had wrought
iron, phosphorous steel (both not given to hardening upon quenching) or proper carbon steel that would produce some martensite. |
 |
Having halfway well-defined and different grades of carbon steel
is the key to steel technology. You might get that by mastering smelting to a degree where you could produced rather homogeneous
blooms with a certain carbon concentration or by picking various grades from a bloom with mixed compositions.
Let's
see if looking into the iron / steel trade will help us there. |
| | |
|
| 1) |
Susan Sherratt: "Commerce, iron and ideology: Metallurgical innovation in
12th-11th century Cyprus"; in: Karageorghis, Vassos, Cyprus in the 11th century B.C.: proceedings of the international
symposium organized by the Archaeological Research Unit of the University of Cyprus and The Anastasios G. Leventis Foundation,
Nicosia 30-31 October, 1993, 59-106, Athens: A.G. Leventis Foundation |
| 2) |
Radomir Pleiner: "Iron in archeology. The European bloomery smelter". Archeologicke
Ustav AVCR. Praha, p. 400 |
| 3) |
Gerhard Dobesch: "Zweierlei ferrum Noricum?", Mitteilungen des Montangeschichtlichen
Vereins Hüttenberg - Knappenberg, Folge 18, Nov. 2011, p. 1 |
| 4) |
H. Preßlinger, E. M. Ruprechtsberger and O. H. Urban: "Stahlwerkstoffe in der Kelten-
und Römerzeit – Teil 1 und 2"; BHM, 152. Jg. (2007), Heft 5, p. 146- 150, and BHM, 152. Jg. (2007), Heft
5, p. 232 - 234. |
| 5) |
Peter Crew: "Twenty-five years of bloomery experiments: perspectives and prospects",
in D Dungworth and R Doonan (eds) 2013, Accidental and Experimental Archaeometallurgy [25] (London: Historical Metallurgy
Society), p. 25-50 |
| 6) |
Andreas Schäfer: "Zwischen“ Dünsberg und Waldgirmes. Wirtschaftsarchäologische
Untersuchungen an der mittleren Lahn" in: Berichte der Kommission für Archäologische Landesforschung in Hessen,
Vol. 10 (2010) p. 69 – 90 and in Abstracts of Second International Conference Plas Tan y Bwlch 17th – 21st September
2007: Early Ironworking in Europe II, archaeology, technology and experiment. |
| | |
© H. Föll (Iron, Steel and Swords script)