|
9.1.2 Problems with Alloying |
|
Setting the Stage |
 |
In everyday life we use almost exclusively metal alloys and not pure metals because
alloys are better. All alloys with iron are called steel a long as iron is the base
metal with more than 50 wt% of the total. With copper (Cu) as base metal, we use more names and call its alloys bronze or brass,
for example. There are no generic names for aluminium
alloys, even so they are very important.
So alloying is good. Well, yes - but there are also problems. I shall
start reminiscing a bit about the history of alloying and go from there to point out some problems
with alloying. Finally I will delve deeply into the joys of alloying |
 |
In writing this article I found it surprisingly hard to find
data or good metallographic pictures for the ideal plain carbon steels, i.e. steel with only
carbon as alloying element. That's the kind of steel I have discussed in the preceding chapters. There is a simple reason for that:
|
| |
| |
| |
|
There was and is no such thing as
ideal plain carbon steel
|
|
| |
| |
| |
 |
There was and is no such thing as ideal plain carbon steel since nowadays we
always add some other elements (typically manganese and silicon) , and in the old times you always had some stuff (often
phosphorus) in the iron unintentionally. |
 |
Steel is iron with some alloying elements. That is a relatively recent
insight. Looking back three millennia, we might distinguish several steps on the way to understanding the steel alloy system
we are dealing with here: iron plus a bit of the rest of the periodic table. The times given below are approximate. They
may vary between here and there, and there is always considerable overlap.
- 1.700 BC (?) - 1.500 AD: Iron was "invented". Smelting proceeded by a solid state process. The big
trick for getting steel was to have it as part of the bloom in the first place. This necessitate the ability to recognize
and to separate it from the wrought ion probably also there. You never made steel, as stated in much of the literature,
by employing a second process called "carburization" that inserted some carbon into the bulk
of your wrought iron (or something else suitable
like phosphorous (P)). In any case, your iron or steel was full of inclusions (except for crucible steel). Where and when
steel making differentiated itself from (wrought) iron making by optimizing bloomery processes for either extreme is not
all that clear, but around 500 BC there was some intentionally made steel around. Of course, the steelmakers most likely
did not have the faintest (correct) idea why whatever they were doing actually "worked". If they had any ideas
at all, we don't know about them because these guys couldn't write, and nobody who could write would even dream of recording
anything that he heard from mere artisans / slaves.
But even without any clear understanding of what one was doing,
iron and steel technology became incredibly sophisticated during the 3000+ years we talk about here. Complex and beautiful
pattern-welded swords and equally enticing wootz swords were produced.
- 1.000 BC - 1.500 AD: Some people called philosophers thought about the "nature" of materials and wrote
it down. Most results of that thinking were utterly wrong. Small wonder. As long as you don't really know what atoms and
chemical elements are, you cannot possibly understand what is going on while making steel (or people). Some early guys had
a few thoughts into the right directions but the prevailing
view of guys like Aristotle was utter nonsense.
- 1400 - 1700: With the advent of the blast furnace steel making changed completely.
Surplus carbon and other stuff now needed to be taken out of the dirty cast-iron (called pig iron) that was produced from now on until today.
- 1700 - 1830:
It became clear that carbon is
responsible for the hardness of steel in comparison to pure iron, and that "chemistry" mattered in the sense that
it was important what your steel contained or came into contact with. It also became clear in the late 17th century what carbon is. Alloying with elements beside carbon happened too, but more
or less accidentally.
- 1830 - 1900: The major breakthroughs in steel making occurred, tied to names
like Austen, Bessemer, Thomas, Siemens, and Martin. Steel production increased dramatically and so did the understanding
of what is going on. In 1897 Robert Austen describes the first
iron - carbon phase diagram. Looking at the microstructure with microscopes and intentional alloying started.
- 1900 - 1950: "Discovery" of atoms, statistical
thermodynamics, and quantum theory. Based on this, the "discovery" of crystals, and crystal defects could follow.
The ingredients for a thorough understanding were now all there. Alloying was done consciously on a rapidly growing empirical
and scientific base.
- 1950 - Present: Modern analytical tools allow to find out the concentration of the elements in your steel, how
they interact and generate all kinds of microstructures, and how that relates to the properties observed. Alloying is based
on a deep understanding of what is going on and a huge data base. There are, however, still some open question and occasional surprises.
- 1990 to Present: Computer power started to allow "first principle" calculations about properties of
real crystals. The age of scientific steel (and other solids) has started. Its
impact on steel technology, while still minor, is nevertheless felt and is sure to increase. Semiconductor science and
technology is utterly impossible without computers.
- Present - 2050: Wait and see. There will be new and superior steels for sure.
|
 |
Whatever is in your steel might have been put in intentionally or unintentionally.
In a strict sense, putting specific elements intentionally into your steel did not start
before, roughly 1850. Steel makers through the millennia, however, might have used certain ingredients (like leaves of some
special plants in crucible steel making) for the same purpose.
What you had in your steel depended on what you used for making it, and trial and error lead to recipes that "worked"
- sometimes by alloying the right stuff by good luck. |
|
 |
Your forebears put (mostly) the right things into their personal systems, too. They did not
eat certain mushrooms, fruits and plants even so they had no idea about the biochemistry of toxins and had never heard of
cholesterol or calories. But they did not consume the really good stuff either (chocolate ice-cream, coffee, cognac, my
wife's gooseberry cake, .. ) because they lacked the ingredients and the know-how for making it. Try to make ice-cream without
a freezer. It can be done but I bet you don't know how.
Most of the time your ancestors ate mediocre food that was
easy to get. But they had very good food on occasion, using special and expensive ingredients and followed time-tried complex
recipes.
In analogy, very good steel was produced sometimes and then used for swords or other high-value items. Most
of the time, however, the stuff was mediocre or bad. |
| | |
|
| |
Major Problem: Embrittlement |
 |
If we now look at whatever was in there intentionally or unintentionally, it
either could be good or bad (and anything in between). Sulfur (S) is practically always bad, manganese (Mn) is practically
always good, and phosphorous (P) is ambivalent. Of course, as with all medicine or spices, the dosage is important. The
headline of this sub-chapter was "Low
Alloy Steels", meaning that we look at low concentrations below at most a few percent. |
|
 |
If a given steel gets worse upon alloying it with something depends on how you
define "worse". That depends on your intended uses. Does a whisky get worse when you alloy it with soda?
A decreasing hardness, for example, might be bad for sword users but not for others.
When you need to make a chastity belt in a hurry,
it's easier with not-so-hard steel. Since your wife doesn't appreciate a hard one any more that a soft one, why bother and use hard steel?
|
 |
Everybody agrees on one issue, however: |
| |
|
| |
|
| |
|
 |
If your steel undergoes a ductile-to-brittle
transition (or transformation), you have a problem! It is such a well known and big problem that it has its own
abbreviation: DBT! If alloying induces a DBT transition, your steel is now perfectly brittle and behaves essentially
like glass. Not good. Neither for swords nor for chastity belts.
Phosphorous and sulfur are notorious for causing DBT
behavior. They are also particular hard to keep out of (ancient) steel, so we now have one of the major problems with (typically
unintentional) alloying in ancient and recent times: |
| |
|
| |
|
| |
|
|
 |
The terms mean shortness, nowadays called brittleness, that occurs at
low or high temperatures, respectively. I've already
introduced this in chapter 3.2, now we will go to the core
of these really big problems in steel technology that haunted steel makers and users up to about 1950.
To be perfectly
clear: phosphorous (P) causes a DBT when the temperature drops below some critical temperature
TDBT, and sulfur causes a DBT if it gets above
TDBT. Other alloy elements might also cause these effects. |
 |
Cold and hot shortness were two of the banes of steel throughout the millennia.
Even today DBT causes major worries for users and makers. |
|
 |
Cold shortness is a major problem for you, the user.
Just imagine that your steel turns brittle in the winter, when the temperature is around freezing. Your sword will shatter
into thousand parts on impact, your steel steamer will just break apart (that
happened!; see below), and your chastity-belted wife only needs to sit in the snow for some time and then look for that
nice smith apprentice with his hammer. One little bang will do it (for starters).
In other words, cold shortness is
bad for you if it occurs in the range of temperatures where regular humans live, love, work, and do business. If cold shortness
occurs at temperatures too low for regular humans, you can live with it. However, if you, like me, belong to the irregular
humans who, for example, keep liquid nitrogen (N) or helium (He) in steel containers at temperatures around -200 oC
(-328 oF) or -270 oC (-454 oF), resp., you better watch out for what kind of steel is used!
|
| |
| |
| |
|
|
| |
| |
| |
|
Hot shortness doesn't affect you, the user, all that much - except if you plan to go to hell - because sword fights rarely take place around 500
oC (932 oF) or so. The smith has the problem now since his steel turns brittle when its hot. If red
shortness occurs, there will be no forged steel objects because the material shatters when the smith is trying to forge
it at high temperatures. So all embrittlements are simply bad. |
 |
I hear you. You now want me to answer your why
questions. Why are phosphorus and sulfur causing DBT transitions at some special and
inconvenient temperatures? And how? For starters I give you a first clear answer: |
| | |
|
| |
|
| |
| |
|
 |
Well, let's take that with a grain of salt. I don't really know
the deep-down details - and that's also true for my colleagues. In the words of Jianming Huang, who wrote a PhD thesis entitled: "Ductile-to-Brittle Transition in
Body Centered Cubic Metals..." in 2004: "The (..) mechanism of this (ductile to brittle)
transition still remains unclear despite of large efforts made in experimental and theoretical
investigation". |
|
 |
By the way, there are many more things in Materials Science that present-day Material
Scientists don't know about for sure. What a relief! This means that there is still enough work for Materials Scientists
and Engineers in the future. My kids can go for decent careers and do not need to become lawyers or bankers. (The brats
actually went for medical doctor (2) and literature science (1)). |
 |
OK - so I don't really know what exactly is
going on at DBT transitions. But I do now quite a bit of general stuff about DBT's, however, and I will share that with
you. |
|
 |
I do know, for instance, that hot shortness,
in very general terms, occurs if some impurity forms precipitates with a low melting point that like to sit in grain boundaries.
When it gets too hot they melt. The ferrite / pearlite grains of your steel then are held together in parts only by a liquid.
They will still cling together - even if the grain boundaries are completely liquid - just like wet hair. But hit it with
your hammer and the steel falls apart. Sulfur (S) does just that, so hot shortness is now explained (haha). |
|
 |
What happens at cold shortness is far more
trickier. Quote: "Large quantities of phosphorus (in excess of 0.12% ) reduce
the ductility thereby increasing the tendency of the steel to crack when cold worked. This .... is called cold-shortness".
You will find a statement like this in most steel books.
Notice that the quote above doesn't say that phosphorus causes cold shortness, only that it increases the tendency
for it. But why is phosphorous increasing the tendency for cold-shortness, or as we
call it now, a ductile-to-brittle transitions (DTB) at low temperatures?
And how does it do that? I have promised (or threatened?) to tackle these hard questions in this Hyperscript, so I will give it a shot.
| |
|
|
 |
In what follows here I give you a few simple
hard facts about cold shortness. Any attempts to delve deeper into that subject will open a humongous can of major and very
wriggly worms. That's why I will deal with the deeper aspects of cold shortness mostly in the science module. |
 |
When I started to look into the topic in more detail, I had no idea about the
size of the can of worms I was about to open. I spare you the gruesome details, you will probably find more than you are
looking for if you use the link above. The explanation for cold shortness, in very simplistic terms, follows a line of arguments
like this: |
|
|
- Any crystal under large stress will either deform plastically or fractures.
In other words, it is either ductile or brittle.
- Plastic deformation means that sufficiently many dislocations are generated and
moved across the crystal. This needs some minimal energy, some of which is supplied
by the stress and some by the temperature. If you lower the temperature the dislocation
process gets more sluggish.
If dislocations hit an obstacle like hard cementite, they might be stopped or slowed down.
Grain boundaries also slow them down, irrespective of some dirt in there. The minimal stress needed to promote dislocation
movement thus depends on the microstructure and the temperature in a rather tricky way.
. - Fracturing
means that some tiny pre-existing nano
cracks grow. This implies that the crack tip runs through the specimen and induces fracture. In order for this to happen,
a certain minimal stress is needed.. If the crack tip hits an obstacle like hard cementite,
it might be stopped or slowed down. If the crack tip hits a grain boundary with weak spots because some impurity atoms or
small precipitates are sitting there, it might speed up. The minimal stress needed to promote cracking thus depends on the
microstructure in a rather tricky way but not much on temperature.
- If you apply serious stress to your specimen, there will always be a competition
between the two mechanisms. If cracking wins, your specimen is brittle, if plastic deformation by dislocation wins, your
specimen is ductile. In most fcc crystals like aluminum (Al), gold
(Au) or nickel (Ni), the dislocations always win at pretty much all temperatures. But this is not necessarily true for other
crystals and you will find a DBT transition at some temperature.
- Since both mechanisms depend in different ways on the microstructure and temperature, phosphorus and
any other element in your steel cannot but influence the DBT transition behavior.
- In fact, even pure iron or ideal plain carbon steel, or just about all
bcc crystals, show DBT transitions - mostly at critical temperatures
TDBT substantially below freezing, fortunately. Before the advent of refrigerators that simply
wasn't much noticed. Vikings may have lived in areas where the temperature falls substantially below freezing. But they
didn't do major sword fights then, or knew and accepted that their swords broke as a natural thing. They might even have
had two swords, one for cold, and one for not-so-cold weather. So they only were pissed at the smith (and shortly after
probably dead), when their swords broke while fighting in decent weather.
|
|
 |
So the long and short of cold shortness in (ferritic) steel is
not if it occurs (it will), but at what critical
temperature
TDBT
you will encounter it. With respect to alloying we therefore want to know how the alloy element affects the critical temperature
TDBT and possibly the magnitude of the effect. The figure below makes clear what I mean:
|
| |
|
| |
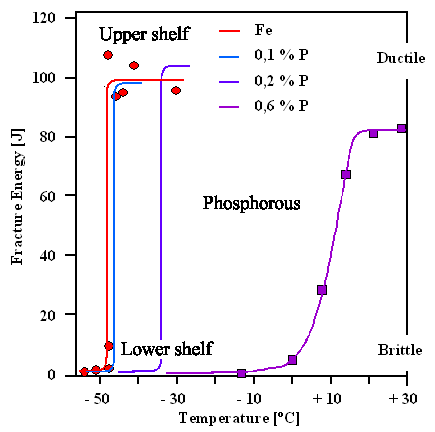 |
Brittle to ductile transition with various phosphorus (P) concentrations
A few of the original measurement points are shown to give an idea about the experimental scatter encountered |
|
Adopted from: W.A. Spitzig, "The Effects of Phosphorus on the Mechanical Properties of Low-Carbon Iron",
METALLURGICAL TRANSACTIONS, Vol. 3, MAY 1972, p 1183 |
|
| |
| |
| |
|
A Charpy impact test
provided the fracture energy data on the left scale.
The specimen for the red line, shows
essentially pure iron. Its detailed composition was: 0.005 % C; 0.001 %N; 0.0045 %O; <0.005 %P; 0.004 %Si; 0.01 %Mn;
0.004 %Si; 0.002 %Ni; 0.002 % Cr.
"Upper shelf" and "lower shelf" is steel engineering slang for the high and low fracture energy associated with
ductile or brittle behavior, respectively. Their difference is a measure for the magnitude of the effect. |
 |
Those are rather dramatic curves that tell us a lot about the DBT transition phenomena.
Let me enumerate the major points for you:
- The critical temperature TDBT increases from about –50
oC (–58 oF) for pure iron to about 10 oC (50 oF) for iron containing 0.6
% phosphorus (P). This kind of phosphorus steel thus is only good at temperatures around
and above room temperature.
- The effect of cold shortness is nothing less than dramatic. The energy needed to
fracture the steel in a Charpy impact test comes down from
an "upper shelf" value around 100 J to almost zero at the "lower shelf".
- The results you get in an experiment as illustrated in the figure will, of course, not only depend on the nominal phosphorus
concentration but also on details of the microstructure and on exactly how you tested. You could, for example, have different
average grain sizes for the same phosphorus concentration, and then you would get somewhat different data.
- The DBT transition region can be extremely sharp. Take the phosphorus steel with
0,2 % phosphorus, for example. At –30 o (–22 oF), it is not brittle but ductile. Hit it
hard with a hammer (or another sword), and it will just bend or dimple somewhat. Go to –40 o (–40
oF), and just hitting it lightly will cause it to shatter into pieces. For higher phosphorus concentrations,
the transition is not quite as sharp but still occurs within a narrow temperature interval.
|
|
 |
Now you see why "the (..) mechanism of this
(ductile to brittle) transition still remains unclear...". We can account more or less for the points 1 and
2 right above, but point 4 is a tough nut to crack with theory. There are other open questions concerning DBT transitions;
look up the science module if you need to know. |
 |
Alloying iron with anything, or alloying iron already containing
a mix of alloying elements (then called steel) with one more element, will always change
the DBT transition behavior. The changes might be small but you must be aware of it. |
|
 |
Generalizing a bit, alloying will always
change all the properties of your steel. Playing around with one specific
element might leave some property almost unchanged, make it better, or make worse. The "playing around" part,
meaning the concentration and detailed processing you use, is important.
If you only look at the information about
phosphorus (P) as alloying element that is contained in the figure above, your conclusion would be to avoid phosphorus at
all costs. To some extent that is what we do. But before you throw out phosphorous (P) forever (assuming you can do that),
you should first look at what it can do for you.
If you cannot throw out phosphorous because you, the ancient smith,
do not know how to do that, you better figure out how to live with it.
We have few modern
steels that contain phosphorous (P) intentionally because whatever good it can do for you is mostly far easier done with
other elements. But we do have plenty of ancient steels that contained phosphorous (P)
unintentionally. Despite all the problems it causes, it still could be used to makes steels that were better than wrought
iron or bronze. In fact, phosphorous (P) steel was so important in old times that it has its own sub-chapter, coming up
soon. |
 |
The basic problem with alloying
should be quite clear now: |
|
| |
| |
Making a good alloy is an extremely
complex undertaking.
|
|
| |
|
|
 |
If you intend to "invent" a better metal alloy today, you are well advised to assemble
a highly knowledgable group of 5 - 10 people, supply a infrastructure with the necessary hardware like furnaces and electron
microscopes, and give them at least 10 years and a sizeable budget.
The odds are actually rather low that they will
come up with a new alloy that will be successful in the market place. Nevertheless - it's the only way to go. |
| |
|
© H. Föll (Iron, Steel and Swords script)

