|
10.2.3 Smelting Wrought Iron, Steel and Cast Iron |
| |
Steel
by Carburization? |
 |
Let's start by a quick look at a controversial issue: Can one make steel from
wrought iron by "carburization"? The answer is certainly "yes" - as long as the term isn't specified.
It is certainly possible to get carbon from the outside into originally clean iron ("wrought iron") if you have
high temperatures at your disposition - and all the time in the world. Steel has indeed been made in this way in the 18th
century (by a process called "cementation"); I will get to that.
|
|
 |
However, the issue here is not the 18th century but the first millennium BC and
the many publications stating that some old artifacts consist of "carburized iron", vulgo steel. The directly
or indirectly implied message is that some ancient smith started with wrought iron and managed somehow to get substantial amounts of carbon into
an object like a knife blade by carburization while he is forging it.
Here are a
few examples:
- "Ideally the smelter produces steel. If not, the iron will be carburized in the forge".
- "The practice of steeling (i.e. carburizing) such items as bracelets is curious since there is no functional advantage
to be gained...."
- "One of the knifes was not carburized, one was mildly carburized, the other two were extensively carburized".
The list could go on for quite a while. I do not give references because what we have is a general pattern, and because
authors I regard very highly have fallen into the fallacy of implying that ancient smiths somehow "carburized"
wrought iron in order to get steel. The truth, however is: |
| | |
|
|
|
You cannot "carburize" a volume of iron
in your forging hearth.
|
|
| |
| |
 |
Rehder, in his invaluable book, makes that quite clear. He has two independent arguments
for his claim:
- The first argument is based on the necessity that the source of carbon can only
be the carbon monoxide produced by burning charcoals in the forging hearth. Based on simple calculations he argues that
one cannot not have enough of that gas in all the places were it would be needed. One rather has carbon dioxide and oxygen,
and the usual effect of heating a piece of iron in a forge-fire is actually de-carburization and iron oxide ("hammer
scale") formation.
- The second argument considers the time needed for getting carbon deep into iron. Rehder maintains that even with enough
carbon monoxide, "carburization" would take far too long to be of practical value.
|
|
 |
The eminent archeometallurgist Donald B. Wagner,
undisputed expert for the Chinese metal history, begs to disagree1). He bases his criticism on Rehder's CO concentration calculations, reasoning that
enough of the stuff could be around in a forging hearth. He also refers to writings from the 18th century and to some more
recent experiments were "carburization" during forging was claimed or demonstrated, respectively. What Wagner
proves beyond doubt is that one can carburize wrought iron, indeed - as long as only thin layers or tips of needles are considered.
|
 |
The issue is rather easy to resolve. How much carbon is available
for moving into the iron from the outside does depend on the source of carbon, of course. Carbon monoxide, splitting into
carbon and oxygen on the surface of hot iron, is far more efficient in providing carbon atoms at the iron surface than solid
charcoals. A gas can envelop the iron at any moment, whereas a solid piece of charcoal only touches the iron at few spots
at best (see all the way below), and the carbon - iron contacts are even destroyed as soon as a carbon atom breaks off and
disappears into the iron.
This is clear from the picture below. It is our old
copper picture, of course, just slightly changed for smelting iron now. It has one added feature: carburization by CO.
It still does not show slag formation and other "details".
So the source of carbon for "carburization"
is important, indeed - but not crucial for the topic. |
| | |
|
|
|
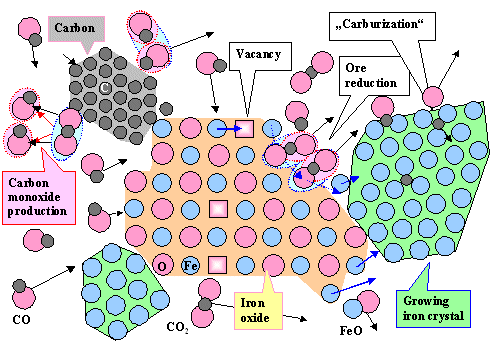 |
| Inside an iron smelter. Extremely simplified |
|
| |
| |
| |
 |
The real problem comes from diffusion,
from the way carbon atoms move into the inside of iron. Once a carbon atom has made it below the surface layer of the iron
crystal, it moves around randomly, as illustrated
here. Looking at
numbers one learns, for example, that you need to keep your piece of iron at 1000 oC (1832 oF)
for about 3 hours if the carbon should penetrate to a depth of about 3 mm. That would
just about "carburize" a knife blade. Not uniformly though, mind you. The carbon concentration near to the iron
surface would be much higher than in the middle of the blade.
So you can get carbon inside iron. In principle as deep
as you like, but at "normal" temperatures, this takes far too long to be practical. If you want to go for 6 mm
in the example above, it will take 9 hours; 9 mm take 27 hours and so on. If you increase the temperature a lot (not all
that easy to do in an ancient forge), the time required does come down but grains will grow larger and so on, counteracting
your efforts to "harden" the iron with carbon.
What you can do and what has been done, is to "case-harden" your workpiece. You supply a thin layer of
carbon-rich iron or steel around your wrought iron inside by keeping it in your forge fire at the right place (where the
carbon monoxide is produced) for a while. If you quench and possibly even temper your piece after that, the outside or "case"
could well be very hard steel.
However! While this is not too difficult to achieve in a modern hearth with a constant
air supply, ancient hearths were "blown" with an intermittent stream of air, and this caused the reducing part
of the fire to "breath" - moving back and forth. The gradual disappearance of the coals also moves the proper
place around, and it is not all that easy to move your workpiece accordingly. If your workpiece is large like a sword blade,
you can only keep a small part of it in the right place anyway.
And don't forget: whenever your iron is not in the
reducing part of your fire, de-carburization takes place! First, you oxidize the iron,
consuming the surface-near and "carburized" parts, and second, the carbon diffuses out
of the iron just as readily as into the iron. If your pieces carburizes or de-carburizes
simply depends on the balance of depositing carbon atoms on the surface and sweeping them off. Only in a reducing CO-rich
atmosphere deposition wins.
All in all, carburization of a thin layer of iron in
an ancient forge fire is possible - but not easy. Carburizing a solid large piece of
iron this way is impossible, though. |
 |
The long and short of this is simple: |
| |
| |
| |
Ancient fully "carburized" iron (=steel)
was never made in a forge fire
but right in the smelter!
|
|
| |
| |
|
Smelting Wrought Iron |
 |
Iron smelting throughout the millennia supposedly produced wrought iron with a
very small carbon concentration. Maybe that's true. At this stage I'm not saying it is, and I'm not saying it is not. What
I am saying first is: Why bother? Wrought iron's properties as a metal are supposed to be inferior or at best equal to those
of bronze. We know that this is true for the hardness of the material from comparing it with bronze.
However, wrought iron (and mild steel) offers large advantages
on other counts:
- If you have the know-how, it is far easier to make than bronze. It doesn't need
an extensive infrastructure for procuring rare ores or ingredients like tin from far away. In contrast, if you have some
wood at your disposal and access to some iron ore deposits in mountains or to a working or ancient bog in the low lands
where you find bog iron, you can make it all yourself.
- Iron is therefore much cheaper than bronze.
- Iron is easy to work with by forging. You must have the know-how, of course, but
banging a lump of wrought iron into a knife-blade shape is altogether far easier then making a mould, making a crucible,
melting some bronze, casting the bronze, finishing the cast.
- You can hammer or fire weld small pieces to
get a large piece; the link gives an
example. That is not possible with bronze and most other metals
- Your wrought iron tools can be repaired. If your knife breaks, fire-weld it together again. You can't do that with a
bronze blade.
- If you can "do" wrought iron, you have the door wide open for moving up to making and working steel,
including the "in-between" like case hardening your wrought iron knife blade.
|
|
 |
Now we know better. While wrought iron is
not harder than bronze, it does offer lots of advantages. As soon as people learned the many tricks needed to make and work
iron, there was no good reason anymore to stick to bronze except for some special cases or because you are conservative.
Getting into the new technology was torturous and painful, with many disappointments and unexplained mishaps. One only embarks
on such an enterprise if there is no choice.
People only switch to unaccustomed food shortly before starving. When
was the last time that you ate insects, worms, grubs, or just cats and dogs (or pigs, cheese, ...)? Potatoes weren't originally
welcome in Europe, many Asian people wouldn't know what to do with wheat, and I still don't know why my wife thinks that
corn is food for people before it has been processed by a pig.
It is thus no accident that iron came into its own at
the collapse of the Bronze Age. As we have seen in the preceding sub-chapters, the occasional very early iron came in all
kinds of variants - copper-rich, "carburized" - with no recognizable relation between the composition and the
intended use, see above. Some bracelets were "steeled"
while some knifes were not. It all points to those ancient smiths simply not having a clear idea about the various qualities
of iron / steel. They just did their best with the haphazard pieces they had and in all likelihood were quite happy if they
could make anything worthwhile for keeps. We can be rather sure that most products of early iron technology didn't "work"
and were simply thrown out.
Eventually, however, iron could be made reproducibly and reliably, and the secondary technologies
like dealing with the red-hot bloom, making half-way clean iron bars on a hearth by forging, plus making various shapes
from these bars by forging and fire-welding, became known and were mastered. |
 |
Above I have exorcised the "carburization demon". Now I need to remove
the "wrought iron was first" demon. Did you realize that all the carburization talk only
makes sense if one assumes that early iron workers could only make wrought iron? Only
then do you need to "carburize" the carbon-free stuff if you want steel.
That's indeed what is mostly assumed.
And that assumption was backed by theory
as far as it goes. Nevertheless - it is wrong! |
|
 |
Copper smelting technology at 1200 BC was developed to a point where it would
have been no problem at all to produce anything ironwise: wrought iron, steels with little or a lot of carbon, and cast
iron. All you needed to do was to use the existing technology with iron ore instead of copper ore and a somewhat increased
charcoal to ore ratio. A few minor adjustments here and there and off you go, making cast iron, for example.
True,
we have not found cast iron artifacts in the Mediterranean between 1200 BC and 1000
AD. Does that mean that nobody could make cast iron? No! It just as well could mean that nobody bothered to make it since
it was considered useless. But what about accidentially made cast iron in antiquity? Have we found any? Not really. But
then nobody looked for it and if found, it wasn't recognized.
Not everything one can make is actually made. Nobody in
the the South American empires (Maya, Aztec, Incas and so on) bothered to make wheels for carts and so on, even so they
knew how to do this (they made children toys with wheels). Wheels, considered to be one of the greater inventions of mankind,
are rather useless in ancient South-American cultures since there were no horses, oxen or any other animals that could pull
your cart around, not to mention the rather steep roads in the mountainous regions or narrow and muddy paths in the jungle.
It is similar for cast iron and high-carbon steel. As all modern smelting
experiments prove, you had no problem producing highly "carburized" iron in your smelter. You just didn't
want to do that most of the time. Steel is so much more difficult to work with and rusts like crazy in comparison to wrought
iron. Its larger hardness isn't so important for most of your everyday iron objects either. It might even be a nuisance.
For example, carpenters still use nails that are not made from steel but from rather soft iron for obvious reasons. And
cast iron is extremely brittle and thus useless on first sight. On second sight one might do something with it, as the Chinese proved.
We need to realize that the main product of the town
smith for the last 3000 years weren't swords, mail and armor. That was made by a few specialized guys. The vast majority
of smiths made every-day wrought iron or mild steel things for the always overbearing reason: It is good enough for the
job and much cheaper. Your car body isn't made from stainless steel for exactly the same reason. |
 |
In other words: After the iron age proper had started and enough experience with
the new metal was acquired, the smelters produced the quality of iron that the customers wanted. And that was mostly wrought
iron or mild steel, occasionally steel, and never cast iron. It is thus small wonder that smelting technologies were optimized
- by trial and error - towards making wrought iron and mild steel.
The question now is: |
| |
| |
| |
How do I smelt wrought iron only?
|
|
| |
| |
|
 |
The answer is: I don't know. It is easily possible
in theory but seems to happen rarely in all those experiments people
make! The best one can do is:
Take advantage of the iron-carbon phase diagram. And the peculiarities of the diffusion
of carbon in iron. Also don't forget to optimize the movement of the burden down you smelter in relation to the movement
of liquids like slag. This involves optimizing the smelter geometry and the kind and quantity of flux you add. Not to forget
either: having the right ore lump size distribution is essential. And it goes without saying that how much air you blow
in at what kind of velocity is crucial.
In other words: those old smelting masters needed an advanced degree in Materials
Science and Engineering. Or did they? |
 |
When my wife makes her famous "Hefekranz" (braided yeast dough thing,
sort of like halla, just better), she optimizes the relation of yeast to flour, the time at the right temperature for allowing
the yeast to eat the flour and multiply, the mixing of the ingredients, the nucleation and growth of CO2
-filled bubbles (this takes optimizing the reaction temperature and time to allow proper diffusion and Oswald ripening)
and the addition of secret ingredients like sugar and God knows what. I gather that the right kind of flour (particle size?) is very important, and that various
temperatures play a significant role. It is an incredibly complicated process and I have never managed to calculate what's
going on.
As far as my wife is concerned, she bakes a cake, following the working recipe
of my mother to a dot. Nothing to it. Works all the time. |
| |
| |
| |
 |
| It worked (Hefekranz) |
|
| |
| |
 |
Long ago I suggested to try a few changes to the recipe. Use different flour for
a chance - maybe the product gets better? My wife just gave me that look. Of course she was right. The probability that
an already very good product gets worse if you make changes in the recipe are far larger than that it gets better. |
|
 |
Good cooking recipes were obtained by a long series of trial and error, and it
doesn't matter if you cook food or iron ore. Eventually there are working recipes. They are somewhat different from region
to region but they work for the local customers. Maybe the Italian food is better than the German food; in Germany we use
German recipes anyway. But even in Germany, recipes for "hefekranz" vary quite a bit from here to there. So all
the good wives, about to make a hefekranz for their deserving husbands, use different recipes - but only up to a point.
All hefekranz recipes must have a lot in common after all, if the final product is to be contained in the bandwidth of what
constitutes a hefekranz. Leave that common ground and you do not get a hefekranz but a Gugelhupf, for example. Also very
good to eat but just not a hefekranz.
Maybe the Roman "Ferrum Noricum" (made from siderite)
is better than the North German iron (made from bog iron)
but smelting recipes in North Germany were not changed. Using the Roman technique would not have worked in North Germany
because the guys there just didn't have the exact same ingredients.
We can be rather sure that every region had their
wrought iron smelting recipe that had evolved by trial and error to give satisfactory results for whatever local conditions
prevailed. Satisfactory results, not the very best result, mind you. Evolution only goes that far and always leaves a bit
of room for future improvements. Just look in the mirror to recognize that. Evolution, after all, has made you. |
 |
What is the common ground in making wrought iron that all smelting recipes must
recognize? Looking at that from the product, your recipe must assure that
- Pure iron is produced during the primary reduction processes.
- This pure iron should not get carburized (or messed up in other ways) on its way down from the reduction zone to the
bloom.
|
|
 |
The first condition is easily turned into a recipe: Keep the temperature low (around
800oC (1472 oF). The you produce your iron only as ferrite that
cannot dissolve carbon. If there is some carbon for some reason, it will precipitate as cementite (Fe3C).
Since reduction of iron ore with carbon monoxide (CO) starts well below
this temperature, there is no big problem there (except, of course, setting and keeping the temperature). In the freshly
reduced ferrite there is no carbon. If it does not get hotter and turns to austenite there never will be. |
|
 |
The problem starts with getting your carbon-free ferrite into some protected position below the tuyere. Typically you need liquid slag for this. But there is no liquid slag at 800 oC. Nothing
helps, you must have temperatures well above 1100 oC (1472 oF) in the hot part of your smelter to
liquefy slag. You must get your carbon-free iron somehow through that hot zone without
carburizing and oxidizing it too much.
If you want to make steel or cast iron, the opposite is true. |
 |
This is where the art of smelting wrought iron starts. It turns
into a high art as soon as you don't just want to produce wrought iron but any kind of iron / steel, e.g. steel with 0.5 % carbon with some predictability and reproducibility.
If you can do that also with high efficiency, meaning with as little fuel and ore as
possible, you are worth your weight in gold, I guess.
Let's review what we have
in our bag of tricks for moving our carbon-free iron particle down through the hot CO producing and oxidizing zones and to keep them carbon-free if we want wrought iron. I include tricks that might come to mind
but don't work: |
|
 |
1. Envelope your pure iron in liquid slag at temperatures below 720 oC
(1330 oF).
Nice try but can't be done. There is no liquid slag at temperatures that low. Never ever. |
|
 |
2. Allow some carburization in the reduction zone and remove the carbon
later again.
Carburization, as we learned above, proceeds by diffusion and thus may only affect the outside of our
ferrite particles as long as they are not too small. "Burn" off that carburized layer in the oxidation zone by
oxidizing and what is left is carbon-poor. That requires that your particles are not to small so only an outer layer becomes
carburized, but not so large that they are not fully reduced. |
|
 |
3. Just run the process messily (and inefficiently).
Keep temperatures
as low as possible and inhomogeneous. Make only the parts above the tuyere as hot as needs be, keeping the opposite parts
cooler. The CO produced in the hotter part will spread out as it moves up, reducing ore also in regions closer to the other
side. Same thing for the slag. It is liquefied in the hotter parts above the tuyere but in percolating down will spread
to the outer parts, sweeping down whatever it finds. Iron particles moving down on the opposite side thus will not spend
all that much time in the austenite phase, where the can absorb carbon, and thus stay pure. |
|
 |
4. Don't mix ore and charcoal, produce layers.
When a charcoal rich
layer burns, producing lots of hot CO, you reduce the ore above it. When the ore layer with the iron comes lower, temperature
will go down because the charcoal content is low, and you get your iron down without carburizing it too much. |
|
 |
5. Oxidize your (more or less carburized) iron somewhat or even a lot in
the oxidation zone. As long as enough iron is there to make it stick to the bloom developing in the "cold" region
below the tuyere, you need not worry as long as you are producing carbon monoxide around the bloom, too.
In other words:
you now reduce very finely dispersed iron oxide right on the bloom. You might even reduce the iron oxides in slag that lingers
there long enough. Since this happens in close contact with some "seed" bloom, you are now growing
a wrought iron bloom. |
| |
 |
6. Keep your iron particles large and compact (in contrast
to spongy), or more generally, have a small surface to volume ratio. Carbon must diffuse in from the surface and you get
lower over-all carbon concentrations in large particles compared to smaller ones at the same conditions. How you can achieve
that I don't know. Use the right ores, in not too small lumps? Have the iron particles attached to yet unreacted ore / gangue
(protecting their surface)? |
|
|
7. Keep the carbon monoxide busy to reduce FeO wüstite
so it does not diffuse into the iron already produced out of sheer boredom because there is nothing else to do. In other
words: move your burden down fast enough so when it hits the hot zone some ore is still around. Expressed in scientific
words and with equations, that is the official
reason for the (wrong but time-honored) theorem that bloomeries can only produce
wrought iron. It will work, though. Just not all the time and in all circumstances. |
|
 |
8. Your idea. |
|
 |
9. Measures that really work and neither I nor you know about. |
|
|
|
 |
10. Measures that seem to work and that W.
H. Lee Sauder and Henry G. Williams III stumbled
across by doing lots of bloomery experiments under the influence of beer. My interpretation of what they are doing - and
I might be wrong - is: Run your smelter hot and with as much ore as possible. Reduce all iron oxide before it hits the oxidation
zone. The iron will re-oxidize but the oxide shell forming around an iron particle will form slag with the other now very
hot stuff around, envelop the remaining iron in a liquid slag shell, and trickle down fast to join the bloom below. |
|
 |
I'm not really certain about all the stuff above. Points 2 - 6 might contribute
to making wrought iron (or any iron / steel) to some extent but I don't know. Sauder and Williams go far in finding solutions;
time will tell how far exactly their approach (and my interpretation) will get us.
Personally, I still wonder if one
can produce wrought iron, meaning iron with hardly any carbon inside, in a smelter at
all. A bit further down I will make it clear that making solid iron with a lot of carbon
inside is also quite hard.
From "theory" one should expect that a bloomery produces iron with some
carbon - not a lot but more than almost nothing!
I'm certain, however, about one thing |
| |
|
| |
|
| |
|
|
 |
At temperatures above A1
the iron assumes the austenite phase. Carbon is soluble in austenite and can get "in" by diffusion. The distance carbon atoms can cover at a given temperature during a given time I called "diffusion length", invoking Einstein. This
diffusion length increases exponentially (really fast) with the temperature but only
with the square root of the time (slowly). If you increase the (absolute) temperature
just 10 %, for example from 1300 K (1027 oC;1880 oF) to 1430 K (1157 oC; 2114oF),
the diffusion length increases by 65 %, while even doubling the time (100 % increase) produces only about a 40 % increase.
Let's simplify that in a strong statement: |
| |
|
| |
Wrought iron production depends on
diffusion limitation
|
|
| |
|
|
 |
In other words: you can't keep your iron clean by limiting the supply of carbon. There is always enough around to mess up your wrought iron. You must limit
the diffusion. You must either make diffusion in the iron slow or you must make it hard for the carbon to get into the iron
by coating it with a diffusion barrier. Low temperatures in the reduction zone, by
necessity above A1 but as low as possible, were thought to provide the key to that. But if you keep the
temperatures too low, not much will happen.
A completely different approach - the
Sauder / Williams way - is to use high temperatures. It looks like this enables efficient slag formation and then efficient
protection of the iron particles.
That is quite similar to what we do in microelectronics. We must keep bad atoms like
copper or iron out of the silicon at all costs. Low temperatures would be fine but then you can't process your silicon either.
So use a diffusion barrier, a thin coating of something inside which the bad atoms diffuse so slowly that they will not
make it to the silicon or are "eaten up" because they react with the stuff |
 |
All things considered, I'm not too sure about what exactly you need
to do to produce only wrought iron. I'm not sure either if that was ever done. The complexity
of the problem will become a bit clearer when we now look at making steel and cast iron. |
|
|
|
Smelting
Cast Iron |
 |
Now let's go to the other extreme. Let's put lots
of carbon into the iron, making it cast iron.
In principle things are clear. Just do the opposite of what is described
above. Allow the iron particles to get really hot in the CO rich part of the smelter. Then carbon can penetrate deeply and
in quantity. To get a feeling of what the task implies, let's look a bit more closely at how much
carbon you actually need, and where it must come from. |
|
 |
The answer to the last question is simplicity itself. The only
source of carbon in your smelter is the charcoal. It turns into the carbon monoxide (CO) that acts as carbon source, indeed,
but you first need the charcoal. Charcoal has a density of 180 kg/m3
- 280 kg/m3; it is very "light". Iron, on the other hand, weighs
in around 7.800 kg/m3 - it is called a heavy metal for good reasons. Considering that you don't feed only
solid charcoal to your smelter but a charcoal - air mixture, which charcoal accounting for about 50 % of the volume, the
available carbon comes with an effective density of roughly 100 kg/m3.
You can imagine the production of carburized iron by taking a quantity of pure iron and then pile as much charcoal on top
as is needed for getting the right carburization level. Dissolve that pile in the iron, and there you are. Here is the picture: |
| | |
|
|
|
 |
How much charcoal you need to dissolve in the
iron shown to get the percentage indicated |
|
| |
| |
|
 |
It sure looks like you need quite a bit. In smelter reality, however, you need far
more - multiply by two or three, would be my guess. Why is that? Because you can't put all the carbon from the
charcoal into you iron! You need to burn quite a lot to produce the energy needed. And burning turns the carbon of the charcoals
into carbon dioxide (CO2). In the reducing layer this CO2 is reacted to the carbon monoxide (CO) that
reduces the ore. That produces carbon dioxide once more. Slag production might also eat up some carbon. Whatever happens,
a lot of carbon dioxide (and possibly some monoxide) will come out on top of your smelter, and the carbon in these gases
cannot carburize your iron. |
 |
If we now want to make cast iron with 4 wt% carbon in the iron, we encounter a
new problem: |
| |
| |
| |
Cast iron production is
carbon supply limited!
|
|
| |
| |
|
 |
It is not diffusion limited as the production of wrought
iron just discussed above. Why isn't it diffusion limited, too? Because you now run your smelter at very high temperatures.
The copper people knew how to do this; it is pretty obvious anyway: Increase the ratio of charcoal to ore and blow more
air trough the tuyere. In case of doubt use two tuyeres. What you need to achieve is that the iron formed way up in the
stack incorporates enough carbon in the CO-rich reduction zone to lower its melting point to a value were it becomes liquid
before it hits the oxidation zone. Then it can trickle down as cast iron and collect as a liquid in the bottom of the smelter,
protected by the slag layer that swims on top of it. |
 |
As already discussed above, your only chance
to get enough carbon into the iron is to use carbon monoxide as source of carbon. From the picture above it becomes clear
that you need to turn a lot of charcoal carbon into carbon monoxide to have enough.
And don't forget, you need carbon monoxide also to reduce the iron ore. For doing that you need far more carbon dioxide
and energy than in the case of making wrought iron. That's why you need to burn far more charcoal.
Hence the simple
recipe for making cast iron once more:
- Use more charcoal relative to the ore content, i.e. increase the charcoal to ore ratio.
- Make it as hot as you can inside your smelter. You need the high temperatures for more efficient CO production and for
fast diffusion.
|
|
 |
Isn't that interesting? Putting a lot of carbon into iron is just about as difficult as keeping
it out! Maybe making wrought iron isn't that tricky, after all? If you don't know a thing about iron and carbon and just
run your smelter on the "lean" side, you will get some carbon into the iron but maybe not all that much? Maybe,
but Lee Sauder, based on experiments,
disagrees. He rather goes for running your smelter hot and being smart about slag production. Who knows. |
 |
What we do know is that the early iron smelters around 1200 BC and some time after
that produced all kinds of iron and steel. Wrought iron, mild steel, hard steel, cast iron, phosphorous iron, and so on.
The bloom from just one run might have contained a whole mix, and the results from one run to the next must have varied.
That is what modern smelters
tend to find
Forging the stuff must have been frustrating.
Sometimes it was easy, sometimes it was difficult and sometimes it couldn't be done at all. That explains up to a point
why sometimes jewelry was made from hard steel and a knife blade from wrought iron, and why there seems to be no system
in the amount of "carburization" if we look at early iron products. It is one thing to discover the basic technique
of smelting iron, and quite another thing to make it a reproducible process that produced high-grade blooms with consistent
qualities and carbon content. It also takes some time to establish (and pass on) the art of forging with all its many tricks.
|
|
 |
It is usually assumed that the "West", including the Romans, was not able to produce
cas iron in antiquity - in contrast to the "East," i.e. the Chinese.
That is not the case. It looks rather that people didn't want to make cast iron. Old cast iron has meanwhile been found
in Europe, and there is probably more. One problem is that you only find cast iron or residues of cast iron smelting you
look for it (and know how to interpret what you find. I give you two references for that 2),
3) but for the time being leave it at that and now turn to the art of smelting steel. |
| |
| |
| Smelting Steel
|
 |
Making iron with carbon contents between 0.2 % and 0.8 %, say, is now simplicity
in itself (haha): Don't do anything special. Not too cool, not too hot and so on, and you will end up with a bloom that
contains some average carbon content around, say, 0.4 %. If you look closely you find
that the carbon concentration varies from one spot to the next. It is easy to see why. Every iron particle, deposited on
the existing bloom by some slag droplet, has its own history and acquired more or less carbon. If the lumps of ore fed into
the smelter had a large distribution of sizes, differences will be large, if they all had about the same size, their carbon
contents will be more similar.
For some hands-on experiecne on this read Adrian
Wrona's "hands-on" account. Here
it is. |
|
 |
There might also be differences in the average carbon contents from the bottom part of the
bloom to the top part because conditions changed. When you first feed ore into the smelter, you cool it to some extent and
it may take some time before a stationary state is reached. When you stop operations, your smelter is still hot and things
still happen for a while. |
 |
With increasing experience you learn how to make a bloom with some halfway predictable
carbon content. Surprises are likely to to happen whenever one of your parameters changes a bit without you noticing. The
quality of the charcoal from that new supplier is a bit different, your new tuyere has a slightly smaller inner diameter,
your bellows developed a tiny leak, the gangue coming with the ore changed a bit in its compositions, you had too much beer
while controlling things, ....
Enough "theory". Now let's go out and smelt. |
| |
| |
|
| |
| |
 |
I have (of course) never heeded my own advice. But others have, in particular
Darrell Markewitz, who works in the Lee
Sauder tradition. His account in what it takes to do proper smelting is quite "hands-on" and fascinating.
Here is his report. |
| |
| |
© H. Föll (Iron, Steel and Swords script)


