6.2 Things Get Complicated
6.2.1 Creamy or Chunky?
Since you only have a certain amount of the hard stuff, you can turn it into a lot of little particles, into just a few big ones, a mixture of big and small ones, or anything in between.
| What's the best? |
The answer is clear, of course: It's a matter of taste as far as chocolate ice cream or peanut butter is concerned. As far as nacre or steel is concerned, it's also a matter of taste with regard to the properties you want.
As far as steel is concerned, the general agreement is that you want it hard but ductile.
Hardness, as we have learned before, is simply a measure of the yield stress, the force per area at which dislocations begin to move. Hard but still ductile steel means that it's difficult but not impossible for the dislocations to move. If dislocation movement is impossible (like in cementite) you have a brittle material, and that's not what we want.
The long and short of this is that we must allow the dislocations to move around a bit. Movement shouldn't be easy, however, and we want to be in full control.
It's a bit like keeping teenagers under control. If you ground them completely, things break. You must let them roam around a bit but it is wise to slow them down, e.g. by keeping in control of car keys and money.
For slowing down dislocations in steel, we use our hard cementite particles.
| Hardening steel (or any other metal for that matter) means to put obstacles in the way of its dislocations. |
- Extrinsic point defects or foreign atoms dissolved in the matrix.
- Precipitates, made from the foreign atoms we have inside the steel.
- Grain boundaries.
- Other dislocations.
- A metastable fourth phase of iron called "martensite".
I you want to use martensite hardening you really must torture the crystal. You must force it to make something it really, really hates: martensite!
A metastable phase is often just a transient structure encountered on the way from one clean and desired phase to another one. In other words, it's something like a teenager—no longer a child but not yet an adult.
A diamond, by the way, is a metastable phase of carbon. So you teenagers out there can stay cool and refrain from sending me hate mail. Metastable states can be quite valuable and they can last a long time. In this case we treat them like a stable phase and use them even in phase diagrams.
Nevertheless, they are not the nirvana states or the best of all possible states. You might perceive them in terms of an extremely simple diagram:
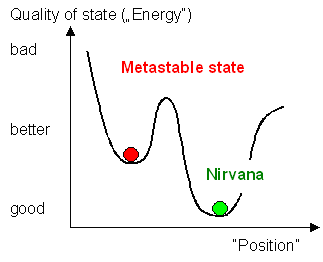 |
| Stable and Metastable States |
Words like "position" and "way" do not exclusively refer to common space but might be metaphors for more abstract entities.
However, you might get stuck in a side valley that does not have the lowest possible free energy. If it is difficult to get out of there, you are stuck in a metastable state. You only get out of it and over to the deepest point by first worsening your situation again. You must first go up to be able to get down to nirvvana.
If you don't have the energy any more to do this you are now trapped in the metastable state until you can get it up again. That might take quite a while, depending on circumstances.
Cementite in iron, by the way, is also only metastable. The stable state of carbon in iron would be graphite. Cementite is, however, stable enough not to do anything in your and my and our offsprings lifetime, so we do not need to worry about this.
| What happens when we go from the liquid phase to a two-phase region? |