| |
10.1.5 Copper Final |
| |
Arsenic Copper |
 |
You are currently reading the 10th chapter of Iron, Steel and Swords. The nine
chapters ahead dealt essentially with the question why alloying iron with some carbon makes a big difference to properties.
If you grasped just the general ideas, it should not come as a surprise that alloying copper with this and that will also
change its properties. You also learned that some alloying might happen during smelting, and that you may not be pleased
with that. In other words: Alloying is a great thing to do - as long as you do it "just right". |
|
 |
Hardness was surely the first property the ancient metal workers would have liked
to improve. Or was it? We know that pure iron or copper is just too soft for chopping off heads, for example. Those old
guys (and Judith) thus must have gone for improved hardness one should
think. Well - not necessarily. Before you can chop off something with your copper sword or axe, you must first make it.
In the case of copper and copper alloys you make it by casting. Something easy to do, one should think.
No! In contrast
to popular misconceptions, casting is not easy. Even if you can get the stuff completely liquid, just pouring it into a
mold will not automatically make a good cast object. You might get porous stuff, for example, since gases trapped in the
liquid metal couldn't get out and cause bubbles like in swiss cheese or yeast dough. Or your mold simply explodes, not so
good either. Here is an example of some modern copper axe casting attempts, done by Julia Wiecken who isn't a clueless amateur
but knows exactly what she is doing: |
|
|
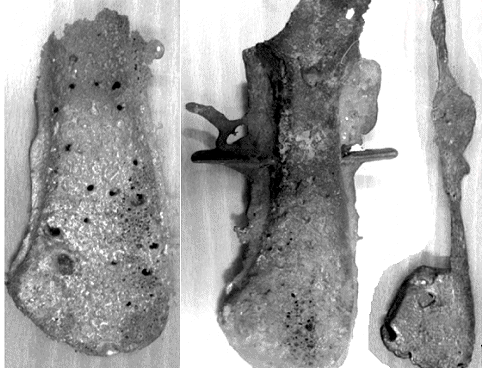 |
| Three tries at casting a copper axe following old "recipes" |
| Source: Julia Wiecken "Report on actualistic casting preexperiments – The
copper hammer and adze-axes from the Carpathian Basin" Historical Metallurgy Society, Coghlan Bequest. HMSN. NEWS Historical
Metallurgy Society 69, Summer 2008. |
|
| |
 |
Julia tried to cast a copper axe, following the old technique as closely as possible.
Here is the link to the actual paper. It just is
not quite as easy as you might think. |
 |
Proper alloying can indeed make casting much easier. And, as an added bonus, your
copper alloy might even get considerably harder! The only question is: how much of what
should I alloy for getting the best results? And why does the winning recipe work so
well?
If I would try to answer these questions in any detail and with scientific reasoning, I would have to write a
few more chapters. I won't. Just believe me without me proving everything.. |
| |
 |
The ancient metal workers in what is now Iran did not spend much time either worrying
about how to answer the questions above. What they worried about, I surmise, is why the guys down the valley got "better"
copper on occasion than what they produced, considering that those guys made copper the same ways they did as far as they
could tell.
In other words: there is no doubt at all that in smelting copper a lot of unintentional alloying took place.
You might also call it "getting dirt into the copper". Some "dirt" might be good for some properties,
others might be all bad. Look at the table below to get an idea of the wide range of unintentional
copper alloys produced as late as 1400 BC.
Chances of encountering all kinds of alloying / dirt effects while smelting
copper increased with increasing business. In very early small-scale smelting, alloying and other effects might not have
been very strong because simple and rather pure ores of the oxide or carbonate types were used, for example azurite and
malachite, that were picked up from surface-near deposits. Smelter temperatures
were not very high either, and no fluxes were added intentionally. That simply restricts what can happen besides making
some copper. Prying off the copper prills from whatever slag and residual charcoal was left, and melting these copper pieces
in air, purified the copper quite a bit since a lot of impurities evaporated or
oxidized (and then could be skimmed off). Very early copper therefore is often quite pure. |
|
 |
As production increased, you had to dig deeper for copper ores. Serious mining
started, and down there in the bowels of the earth you encountered more complex ores, typically sulfides, in all kinds of
mixtures. As production increased, all kinds of ores or ore mixtures made it into the smelters. Deeper down into ore deposits
you also tended to encounter minerals of other metals and mixed minerals that contained copper and
some other metal like iron, nickel or arsenic.
Temperatures in your more advanced smelters were higher, too, and weird
reactions could now take place. | |
|
 |
Elements that are often found in connection with copper ores had a higher probability
if ending up in your copper than the rest. Tin (Sn), the key element for making bronze proper, is not among the likely ones
so making tin bronze by accident is unlikely. The more likely elements were arsenic
(As), antimony (Sb), nickel (Ni), iron (Fe) and I don't know what else.
Almost everything was possible now, including
making some copper-rich (and thus useless) iron. Oversimplifying a lot, your smelting could go along four different ways:
- You produced "better" copper, i.e. a copper alloy with improved properties.
This happens, for example, if some arsenic gets incorporated, often together with antimony and nickel.
- You produced "bad" copper, for example if iron becomes alloyed.
- You produced usable copper plus other stuff like so-called "matte" (copper
sulfides) and iron.
- You produced no copper but something else like so-called "speiss",
an arsenic - iron alloy.
|
|
 |
Those must have been exciting times in metallurgy. Of course, what we find many
thousand years later are almost exclusively artifacts left from successful copper and copper alloy smelting. Nobody kept
the useless stuff, and nobody recorded the curses uttered when a smelting batch went wrong.
As Ünsal Yalçin
showed in the paper mentioned before, the "dirt"
concentrations contained in the earliest copper artifacts produced by smelting varied considerably but on a relatively low
level. Large variations in compositions seem to occur for pretty much all of early copper production, with the over-all
purity of the copper produced declining with increasing production in large furnaces. |
 |
Eventually some people somewhere realized that certain kinds of copper - like
arsenical copper - were just better than others, and that one could optimize the production of the "good" stuff
by putting the right ingredients into the smelter. Contrariwise, avoiding certain other stuff and practices made it less
likely that "bad" copper was produced.
That is straight evolution once more. The archeological evidence clearly shows that arsenic alloying was encouraged, while iron alloying was
avoided. To us that is clear because arsenic copper is "better" copper, indeed, while ferric copper is not good for you.
|
|
 |
About everybody in ancient times sooner or later produced arsenic copper intentionally. Iran is exceptional in its long-lasting preference for arsenical copper at a time
when most surrounding regions had already switched to tin bronze. Why is arsenic copper preferable to pure copper? There
are several reasons:
- It does make casting easier. Not only does the melting point decrease with As concentration, it also helps to avoid
all the negative effects of gases contained in the liquid metal.
- It does produce substantial hardening. Just 2 wt% As may increase hardness and tensile strength by 30 %. You also get
a larger work-hardening effect.
- It changes the color. In particular, arsenic copper may show a silvery sheen.
It may actually appear to be silver if the concentration of arsenic was high enough.
However! The first law of economics
still applies. You must pay a prize. . |
| | |
Arsenic (and its compounds) have this
unfortunate tendency to kill you!
|
|
|
 |
Even worse, arsenic tends to kill you sneakily. Arsenic and its poisonous compounds
easily vaporize, streaming out of your crucibles and smelters and poison the air you breath. Don't try to make arsenic copper
yourself!
Ötzi the Iceman, the well-preserved natural mummy of a man
who lived about 3.300 BC and was found in the Alpes in 1991, had a considerable concentration of arsenic in his body and
hair. This, along with Ötzi's copper axe made from 99.7% pure copper, has led scientists to speculate that Ötzi
was involved in copper smelting. |
 |
Anyway, arsenic copper was first produced more or less accidentally by "co-smelting"
copper and arsenic bearing ores like arsenopyrite (FeAsS), enargite (Cu3AsS4), domeykite (Cu3As)
and many others found here and there in copper ore mines. When you realize that there is some connection between the "input"
and the "output" of your smelter, you pay some close attention to what ores you use and how you process them before
smelting. You might even differentiate what you do. Some make regular, relatively pure copper for everyday uses, some others
(and somewhere else) make highly alloyed "silvery" prestige objects that are traded over long distances.
|
|
 |
Witness the Nahal Mishmar hoard found
in 1961 in a cave on the northern side of Nahal Mishmar, Israel. It dates to 3500 BC or older and includes 432 artful copper,
bronze, bone and stone objects; 240 mace heads, about 100 scepters, 5 "crowns"", and more. The copper objects
alone weigh 140 kg, constituting an immense value for the time. It is probably the collected treasure of a major temple
that was hidden for some reasons. Here is a picture of some of the objects; more can be found via this
link. |
|
|
 |
Parts of the Nahal Mishmar hoard
Large picture |
| Source: Courtesy of the Israel Museum |
|
|
 |
What exactly some of these objects signify is not all that clear.
There are also researchers who argue strongly that at least som of the objects are form a much later date 3). The round balls with a hole are mace heads. It is interesting in this context that a mace head
was the first metal weapon found so far, and that maces, good
for bashing your opponents skull in, were employed for a long time. The picture below shows a Hittite warrior from around
600 BC with a sword and a mace. Note also that there are no swords either in
the Nahal Mishmar hoard.
An amazing skill in casting technology 5500 years ago is demonstrated quite nicely by all
these objects. Particularly interesting for us is the large range of alloy compositions used for the objects as is directly visible from the quite different colors. Indeed, we have objects made from rather pure
copper, objects made from copper alloyed with up to 20 % of an arsenic (As) - antimony (Sb) mix, and probably anything in
between. Some more pictures are here.
One picture shows the "silver sheen" quite nicely.
There are indications that the alloyed copper objects
in the hoard were imported from somewhere. It is no yet known from where exactly but definitely not from close by. |
| |
 |
| Late (600 BC) Hittite God / warrior with sword and mace. |
| Source: Photographed at the Karatepe / Turkey open-air museum. |
|
 |
So how do we alloy copper with arsenic? Paying close attention to what you put
into your smelter will only get you that far. For achieving copper with a defined concentration of arsenic - say 3 % ±
1% - too many things need to be "just right". You simply can't monitor your smelter and its input closely enough,
and there is no guarantee that you get the 3 % stuff all the time. While co-smelting most likely was the technology used
most of the time, the archeological evidence strongly suggests that some more sophisticated alloying process must have been
used after an initial (long) phase of co-smelting and hoping for the best. So how was it done? |
|
 |
"Where is the problem?" you might ask. "So these guys eventually
smelted pure metallic arsenic and mixed it with pure copper, melting it together, just as they did with tin somewhat later".
Good thinking. Just not correct, sorry. You cannot smelt arsenic in the usual way because arsenic doesn't melt but turns
into a gas right away when it reaches a relatively low temperaure of 613 oC (1135 oF). Skipping the
liquid phase and turning from a solid to a gas is know as sublimation. You know it from
frozen carbon dioxide (or dry ice) that produces all these dramatic "fog" effects in theaters and night clubs
when it sublimates in a water-rich environment (the freezing / sublimation temperature is -56.4 °C (-69.5 °F)).
Arsenic is even worse than zinc that also vaporizes inside the
smelter. Arsenic gas, of course, will quickly move up the burden, oxidizing as soon
as it meets oxygen. It will solidify again on cold surfaces - and it kills about everything in its way!
So nobody made
solid arsenic in ancient times. Co-melting solid arsenic with solid copper wouldn't have worked well anyway, since most
of the arsenic would have evaporated off long before the copper would get liquid. |
 |
Making a defined copper - arsenic alloy thus
is quite difficult. But some of the ancient guys obviously had figured out some way of doing that - but to archeometallurgists
it is far from obvious how, exactly, they did it. There is a lively discussion going on just now, fueled by new results
coming in regularly. I will only give you a taste treat of what is going on. |
| |
 |
In a very recent paper Pernicka
and others 1) present rather convincing evidence that at least the old Arismanians
in North-West Iran around 3000 BC deliberately produced speiss, an iron-arsenic alloy, which
then was used to produce arsenical copper. Most likely the ore "arsenopyrite" (FeAsS) was first smelted to produce
speiss, which then was added to metallic copper or as ingredient for a smelter dealing with "proper" copper ore.
The copper produced contained arsenic (good!) and iron (bad!), but the iron more or less removed itself by oxidation if
the copper was melted in air. |
 |
Let's leave it at that and only give a passing look at a magnificent cast arsenic copper head that may be linked to the art of the Elamites, early masters of metallurgy. It is almost solid and thus rather heavy. The Metropolitan Museum in NYC
does not state if the arsenic content was high enough to produce the "silver sheen" possibly sought after for objects like this.
|
|
 |
Of course, the Metropolitan doesn't know exactly where this object comes from
because, like most objects shown in the Metropolitan and many other museums, it goes back to illegal digging and trading
if you believe Oscar Muscarella, something I tend to
do. |
| |
 |
Head of a Ruler; arsenical copper, Iran; ca. 3200 BC - 2000 BC
This might be Ur-Nammu
(or Ur-Namma, Ur-Engur, Ur-Gur) ca. 2047-2030 BC who founded the Sumerian 3rd dynasty of Ur 2) |
| Source: Photographed in the Metropolitan; NYC |
|
| |
 |
A lot more could be said about arsenical copper but, to the best of my knowledge,
archeometallurgists are far from having come up with the "final" words to the topic. So let's desert the issue
here and move on to: |
| |
| |
| |
Bronze |
 |
Copper bronzes still are a diversified and thriving part of modern metallurgy.
We use aluminum, lead, phosphorous, silicon, and (insert name of chemical element) bronzes
nowadays; plus tin bronzes, the "classical" bronze. Just to be clear: if I look at bronze
in antiquity, I only mean tin bronze. Of course, antique bronzes also contain all kinds
of trace or "dirt" elements besides tin - but let's not be picky and forget about that.
Bronze was THE first
"metal" that changed humanity for good. Actually, you just as well could say it changed humanity for worse because
"industrially" produced bronze weapons allowed large-scale warfare. Slaughter on a magnitude unprecedented in
earlier times commenced around 2000 BC. |
|
 |
The word "bronze" appears first around 1730; it goes back to 13th century
Italian "bronzo" meaning bell metal or brass. The deeper roots are unclear; the Persian word for copper, "birinj",
might be the source.
Bronze contains typically around 10 % tin; up to 20 % might also be encountered. The microstructure
can be rather complex. If you don't scare easily, you might consider taking a look at the phase
diagram. |
 |
As we can see from the time / geography diagram of copper uses given before, all civilizations sooner or later changed from making copper and arsenical
copper to making bronze. It is just so much better:
- Alloying is easy and controllable (provided you have tin) and doesn't kill you because the ingredients are not toxic,
in contrast to arsenic.
- Casting is much easier (lower melting point and less problems with gases, etc.)
- It has a very good corrosion resistance, making it suitable for ship fittings (very important around the Mediterranean).
Even ship propellers today are still made from some bronze.
- Bronze is
considerably harder then wrought iron or other Copper alloys
then in use. Its Vickers hardness ranges form 60–258 vs. 30–80 for the others.
|
|
 |
How good is bronze with
respect to hardness? Here is the decisive figure from the extensive work of Heather Lechtman: |
| |
 |
Hardness of Tin (Sn) and arsenic (As) bronzes in comparison to carbon steel.
|
| Source: Heather Lechtman: "Arsenic Bronze: Dirty Copper or Chosen Alloy? A View
from the Americas"; Journal of Field Archaeology, Vol. 23 (1996) p 477 - 514 |
|
| |
 |
Surprise! Not only is arsenic copper just as good with respect to hardness than
tin bronze, both are just as good as normal steel not containing martensite
from quenching. However, looking a bit more closely, and also at concentrations of arsenic or tin far above 1 %, tin bronze
wins against arsenic bronze with respect to mechanical properties and all the other points made above. Wrought iron or mild
steel, however, has no clear advantage. |
 |
Making and casting bronze became a large-scale industry that came into its own
around 1500 BC in the countries around the Mediterranean. Many beautiful
works of arts, in particular from the "late bronze age" (after about 1500 BC), with intricate details are
displayed in museums, including large sculptures and useful, if expensive objects like the one shown below: |
| |
|
 | Life size bronze statue of some Roman senator.
2nd century AD, found submerged |
| Source: Photographed in the Archeological museum, Adana, Turkey. |
|
| |
|
| |
 |
The cauldron stand is displayed in the "Neues Museum",
Berlin; Germany (that's where you find Nefertiti, too). It is probably from Kition / Cyprus. Note that it was broken
and mended rather clumsily.
A large-size picture of a ritual Hittite bronze axe can be found here |
 |
Tin bronze (Cu-Sn) alloy became known in the late 4th millennium BC and the beginning
of the 3rd millennium BC in Mesopotamia and western Iran (Khuzestan and Luristan regions). Early bronzes in some places
might have been produced by accidental co-smelting of copper ores and cassiterite (SnO2), the only tin ore around.
Cassiterite is rather rare, however, and it was and is a matter of much contention in archeology where the many bronze makers
in antiquity got their cassiterite or metallic tin from. |
|
 |
To cut a very long and confusing story short, copper and tin eventually became high-volume
trading items in the second millennium BC. Cyprus was a major producer of copper,
and the "ox-hide"
copper ingots typically recovered from trading ships lost in the Mediterranean
can be found in about any archeological museum in Europe - except the
one on Cyprus, it seems.
A lot of what we learned about the metal making and metal trade in the heydays of the bronze
Age comes from the "Uluburun ship" that sank around 1320 BC.
Below
is the Berlin ox-hide copper "ingot"
An ingot,
by the way, is a piece of relatively pure material, usually metal, that is cast into
a shape suitable for further processing. Pieces of iron used for trading thus are not
ingots, even so some famous museums address their iron this way. | |
|
| |
 |
26 kg copper "Oxhide" ingot; 2 000 BC - 1 000 BC, Cyprus
|
| Source: Photographed in the "Neues Museum", Berlin; Germany. |
|
| |
 |
"Oxhide" tin ingots are less ubiquitous but also exist. Cornwall in England (very close to the edge of the world in those times) was a known source, and a few more
had been known around 1980, but these known tin sources were simply not enough for the bronze industry. Meanwhile, more
than 40 ancient sites of tin mining have been discovered in southern Turkey, and our knowledge of ancient metallurgy has
changed and improved ever since. I doubt that the last word is already out on the topic and thus will not dig deeper into
the subject.
More to tin and other early metals can be found from the link hub. |
|
|
 |
Making tin bronze thus could be done by simply co-melting the metals and I won't
go into this anymore. Much trial and error must have occurred, but eventually copper, tin, and bronze technologies were
mastered quite well. Around 285 BC, for example, some Chares of Lindos had the Colossus of Rhodes erected, a 30 m tall bronze statue, made from 500 "talents" bronze (15 tons)
and 300 talents of iron (9 tons). The Assyrian king Sennacherib (704 BC -681 BC) claims to have cast a 30 ton bronze statue,
and he might not have been the only one. |
|
 |
For comparison: The bronze Bavaria in Munich, cast around 1848, is 18.5 meters
high and weighs about 87 tons. The Statue of liberty in New York was erected in 1886, is 46 m tall with a 27 ton copper
skin. 350 million $ were raised to allow restoration of the statue in 1984, to give a rough idea of the kind of money one
needs for showing off in style. |
 |
The following figure gives an idea about technology development
in the second half of the second millennium BC in Iran |
| |
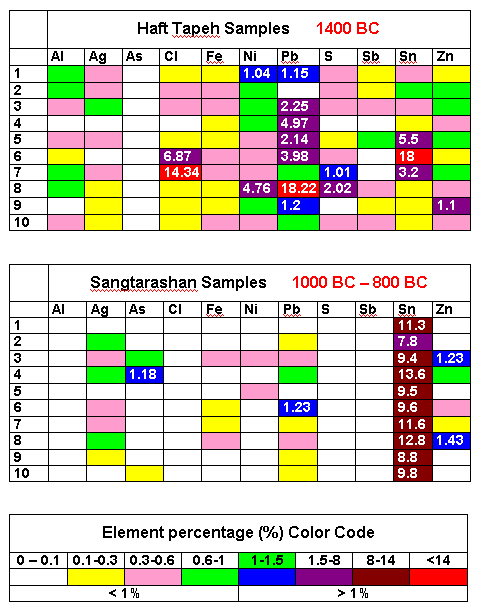 |
| Composition variation of copper / bronze from 1400 BC to 1000 BC |
| Source: Redrawn from: Omid Oudbashi, S. Mohammadamin Emami and Parviz Davami (2012).
Bronze in Archaeology: A Review of the Archaeometallurgy of Bronze in Ancient Iran, Copper Alloys - Early Applications and
Current Performance - Enhancing Processes, Dr. Luca Collini (Ed.), ISBN: 978-953-51-0160-4, InTech |
|
|
 |
While the earlier samples - all from the same place and time - showed wild variations
in the impurity concentrations, 500 years later the alloying
element tin (Sn) was obviously intentionally added with a well-controlled concentration around 10 %. Similar observations
hold for other places. |
 |
All of this adds up to a simple conclusion: around 1500 BC a well-developed smelting
technology was in place in many areas of the Old World that could produce copper, tin, bronze (Cu + Sn) reproducibly and
in quantity. Not to forget, plenty of lead, silver, mercury and even
brass (Cu + Zn) were also produced in more or less tricky ways. Some rather involved techniques for parting and purifying
some metals also existed, look up the "Early Metals" module.
The big question now is: Why the hell shouldn't those guys not have made iron and steel?
I'll come to that but want to point out already here that the emergence of iron and steel technology was not an isolated
breakthrough but part of an existing and already highly developed engineering system. If not much iron and steel was produced
before 1200 BC, it was not so much because it couldn't be done but because those guys didn't want to do it. |
| |
 |
Before I now finally move to iron and steel, let's give a first quick look at
how copper and iron smelting hang together. |
| | |
|
| |
Speiss, Matte and Iron in Copper Smelting |
 |
As mentioned before, things get more complicated when you run out of clean, simple
oxide or carbonate ores. Typically, these minerals formed when the original ores came
in contact with air (and water), and that happens when they are close to the surface. All the way down in the bowels of
the earth you have the original ores, typically sulfides. In between things are complex. Surface-near ore might have dissolved
in rain or ground water and then got washed down deeper, where it precipitated again. Over million of years a complex mix
of copper oxides, carbonate, sulfides and whatnot built up, together with "mixed metals": ores containing arsenic,
iron, antimony, nickel and whatever else was around.
Put sulfides
or more complex stuff in your smelter and all hell will break loose, at least in an olfactory sense. Some of the sulfides
and sulfates will become oxidized, and your contraption will exude hellish fumes like sulfur dioxide, SO2. The
oxidized ores can become smelted, i.e. turned into metal and carbon dioxide. The surviving sulfides will not get reduced,
however, but will generate trouble. It is therefore a good idea to "roast" your ores before you put them into
the smelter. Roasting turns sulfides into oxides. It doesn't need very high temperatures
and can be done on a kind of griddle with a regular fire below. |
|
 |
That's what ancient metal workers did when smelting copper was becoming a real
industry in the second millennium. Roasting ores wasn't particularly healthy, especially if they also contained some arsenic,
and it was a good idea to say upwind of roasting griddles and smelters if you could. Ancient metal workers, however, rarely
succeeded in oxidizing the ores a full 100 %, and some sulfides still went into the smelting furnace.
By
then also some iron oxides went into the smelting furnace. Either because they happened to be around or, more likely, because
they were added intentionally as flux. Iron oxides, together with the ubiquitous always present silicates (things containing
silicon dioxide, SiO2), produce fayalite, (Fe2SiO4), a great slag material.
| |
|
| |
|
We know that this was done because the slags from advanced copper and iron smelters
are virtually identical in their compositions! Smelters were also relatively large and could easily reach high temperatures
around 1300 0C or above. What happened under these circumstances is quite complicated and will lead us straight to iron smelting.
|
 |
I have made clear by now that in the second millennium BC copper
and tin smelting was done on a large scale, and with a rather sophisticated underlying
technology and infrastructure. Around a smelter things still looked
rather primitive but that was and is deceiving.
It is important to realize that bronze wasn't the only metal (alloy)
produced in antiquity. Including iron, there were the "seven metals of antiquity": Gold (Au), Silver (Ag), Copper (Cu), Iron
(Fe), Tin (Sn), Lead (Pb) and Mercury
(Hg). In an indirect way there was also Zinc (Zn) since brass, a copper zinc alloy,
was made but without ever making elemental zinc. This special module
tells it all. Besides the smelting required for making all these metals except gold and mercury, other rather tricky technologies
had been invented for purifying and "working" these metals. And quite a few of these technologies existed before
iron was produced on a sizeable scale.
The question I'm leading up to once more is: why did iron come in so late? Is
it especially difficult to make or what? Before I go into this, let's ask a more general questions: what about the many
other metals the periodic system provides for? Why wasn't there nickel (Ni), cobalt (Co), chromium, (Cr), manganese (Mn),
titanium (Ti), or platinum (Pt), to name just a few? After all, we can be sure that after the discovery of smelting, some
early enterprising engineers and scientists must have tried to pry out metals from about every rock and mineral they could
find. |
|
 |
I have already answered this questions in parts - look up this
module and here you find probably more than you are looking for! Some metals simply cannot be smelted with carbon or
carbon monoxide as reducing agent. Some metals, including iron, could be reduced to the elemental stage but not melted and
thus would not collect as liquid pool in the bottom of your smelter. You only could "make" them if you managed
to condense them in a safe place below the tuyere in the solid state as a "bloom". While this works with iron,
it may not work with other metals. I actually don't know because nobody, it seems, has tried to do this, for example, with
manganese (Mn, melting point 1244oC; 2271oF), nickel 1535 (Ni, melting point 1453oC; 2647oF)
or cobalt (Co, melting point 1495oC; 2723oF). It may well be that for one reason or another, all these
metals were just not "in reach" in antiquity but I suspect that some metals actually could have been made but
had no conceivable uses. Future archeologists digging through the remains of the 20th century will not easily find indium
(In) or gallium (Ga) since they were more or less useless, even so these metals were produced (as by-products of other metals).
Only in the last few decades they have become key metals for some High Tech applications. Manganese (Mn) by itself, for
example, wouldn't have been of much use in the ancient world and alloying it to iron was impossible since iron could not
be melted. |
 |
To make a long and complex story simple, I will just give you some results of what people in
the second and particularly in the first millennium BC experienced in their metal smelting practice. |
|
 |
1. The made Speiss
Speiss
is a collective name for all kind of arsenides and antimonides
produced in a smelter. Typically we have iron and copper arsenide. Since arsenic and antimony are metals (sort of), speiss
is thus a metal (alloy), too. It came up above in the context of making arsenic copper
but it actually comes up all the time. If it wasn't smelted intentionally, like in the case above, it was often a by-product
of copper and lead smelting. Arsenic copper could be seen as an extreme case of speiss on the low arsenic side.
Much
more could be said about the role of speiss through the millennia, not to mention the etymology of that word. It's rather
strange in English - and rather common in German: Speiss relates to "Speise"=food, "Speis" is south-german
dialect for mortar, and bell-makers still use "Speise" for casting bells. The verb "einspeisen" means
to inject, feed, supply with. You get the drift. |
|
 |
2. They made Matte
Matte,
once more, is a collective name for metal sulfides produced in a smelter, typically
Cu, Fe, Ni and Pb sulphides. Sulfur (S) is not a metal, so sulfides aren't metals either. Matte may form as an intermediate
product when smelting sulfide ores, especially for copper and lead. The reason for matte formation is simple: while you
can turn a sulfide into an oxide with heat and oxygen, i.e. by roasting in air, you cannot reduce a sulphide by carbon monoxide
in the smelter. The sulfides getting down into the reduction zone thus end up as molten layer on top of the molten copper
layer and below the slag layer, since these liquids don't mix and arrange themselves according to density.
Matte is
a heavy brittle black stuff when solidified; it was considered a nuisance in early smelting days. It is actually a very
rich copper ore that can be smelted to the pure metals in a second go - if you know how to do it. I give you a hint: Roast
it well in air and add silica as flux - that will do the trick. Much of the ancient matte has been used for copper making
somewhat later.
Matte is once more a strange word in English and not-so-strange in German. |
|
 |
3. They made iron
So you want to make copper.
You feed your smelter with roasted copper (+ arsenic) ore; a mix of oxides and sulfides and God knows what else. You add
some iron oxide and sand /silicon oxide as flux plus plenty of charcoals. Now consider: |
| |
Why shouldn't you get some iron too?
|
|
| |
|
You expect to get some arsenic copper, some matte and maybe some speiss. Fine.
You called the iron oxide that you put in your smelter "flux" - but you just as well could have called it "iron
ore".
The truth is that you did get iron if you weren't careful. Making iron accidentally was a real hazard in
copper smelting, and could best be avoided by keeping the temperatures as low as possible. The reason is simple: you get
iron by either reducing the ore at moderate temperatures high up in the burden or by reducing the iron-rich fayalite slag swimming on top of everything else all the way down. As long as the iron produced remained
solid, it couldn't do all that much. At worst some particles, or a thin layer of iron particles, floated on top of your
molten copper, perhaps. However, if the iron could incorporate enough carbon to turn into cast iron, it liquefied and caused
trouble. At higher temperatures this is more likely to happen.
Copper containing as much as 35 % iron has been found.
It is entirely possible that ancient copper guys produced iron-rich copper quite often. It was, however, not too difficult
to purify iron-infested copper: just melt it. Anything above the solubility
limit around 4 % will precipitate and float to the surface, where you can pick it up as iron!
The rest you can get out by blowing air onto you liquid copper, oxidizing the iron but not the copper. Add a little silica
and you get slag, sitting on top and thus easily removable. There are two points of interest here:
The first
point is: The ancient copper guys definitely knew about iron. However, what they got was iron contaminated with copper,
and that was and is "bad" iron, with properties nobody likes. The second point
is: The ancient copper guys definitely knew about refining a metal by exposing it to oxygen in its liquid state. That, to
refresh your memory, is the way steel is made from cast iron. |
|
 |
4. They made mixes of all of the above
The picture
shows a kind of (highly idealized) worst case where you get everything during copper smelting. Typically, either matte or
speiss dominates in copper (sulfide) smelting. However, in lead smelting you might have gotten everything except, perhaps,
the iron because you could keep temperatures relatively low. |
| |
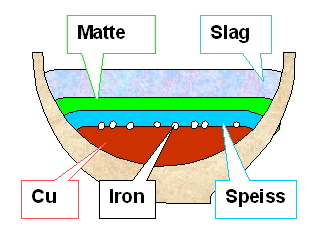 | | What you found in your crucible |
|
 |
Even present-day copper smelters worry about matte and speiss. If you produce
either one, your efficiency is not as good as it could be. Indeed, modern conventional metallurgical wisdom has it that
the production of speiss has to be avoided at almost all cost, since it is more harmful than useful for any practical purpose |
| |
|
© H. Föll (Iron, Steel and Swords script)









