| |
5.4 Weird Denizens of the Defect Zoo |
| |
5.4.1 The dislocation |
 |
I can no longer avoid to introduce you to one-dimensional
defects. There is only one kind: the dislocation. |
|
 |
A dislocation, when you first meet one, appears to be rather fascinating
but weird. You don't know how to handle it, what it is good for, and what to do with it. It's just like a new-born baby.
So let's just make one (a dislocation, not a baby). That's relatively easy, even if you don't know exactly what goes on
during the first few tries. Go through the moves a few times, and you will get the drift, just like - well you also get
this drift. |
 |
First we look at a schematic drawing of a three-dimensional perfect crystal as
shown below. It's schematic because I spared you (and me) all the atoms inside the crystal. It's also a cubic primitive
element crystal that doesn't really exist but is easy tro draw. Since just showing atoms is a bit confusing, I also indicate
some particular planes in blue. |
| |
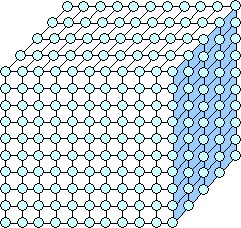 |
Here is a schematic perfect cubic primitive crystal .
One plane of atoms is marked in light blue. |
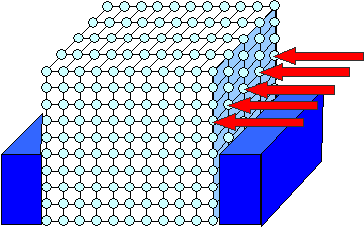 |
The crystal is clamped into a vise and subjected to some shear
stress (red arrows).
The shear stress is applied because we try to
move the upper part to the left. |
| Generating a dislocation; part 1 |
|
 |
We put our model crystal into a vise and now press like crazy
on the upper right hand part. Bang it with your hammer if you like, but the stress distribution generated in this way tends
to be a bit messy (the red arrows would have different lengths and would not be exactly at right angles to the crystal plane)
.
Essentially we want to move or shear the upper part of the crystals somewhat
to the left. If enough force, pressure, bang, or to call it by the correct name: stress is exerted on that outmost plane of atoms, it will eventually do an interesting thing
as shown in the next picture of our little comic strip: |
| |
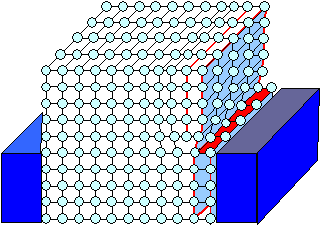 |
The upper part of our schematic crystal has yielded to the shear
stress. The outermost plane of atoms has curved inwards and connected to the atoms in the lower part. There is now
a step and one "half-plane" of atoms ends inside the crystal.
A dislocation
has been generated.
|
 |
Keep stressing and the dislocation from above will move through the crystal as shown.
For simplicities
sake we are only looking at the front side of the crystal. |
| Generating a dislocation, part 2, and moving it |
|
 |
The outermost plane with all the atoms on it is squeezed so much to the left that
it eventually squeezes the neighboring planes of atoms so much to the left that the bonding between one row of atoms snaps.
The outermost plane takes the part of the second plane, bonding with the atoms below. The second plane then ends
inside the crystal. |
|
|
We have generated a dislocation!
|
|
|
 |
To be more precise, we have generated a particular kind of dislocation, called
an edge dislocation.
If we keep pressing, the dislocations moves. The ending
plane of atoms joins up with the plane next to it and thus moves deeper into the crystal. The animation shows that schematically,
but be aware that this happens all along a line at right angles to the screen..
What an edge dislocation deep in a crystal finally looks like is shown below in a thee-dimensional rendering
. |
| |
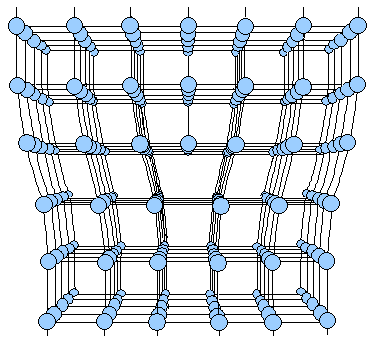 | | Perspective view of an edge dislocation |
|
|
 |
If we keep pressing, the dislocation will move all the way from left to right,
finally emerging on the right (look at the animation). The net result is that the upper part of the crystal has been displaced
relative to the lower part.
In other words: |
| | |
|
|
 |
In yet other words: We have moved a big object—half a crystal—not in
one fell swoop that would have required to break a lot of bonds and thus a lot of energy, but step-by-step, with rather
little effort. We have sheared off the top relative to the bottom and that's why we call that kind of deformation a shear deformation, the kind of deformation you get when you exert force parallel
to a surface.
Dislocations can only produce shear deformations
and never what we call normal deformations, the kind of deformation you get by
pressing normal to the surface on things. This
link illustrates and explains that in more detail. |
 |
Mother nature, of course, has discovered the principle of moving big objects the
easy way long ago and implemented many variants of that basic principle. |
|
 |
In our case we want to move the whole top of the crystal to the left.
The idea is not to move the whole body with brute force, but only small parts of it at any given time with far less effort.
To say it a bit differently: Localize a certain deformation in a small part of
the body and then move that "defect" through the body.
The following pictures show this principle for two
biological objects and a man-made object (big rug). In all cases it is far more efficient to move a "fold" through
the object than the whole object. In crystals, dislocations do just that. |
| |
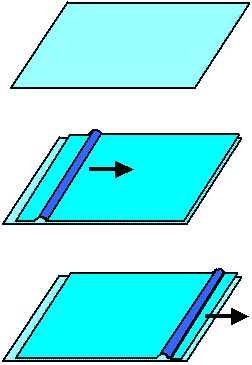 |
| Moving big things with little effort |
| The basic principle in all cases is to move a localized defect through
the structure rather than moving the whole structure at once. |
|
 |
Crystals knew all along, for the better part of 10 billion years, that life with
defects like dislocations is much easier than being perfect. |
|
 |
Humans, however, did not figure out that dislocations are absolutely essential
to sword forging before 1934; we shall see why in a moment. About 3.000 years had to pass for iron technology, and far more
for general metal technology, before we first understood what was going on when "bending" a metal. Dislocations
are not only essential for sword forging but for all the bending done by the "metal bending industry, as we call our still largest and most important industry.
Compare that to the first "use" of electricity by Volta
around 1770, and a full-fledged industry based on an almost complete understanding of electricity around 1900. |
|
 |
Dislocations are also extremely important for most of semiconductor electronics
because there you must not have them! Since you can only avoid something if you know
what it is, silicon technology would be impossible without a thorough knowledge of dislocations (and other crystal lattice
defects), including how one can avoid having any in the silicon wafers one starts with, and
how not to produce them during processing. |
 |
Nevertheless, lots of peoples missed the big moment when Taylor, Polyani and Orowan discovered dislocations in 1934. |
|
 |
Worse, it took even until about 1960 before dislocations became common-place in
scientific and engineering circles. The big breakthrough was the invention of the transmission
electron microscope (TEM). Do you know who invented it?
I thought so. Go to this link.
Do not collect 100 Dollars.
Anyway, since about 1960 one could see dislocations,
and seeing is believing. |
 |
I will allow that a lot of people were a bit distracted beeen 1930 - 1950. Some
other events like the Second World War happened in this period of time and no Noble prizes were awarded. After the war,
lots of Nobel prizes went to the heroes of atomic and nuclear science who unveiled major secrets of nature and produced
atom bombs on the side. That was fine. It is nevertheless inexcusable that no Nobel price was ever awarded to the scientists
who dug down to the very roots of mankinds most important technology. |
|
 |
The Nobel price committee tried to make up for this oversight by awarding a Noble price in 1986 to Ernst Ruska
(1906 - 1988). He was one of the heroes around the invention of the electron microscope and in 1986 the only
survivor from the heroic times when dislocations and other defects in crystals were discovered and we finally understood
what is going on inside a metal when you bang it with a hammer or work with it in any possible way.
So if you, like
me, haven't been awarded a Nobel price so far, you are in very good company.
The special module gives you an idea of
the people who were instrumental for understanding sword forging. |
|
|
 |
Just to make sure, let's look once more at how dislocations achieve
plastic deformation in a schematic way. |
 |
 |
 |
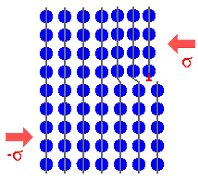 |
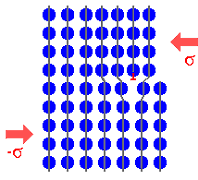 |
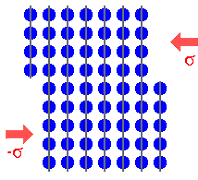 |
| 1. Formation of a small step by generating a dislocation. |
2. Moving the dislocation through the crystal. The upper half has slipped some. |
3. Emergence of the dislocation on the other side, after the dislocation has moved all the
way through the crystal. The upper part has slipped completely | |
The three major steps in plastic deformation as seen from above and sideways. |
|
 |
We produce a dislocation by pressing as shown below (or above); next it moves
through the crystal. When it emerges on the other side, the total effect is that we moved all
the atoms in the upper part of the crystal with respect to the lower part by one atomic distance.
As long as the dislocation is still inside, only parts of the upper half has moved. |
|
 |
Did bells ring in your brain? Did you note the magic word? Here it is: "we moved all the atoms". We moved atoms with a dislocation!
Moving atoms, I'm
sure you will recall, was at the root of changing something in a crystal and that was the key to changing properties. The
magic word was diffusion, or the random
movement of atoms by some mechanism.
With a dislocation we move a lot of atoms too, but pretty much in lockstep, the exact opposite of random atom movements
in a diffusion process. We shall call this kind
of moving atoms around a military movement. Moving atoms around individually and
at random by diffusion, we then shall call a civilian movement on occasion. |
 |
The dislocation itself is the region where things don't fit anymore. It is a line defect, with an extension in just one direction. Along the dislocation line,
atoms would not see their usual environment, so we have a one-dimensional defect. |
|
|
|
 |
There is far more to dislocations than I can relate in this introductionary subchapter.
The (two) science modules give a small taste treat of some of the more elementary stuff encountered when looking a bit closer
at dislocations. | |
 |
Now get ready for the climax! |
| |
|
© H. Föll (Iron, Steel and Swords script)













