| | |
Creep |
|
|
Basic Mechanism of Creep |
 |
Creep is the word for processes that lead
to a very slow plastic deformation (and eventual fracture) of materials that are kept
under a constant load that is far to small to cause
plastic deformation right away in some tensile test. That's
a definition that holds for all materials, not just crystals. |
|
 |
In what follows I confine myself to crystals. Then we may discuss dislocation
creep because there is never any plastic deformation without dislocations.
Here is what creep looks like in a simple diagram: |
| |
| |
 |
| Creep diagram. |
|
| | |
|
|
 |
A "creepy" tightened screw under constant load (= constant stress) slowly "gives",
meaning that it gets longer (= strain increases). Eventually it will fracture or break. This can take many years or just
a few months. You might have experienced the latter case with things like the cheap screws on your cheap bicycle that miraculously
always are loose after a year, no matter how hard you fastened them. That's because of creep and not because the nut somehow
turned. |
 |
Slightly changed dimensions because of creep may not be very visible but are potentially
disastrous. If tightly fitted components in some product change their dimensions just a little bit, the consequences can
be real bad. The example of the screw in the figure above illustrates that nicely. |
|
 |
Any screw is tightened by putting it under some tensile stress. This stress should be below
the yield stress so the screw is only strained elastically. If creep occurs, the screw will elongate plastically v
e r y s l o w l y and this relaxes the stress. The process ends when the stress is zero, and that means
that the nut is now loose.
Note that the diagram above does not really represent
the tightened screw case, implying that the load or stress decreases as the screw gets
longer, but a case with constant stress all the time, e.g. when the screw is holding
a weight. Under circumstances with constant stress, the material will eventually fracture. Before it does that, however,
it will have noticeably changed its shape / dimensions. |
 |
So why? What exactly happens inside the material?
We did not apply stresses above the yield stress, so dislocations should neither move nor be generated. That is still true
but only on a macroscopic scale. We must give dislocation movement a closer look to understand what is going on. |
|
 |
Let's consider a perfect crystal with no defects whatsoever, except a few dislocations.
Now do a tensile test. The material will be relatively soft with a low yield stress. The reason for this is clear: No defects
= no obstacles for dislocation movement besides the natural resistance of the perfect crystal (called Peierls
stress, by the way).
Now harden the material, for examples with precipitates. The "global" yield stress is
much larger now and the dislocations cannot move globally even if the applied stress
is larger than the Peierls stress. There is no global plastic deformation. The dislocations, however, can move locally until they encounter a precipitate where they get stuck. |
| |
| |
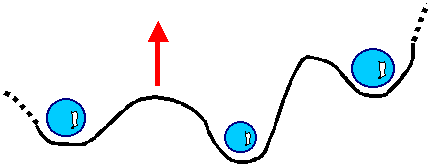 | | Dislocation stuck at precipitates. |
|
| | |
|
|
 |
The picture shows a dislocation (black line) that tries to move in the direction of the red
arrow. It is stuck at precipitates (blue spheres) of a different phase. It can't move globally. All it can do is to bow
out a bit locally between the precipitates as shown. |
 |
If there is creep, there is plastic deformation. That necessitates that dislocations
must move large distances and that can only mean that dislocations get unstuck ever so often. Then they move until they
get stuck again at some precipitate in the direction of their movement. Then they get unstuck and move a little bit once
more. And so on. And every time they move a little bit, there is a little bit of plastic deformation. Over the years it
adds up and the screw gets measurably longer. |
|
 |
So what kind of mechanism can make a dislocation unstuck? Well—simply increase the stress
until the yield stress of the hardened material is surpassed! Yes, quite true, but when we consider creep we keep the stress
constant and below the yield stress.
So once more: what kind of mechanism can make
a dislocation unstuck? I will give you a hint: creep gets more efficient or faster if you increase the temperature. |
|
 |
The answer is, as (almost) always: You need to move atoms. In other words: you need diffusion,
typically via a vacancy mechanism.
On order to understand how this works, let's look at a stuck dislocation from the
side, i.e. we are looking along the dislocation line |
|
| |
| |
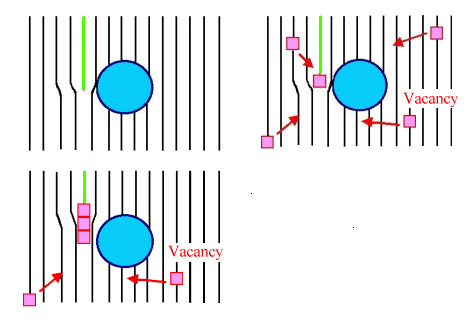 |
| Stuck dislocation becomes unstuck with the help of vacancies. |
|
| |
|
|
 |
Upper left: The dislocation
is symbolized by the ending green lattice plane. It wants to move to the right but is stuck at the precipitate.
Upper right: Some vacancies are always moving around. Very few move very slowly at low
temperatures, far more move far faster at higher temperatures. Some get caught at the dislocation line. This means that
there is now an atom missing in the dislocation line.
Lower left: Several vacancies took a whole bite out of the half-plane defining the dislocation
line. This does not only happen in the plane of the drawing but also "ahead" and "behind".
Can you
imagine this in three dimensions? If not, look at the next figure: |
|
| |
| |
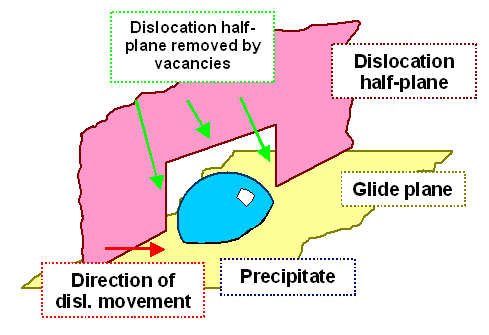 |
| Stuck dislocation freed by vacancies. |
|
| | |
|
 |
A number of vacancies has gnawed off sufficiently many atoms
from that part of the "half plane" of atoms that is stuck in front of a precipitate. The dislocation line now
is curved—and free to move on, elegantly bypassing the precipitate. |
|
 |
"Why, oh why, should vacancies do that?", you might ask with good reason. They are
not good samaritans that help stuck dislocations to get on just because they like to be nice to dislocations. No, they are
perfectly selfish, like the good people from the IAAA (or ADAC in Germany) that help you to unstick your car. It's good
for vacancies because they have an easier life sitting right in the dislocation line (their energy goes down a bit). The
car people have jobs if cars get stuck. |
|
 |
Let's look a bit more closely at that. Consider an area in a crystal that is under compressive
stress. In other words, the distance between the atoms is smaller than it should be. You could also say that the density
of atoms is too large, there are more atoms in a given volume than in stress-free regions.
Vacancies in some region
lower the density and thus release compressive stress somewhat since the distance between the atoms on average becomes a
bit larger. This simply implies that vacancies are attracted by regions with compressive stress since their presence lowers
the stress and thus the elastic energy. Since any dislocation always has areas of compressive stress along the dislocation
line, it always attracts vacancies. If it is piled up against a precipitate, the compressive stress there is even higher
- and that's why the vacancies go right into this part of the dislocation.
It's essentially the same reason why vacancies
also like to sit in grain boundaries or many other defects. It lowers the energy of the system: crystal - defects - vacancies
and thus brings it closer to nirvana. They are not permanently stuck, however. They also can get off again; it's a dynamic
process. |
 |
The long and short of this is that mobile vacancies will help dislocations to
"climb" around obstacles, and dislocation
climb leads to creep of the material. |
|
| |
|
Advanced Creep and Deformation Diagrams |
 |
You probably forgot that vacancies also help grain boundaries to move; I hardly remember that myself. If you consider a poly
crystal under some stress that is below the yield stress, slow grain boundary movement can also cause creep, the slow elongation
of your work piece. The principle is shown below |
|
| |
| |
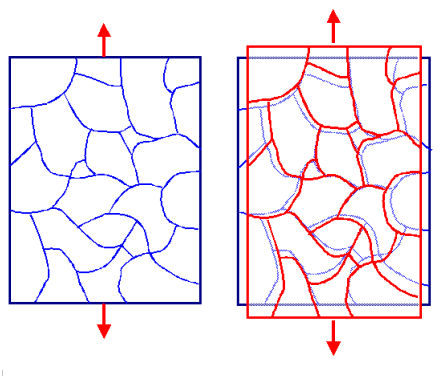 | | Grain boundary creep. |
|
| | |
|
|
 |
Left: Some poly crystal under tensile stress.
Right: Slow elongation because grain boundaries move. |
 |
The grain boundaries must, on average, move "inwards" to produce some
elongation. They move like this because they absorb and generate vacancies in the "right" way. Why
should they do this? |
|
 |
Because the compressive stress is in the left - right direction; this is the
direction where the distance between the atoms is a bit smaller then normal. In the direction of the applied tensile stress
it is a bit larger.
It thus makes sense once more to "work" with vacancies mostly in those parts of the grain
boundary that are under compressive stress, an this automatically leads to the preferential movement "inwards".
Note that neither grain boundaries nor vacancies "know" about this. It is simply a matter of probabilities. Creep
will result if it is just slightly more probable that vacancies get caught in the regions under compressive stress relative to other regions.
|
 |
Somewhat complex but powerful "deformation
diagrams" show it all. They were pioneered, I believe, by Michael
F. Ashby; I took it from his brilliant book
(with David R. H. Jones as co-author)). Here is a
simple one for pure nickel (Ni): |
| |
| |
| |
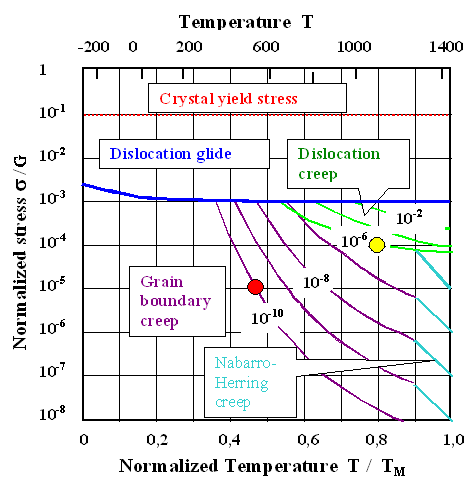 | | Deformation diagram for nickel (Ni). |
|
| |
|
 |
Shown is the kind of plastic deformation that takes part for some combination
of applied tensile stress and temperature. The axes are "normalized" and that makes this diagram—roughly—valid
for all metals not too different from nickel (most fcc metals). |
|
 |
The normalized stress is simply the applied stress
divided by the shear modulus
G of the material; is is
shown on a logarithmic scale.
That supplies a first (red) line; the theoretical maximum strength of all materials at
about 0.1 G, called crystal yield stress. That is the stress that would produce plastic deformation even without dislocations,
simply by sliding one part of the crystal relative to the rest all at once. It is not temperature dependence. |
|
 |
The normalized temperature is simply the actual
temperature T divided by the melting temperature TM. The regular temperature scale for nickel (Ni)
is given on top of the diagram. |
|
 |
Plastic deformation by regular dislocation movement or glide appears for all conditions above
the blue line that marks the yield stress. Note that the regular yield stress is about 100 times smaller then the crystal
yield stress, and decreases somewhat with increasing temperature. |
|
 |
The system of brown lines describes grain boundary creep. The
numbers refer to deformation speeds. 10–10 thus denotes de/dt = 10–10
s–1 or about 5 % deformation in 20 years. |
|
 |
The system of green lines describes dislocation creep, while
the blue lines indicate "Nabarro-Herring" creep, something I will not throw at you.
|
 |
Let's look at two examples of what can happen. |
|
 |
Conditions given by the red point: The applied stress
is about 100 times smaller then the yield stress; the temperature is almost half the melting point; somewhat above 500 oC
in the nickel case. Creep will occur by a grain boundary mechanism (always tied to vacancies, of course) at a rate of 10–10
s–1 or about 0.25 % per year. You, the modern engineer, must now decide if your construction can live with
that. |
|
 |
Conditions given by the yellow point: The applied stress
is about 10 times smaller then the yield stress; the temperature is very high. Creep will occur by grain boundary and dislocation
climb mechanisms (always tied to vacancies, of course) at a rate larger then 10–6 s–1
= 10–4 % / s » 0.3 % per hour. That is definitely too much for any decent
product. |
 |
Deformation diagrams like that are not only very useful for metals but also, for
example, for ice (frozen water) and olivine (major mineral in the earth's crust). What they tell you then is that the "movement"
of glaciers, the continents, the uplifting of mountain ranges like the alpes, and so on, is based on plastic deformation
by creep! |
| | |
|
© H. Föll (Iron, Steel and Swords script)





