|
|
|
Polishing and Structure Determination |
 |
Let's assume you have a quench-hardened pattern welded blade that still contains
some slag particles. You polish the blade until it is absolutely flat. Since there are few absolutes in technology, let's
say it is flat to about ±10 nm.
In cross-section this looks (schematically!) like this. |
| |
| |
| |
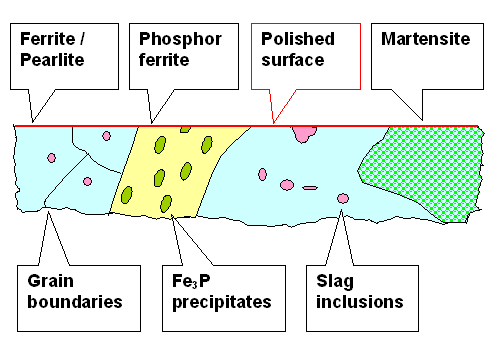 |
| A perfectly polished blade surface and what could be below |
|
| |
| |
|
 |
What do you see when you look at the polished surface? That is a rather difficult
question. The answer has three parts:
- Since different materials reflect the light differently, you "see" the intrinsic "reflectivity"
of the various materials, the reflectivity that is not influenced by surface roughness
or layers on the surface (we do not worry about the very thin oxide layers always there). If the reflectivity differences
between different materials are large enough, some materials will "look" brighter than others.
- You do not "see" small specks of material, even if their reflectivity
is different from that of the matrix. The eye isn't a microscope, after all. And even with a microscope you would see nothing
from spots that are much smaller than the wave length of the light, say 0.1 µm.
- You see yourself. After all, you are looking into a mirror
now. That's why the word "see" is in quotation marks above. Differences in reflectivity you "see" foremost
with an optical instrument. What you see with your eye depends very much on the kind of illumination employed and how you
look a the blade mirror.
We know that all metals reflect light very well. In other words: you can make mirrors from them. However, no metal
or material reflects 100 % of the light falling on it. The best you can get is about 97 %. Calculating the intrinsic reflectivity
of a given material, by the way, is a difficult task, and I'm not sure that it has ever been done for different grades of
steel. I have not seen reliable measurements either. I thus do not know if the reflectivity of a high-carbon steel is different
from that of low-carbon or phosphorous steel and if it is, how much. Nevertheless we may safely assume that there is a difference
- but not a large one.
That means that you might be able to see slight differences in brightness when you look sort
of sideways at a perfectly polished blade. You will not see the small
stuff, however: slag inclusions, precipitates or grain boundaries ending at the polished surface.
Many people in many
cultures at some times wanted their blades to be mirror-like with no discernible features. That is a tall job for the polisher.
There are two different ways of achieving "mirror" polishing and I
have detailed that before so I won't repeat it here. |
|
 |
In this context it is good to note that all
properly polished surfaces have mirror qualities - for example the quite black surface of a Steinway Grand Piano:
|
| |
|
| | |
 |
| Black mirror - a Steinway Grand Piano. Mizuka Kano playing in a barn during the Schleswig-Holstein
Musikfestival 2014 |
|
| |
| |
|
 |
The reflectivity of the black high-sheen lacquer is much smaller than that of
a metal but still high enough to produce nice mirror effects. So what can you tell about the uniformity of the surface of
the inside of the piano lid? Nothing! You can't see it. All you see is the reflection of the piano's inside. There is a
reason why you can order prints and other stuff "glossy" or "matte". |
 |
So perfect polishing by itself will not tell
you very much about the structure of your blade. "Japanese" polishing, however,
does just that. Look at Stefan Maeder's book or just
at the pictures here. While Japanese polishing does produce mirror-like surfaces
for parts of the blade, it also delineates at least parts of the structure of the blade. It's parts
of the structure because you can never be sure that it reveals everything there is. It won't tell you much, for example,
about phosphorous distribution. But it is quite powerful. An expert can appreciate the different grades of steel, can tell
if and how well faggoting was used, if there are (large) inclusions, and so on. |
|
 |
So how does Japanese
polishing work? Why does it reveal structures? Stefan Maeder writes a hell of a lot
pages on this subject without ever going into that "how" and "why" part. I don't know how Japanese polishing
works, to be sure, but I can make an educated guess.
Polishing typically means that you abrade (meaning scratch) the
object to be polished with small particles that are harder than the object. The hard
particles are contained in a slurry or embedded in a disc; read up in the polishing
module how it is done. If you want a "mirror-like" finish, your hard particles must be very small - sizes
below 1 µm are necessary. That is not so easy to come by and how to get or make the right "dust" and "stones"
are well guarded trade secrets. Note that King Theoderic's "Thank
You" letter for pattern welded swords mentioned "The metal your whetstone
so carefully shapes, this your splendid dust (granted to your country by the bounty
of nature) so thoroughly polishes that it makes the gleam of the iron a very mirror of men". Not to mention
Alexander the Great who needed iron for polishing diamonds.
What you get is still only a scratched surface but if the scratches are small enough to not be visible with normal
light.
The polishing action may not be exclusively due to mechanical abrasion (or "scratching" if you like),
however, but may contain a chemical component. Since freshly scratched surfaces are
very reactive, the damaged parts right after scratching might dissolve in the liquids always used with polishing. Even water
might do the trick to some extent. That provides for some additional smoothness and the polisher may not be aware of this.
If he is aware of this effect, he might develop the technique into what is called chemical-mechanical polishing, a technique used on a huge scale to provide perfectly polished (far better
than any sword) silicon wafers in large quantities.
The key word in all of the above is "harder".
Your polishing particles ought to be considerably harder than the material you are polishing if you want a surface as shown
schematically above. That's why diamond particles are so popular for this job.
What happens if this is not the case?
If, for example, you polish a blade with particles that are harder than ferrite or mild steel but not
as hard as martensite?
Right. Most of your blade will have a mirror finish but not
the hard martensite edge. And hard particles in the softer part of the blade might also get visible because they "stick
out" a bit and then become visible even if they are quite small - if you look at them the right way. The shadow of
a little bump at sunset ("glancing angle illumination") can be much larger then the bump itself and now you see
it.
Very schematically the "Japanese" polished surface might look like this: |
| |
| |
| |
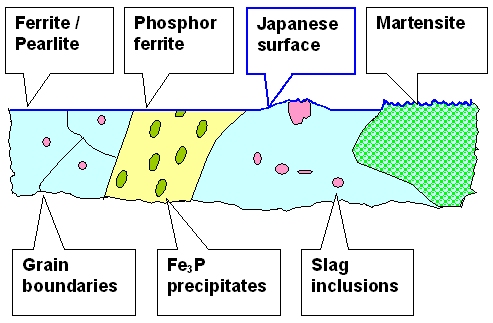 |
| A perfectly polished blade surface and what could be below |
|
| |
| |
| |
 |
The hard edge looks different now because it is rough. That would make it somewhat
darker than the bulk part because rough surfaces scatter the
light in all directions and thus less light finds its way into your eye.
Yes, I know that you now are going to
tell me that you have seen Japanese blades where the body looked darker then the edge.
No problem. You then looked at it under conditions where a lot of light came from one
direction (the sun, for example). Then the well-polished blade reflects most of that light in a specific direction and if
that is not the direction to your eye, you won't see much - the blade appears dark. If you look at the blade with "diffuse"
light coming from all directions (cloudy day; sun not visible), things look quite different.
On the other hand, it is
easy to conceive of a way where the surface looks exactly the other way around relative to the picture above: the soft part
is a bit rough an the hard part is perfectly polished. First polish everything to perfection with very hard slurry particle,
then polish again with relatively soft and largish particles. This won't do much to the hard part but you are now roughening
the soft part. |
 |
This, I believe, is the basic principle behind Japanese polishing. And now you
can start to optimize. Use particles of different hardness in the various steps, use different liquids, .....
Since
Japanese polishing was optimized to reveal the structure of Japanese blades while still providing an aesthetically pleasing
"polished" look, it is certainly a good method for analyzing blades. Stefan Maeder has proved this for early medieval
European swords as shown here, and more recently for a Celtic sword from 200
BC - 300 BC. Here is what he blade looked like after "Japenese polishing" by Maeder:
|
|
 |
We need to be clear about one thing in this context. While "Japanese polishing"
is certainly a very good method for finishing blades in a way that looks good and allows
structural characterization, it is not necessarily "THE BEST" as claimed by Maeder and others. Western (modern)
polishing plus an optimized defect etching might just be as
good or better. As long as nobody makes a direct comparison we simply can't tell. |
| |
| |
| |
Producing the Pattern |
 |
Let's assume you have a pattern welded blade or some blade made by piling various
grades of steel that still contain some slag particles. You polish the blade until it is perfectly flat and mirror-like.
Under the right illumination an looking conditions you now can see the pattern. But only faintly
because the reflectivity of the two kinds of steel used is (most likely) only a little bit different |
|
 |
This is not what you want. You want to have a strong pattern, always visible,
kind of black-and-white. So what you do is to etch the (polished) blade. The German word for this process is "ausziehen" in the sense of "extracting". What you may want should look like this: |
| |
| |
| |
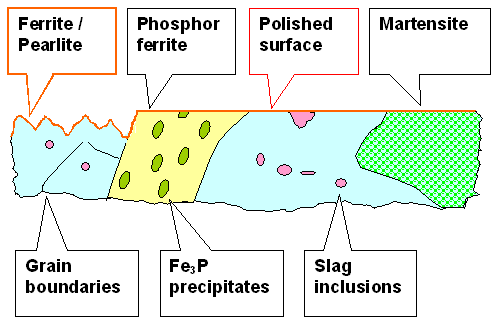 | | Etching for pattern delineation |
|
| |
| |
|
 |
I have chosen to leave the phosphorous iron more or less intact and to only roughen
the ferrite. That is what usually happens if you use mild acids like fruit juices for the etching. You need to dissolve
some of the steel and that means you are actually corroding it. Since phosphorous iron, for rather tricky reasons, is more corrosion resistant than normal iron, changes are that you get what is shown
above. |
 |
We need to be clear - once more - that pattern etching like defect etching is
still counted among the black arts. It is almost
impossible to predict what a given etchant will produce, and it is even impossibler (!) to device a recipe for concocting
a good etchant from first principles. On the other hand, a lot of recipes just work. So doing the etching is simplicity
in itself. |
|
 |
Another way to produce optical contrast is to grow a layer on one of the steel
kinds. Without tricky chemistry that tends to be an oxide layer (best is magnetite, Fe3O4) and if
you succeed in doing so, your iron / steel will become dark or even acquires a kind of color. It will turn bluish for example.
That's why the process is called "blueing" or "browning".
I'm not saying that the
ancient craftsmen who made pattern welded swords used one of the many blueing / browning processes known today. All I'm
saying is that immersion in some corrosive liquids might produce stable oxide layers, in particular if you follow up with
some stabilizing treatment like boiling in water.
Since we have no ancient pattern welded swords with an intact surface,
we cannot presently know how our forebears produced the pattern. The best I can come up with is this picture from Joachim Kinder: |
| | |
|
| | |
 |
| Appearance of a well preserved pattern welded blade at low magnification |
|
| |
| |
|
 |
The blue parts is phosphorous iron. If it reflects an original blueing by the
sword maker is open to doubt. It is not impossible either, however. It is a tantalizing thought that pattern welded swords
might have been colored. King Theoderic's "Thank You"
letter mentions pattern welded swords as "the metal was ... shining with different colors". |
| |
| |
© H. Föll (Iron, Steel and Swords script)




