| |
Science of Alloying |
|
What Can be Done |
 |
What, exactly, do you expect from the "science"
of alloying? Well - it should at least be able to answer all questions coming
up in this context. If you think about for this a bit you realize that the issue boils down to two basic question: |
|
 |
1. How do the properties of my system change if I add an amount x of element y
and process with recipe z.
2. How do I have to compose a system and find the right processing to get the properties I
want?
|
 |
The first question asks for analysis, the
second one for synthesis. The second one is much harder to answer then the first one.
Let's look at some examples. |
|
 |
Example for the first question:
My first example concerns you, the ancient smith.
Your system may be "ideal" plain carbon steel, say iron with 0.4 % carbon (C). Your process is to forge it around
900 oC (1652 oF) into the desired shape. Then you quench it in water, followed by 20
minutes tempering around 350 oC (662oF). You get some tempered steel this way.
Now you wonder how properties like hardness and so on change when you add 0.2 %
phosphorus or 0.15 % sulfur (S). That's a rather good question of the type 1 that you, the ancient smith, could have asked
because it relates very much to what you were doing then. You probably would have been satisfied if someone would have told
you what would happen with respect to just a few properties like hardness and cold /
hot shortness.
Of course you, the modern reader of this hyperscript and fully conscientious of the claim I made early on, want to know more. You want to know what
would happen with respect to a lot of properties and in particular why
that happens.
Can I answer these questions? No, I can't. I'm not a steel expert,
remember? I'm fairly confident, however, that there are scientists out there who could tell you what is going to happen
because they can actually calculate it. They don't have to know from experience or try it experimentally. They can calculate
it because the theory is there. Maybe the computer power isn't quite up to the job right now but it will be there in the
fullness of time.
The question has a definite answer, and we are going to produce it - now or later. |
|
 |
Example for the second question:
My second example
concerns a modern engineer. Working for Audi, she might ask what kind of aluminum (Al) based composition would keep all
the good properties of the aluminium alloy she is using right now for the body of the more expensive Audi cars, but would
add easy weldability to the list of desirable properties. Her present aluminum alloy
can't be welded, causing major cost issues.
A definite answer to that question would be the death knell for that part
of the steel industry that makes steel for car bodies. So her colleague over at the steel plant might ask a question along
a similar line: what composition (plus process recipe) would make a better steel that could compete against the new aluminum
alloy.
Both engineers supply a long list of detailed properties they need,
plus some requirements as to costs to you, the scientist in charge. |
 |
Can you or anybody else give them a definite
answer? No you can't. There are no easy positive answers to their questions. But maybe
the answer will be "Can't be done, ever!" Let's see why. |
|
 |
It is rather simple to come up with a wish list of properties, where the scientist answers
right away: "can't be done". For example, for rather well-known reasons, a single material from which you can
make a solar cell with an efficiency of 50 % does not exist. Never ever. Theory is clear on this.
But how about a material
with 18 % efficiency at a cost of 2 $ per kilogram? This is a question that no scientist
can really answer because money and costs are not part of science. There is no way to compute scientifically,
for example, what the cost of tellurium (Te), will be in 15 years. That is big topic in solar cell business, by the way.
Maybe it is possible (theory doesn't forbid it), maybe it's not. No way of telling. At best you can give probabilities.
|
|
 |
It is one thing to calculate the properties of something from first principle. It is perfectly
possible for example to calculate what kind of X-ray diffraction pattern would result from a given arrangement of the building
blocks of DNA. Given the structure, you can calculate the diffraction pattern.
It's a completely different thing to
calculate what you have to assemble to obtain a certain property. The reverse of the example above, calculating the structure
from a given diffraction pattern, is more or less impossible. In strict math terms: it is always possible to calculate the
value of a definite integral for some function, even if it is a "difficult" one. The result is a number. It is
impossible, however, to calculate backwards and derive this function from a given number. The reason is simply that an infinitely
large number of functions, if integrated, could give the same number.
Questions of the second type fall into this category
to some extent. Sorry. But see the positive side of that: Materials Science will never come to an end. Computers somewhere
and sometime will be able to give answer to all your questions of the first type but not to questions of the second type.
Materials scientists, however, will find answers on occasion because they can do more than just pure math. |
 |
The long and short of this is simple. If you supply a list of desirable properties
for the steel you want, the answer might be that it can't be done. But even if your list does not include the impossible,
nobody will be able to sit down and calculate the necessary composition and the processing that will do the trick.
Now that we know what we can't do, let's see what is possible |
|
| |
| |
Classification
by Influence on the Iron-Carbon Phase Diagram |
 |
The science of iron alloying must start with
binary phase diagrams for all sensible binary iron
- element x compositions. Non-sensible compositions
for example, are iron - noble gases for all concentrations and temperatures or the x-rich end of the iron - element x phase diagram. |
|
 |
The good news is that we can actually calculate
all those phase diagrams pretty well and in the not too distant future we will be able to do this really well (partially
due to the efforts of your's truly and colleagues with silicon and microelectronics, giving us computers with ever increasing
power).
Now that we know what kind of phases we are going to encounter with binary alloys (e.g. Fe3P, Fe2P
for iron - phosphorus systems; Fe2N for iron - nitrogen), we need to know about moving atoms around in those phases.
How often does an atom /vacancy jump per second? That's harder to calculate - but we will get there. |
 |
But let's not get carried away. Even if we can do all there is to do for the binary
system iron with just one kind of alloying element, we will not get very far with complex systems, e.g. iron with 5 or more
alloying elements if we try to calculate everything possible from first principles. There is simply too much that could
happen. Most of what could happen will not happen, but you have to do very precise math on occasion just to figure that
out. It is far easier to figure it out some other way, even if that involves some "intelligent" guessing plus
experience. Rules then can be derived that may not always be 100 % correct but at least most of the time. One such set of
rules comes from the insight that the phase diagrams of most alloy elements and iron fall into just 2 major categories with
two sub-clauses each.
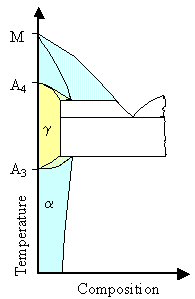
Keep in mind for all comparisons that for very small concentrations you have almost pure iron
with its two phase changes from an a
bcc structure to a g
fcc structure at 912 oC (1.674 oF) and back again
to d bcc at 1394 oC (2541 oF ). We call these temperatures,
or better the lines starting at this points, A3 and A4 (the melting temperature here
is "M"). |
| |
| |
| | |
Any binary phase diagram must converge
to these points for small concentrations
of the alloying element.
|
|
| | |
|
| |
|
There are two major ways to do that, and two sub-groups for each basic possibiltiy.
The two major ways are.
- The fcc phase gets more prominent. In other words, you might find the fcc phase (then called g
phase or austenitic steel) within a larger range of temperatures than for pure iron.
The alloy element is a g-stabilizer.
- The fcc phase gets less prominent. In other words, the bcc phase (then called a phase or ferritic steel) then must be more prominent. The alloy element is an a-stabilizer
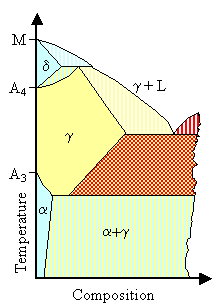
Now let's look at the subgroups of the g-stabilizers
The the figure right below illustrates this schematically. |
|
 |
- Expanded g-phase systems. The alloy
element expands the temperature range for stable austenite compared to pure iron. There is a sizeable g-phase
region but it is still limited from all sides because compounds like Fe3C
can form
- Open g-field systems. The g phase is expanded and has no boundary in a meaningful range of compositions. Alloy elements doing
that encourage the formation of austenite in wide compositional and temperature regions. This could even mean that the g-phase austenite could be stable at room temperature.
|
| | |
|
|
|
 |
 |
| Expanded but limited g-field systems |
Expanded and Open g-field systems |
Adopted from "Key to Metals", The Effects of Alloying Elements on Iron-Carbon Alloys
This way of thinking goes
way back to "F. Wever, Archiv, Eisenhüttenwesen, 1928-9, 2, 193" |
|
| |
|
 |
Let's look at some examples: |
|
 |
Class 1: Expanded g-field
systems.
That's the kind of phase diagram we know because
carbon (C) and nitrogen (N) are the most important elements
in this group. The g-phase field is expanded, but its range of existence is cut short
by compound formation.
The expansion of the g-field by carbon (or nitrogen) is at
the core of heat treatments of steels since it allows the formation of a homogeneous solid solution (austenite) containing
up to 2.0 wt % of carbon (or 2.8 wt % of nitrogen).
Copper (Cu), zinc (Zn) and gold (Au) have a similar influence.
| |
Class 2: Open g-field systems
To this group belong the important steel alloying elements nickel (Ni) and manganese (Mn), as well as cobalt
(Co) and the inert (noble) metals ruthenium (Ru), rhodium (Rh), palladium (Pd), osmium (Os), iridium (Ir) and platinum (Pt).
These alloy elements are also known as g stabilizers.
Both nickel and manganese, if added in sufficiently high concentration, completely eliminate the bcc -iron phase and replace
it, down to room temperature, with the g-phase. So nickel and manganese depress the phase
transformation from g to a to lower temperatures. It
is thus much easier to obtain metastable austenite by quenching from the g-region to room temperature,
consequently nickel and manganese are useful elements in the formulation of austenitic steels. |
 |
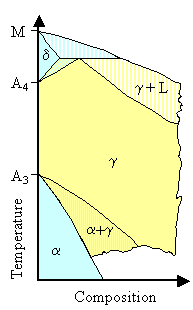
The paradigmatic g stabilizers producing open
g-field systems are nickel (Ni) and manganese (Mn). Their phase diagrams with iron are practically identical for the iron-rich side: |
| |
|
|
|
 |
| Phase diagram iron - nickel or iron - manganese |
|
| | |
|
|
|
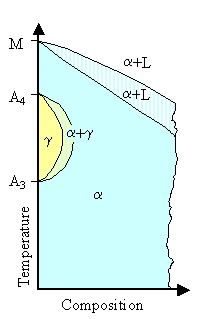
Next we have the two subgroups of the a-stabilizers: |
| |
- Closed g-field systems. The opposite
of Class 2 from above. The g region becomes rather small and is completely contained in
an a field.
- Contracted g-field systems. The
alloy elements encourage the presence of the a ferrite phase in larger regions but the
g is not completely contained in an a field but linked
to compound phases of the alloy elements with iron.
|
|
| |
| |
 |
 |
| Closed g-field systems |
Contracted g-field systems |
| Adopted from "Key to Metals", The Effects of Alloying Elements on Iron-Carbon Alloys |
|
| |
|
|
 |
Class 3: Closed g-field systems
Many elements restrict the formation of g-iron, causing the g-area
of the diagram to contract to a small area referred to as the "gamma loop". This
means that the relevant elements are encouraging the formation of the a phase or bcc iron
(ferrite). One result is that the d and g-phase fields
merge and become continuous, completely enveloping the g region.
Closed g-field systems are therefore not amenable to the normal heat treatments involving cooling through
the g/a-phase. transformation.
Silicon (Si), aluminium
(Al), beryllium (Be) and especially phosphorus (P) fall into this category (phase diagram
here), together with the strong carbide forming
elements like titanium (Ti), vanadium (V), molybdenum (Mo) and chromium (Cr). |
|
Class 4: Contracted g-field systems.
Boron (B) is the
most significant element of this group, together with the carbide forming elements tantalum (Ta), niobium (Nb) and zirconium
(Zr).
The g-loop is strongly contracted, but is accompanied by compound formation
(the white unspecified fields). |
 |
Now for a few rules and examples. |
|
 |
Although only the binary systems have been
considered so far, we can predict to some extent what will happen if we now add carbon. We're looking now at a ternary system where the same general principles usually apply. For a fixed carbon content, as
the alloying clement is added the g-field is either expanded or contracted depending on
the particular solute. Some examples:
- With an element such as silicon (Si) the g-field is contracted and there is a corresponding
enlargement of the a-field.
- If vanadium (V) is added, the g-field is contracted and there will be vanadium carbide
in equilibrium with a ferrite over much of the ferrite field.
- Nickel (Ni) does not form a carbide and expands the g-field.
|
|
 |
Normally, elements with opposing tendencies will cancel each other at appropriate
concentrations. In some cases, however, anomalies occur. For example, chromium (Cr)
added to nickel (Ni) in a steel in concentrations around 18% helps to stabilize the
g-phase, even so it is listed under the "closed g
phase elements". This "Ni-Cr anomaly" is used in the class of (18% Cr 8 % Ni) austenitic steels.
|
 |
This is all pre-war stuff. Even then it was clear in principle how one could go
beyond qualitative rules and get quantitative results. The only problem was that doing the necessary math was impossible.
Present-day computer-based methods overcome these difficulties and the science of alloying is becoming a true quantitative
science, able to make precise predictions. |
| |
| |
| |
Classification
of alloying Elements by "Stabilities" |
 |
Here we look on the effect of an element on the stability of the important phases
in steels containing some carbon. This involves in particular: This gives us four major groups: |
|
 |
1.
Elements which tend to form carbides. In other words, elements that produce stable carbides
rather than compounds with iron. Of course, this implies that there is some carbon around.
We have essentially chromium
(Cr), tungsten (W), titanium (Ti), niobium (Nb; sometimes also called columbium), vanadium (V),
molybdenum (Mo) and manganese (Mn) as carbide formers. The mixture of the complex carbides
formed whenever those elements are present is often - wrongly - referred to as "cementite". The reason is that
the carbides may contain both metals - iron and the alloying element - e.g. (FeCr)3C, (CrFe)3C2
(CrFe)7C3
or (CrFe)4C, to name some iron-chromium carbides.
If we have several metals in the mix, there is a
competition for which one gets the carbon. |
|
 |
2.
Elements which tend to graphitise the carbide.
We have Silicon (Si), cobalt (Co), aluminium (Al) and nickel (Ni). Only a small proportion of these elements can be
added to the steel before graphite forms during processing, with attendant ruin of the properties of the steel, unless elements
from group 1 are added to counteract the effect. |
|
 |
3.
Elements which tend to stabilize austenite.
Prominent are manganese (Mn), nickel
(Ni), cobalt (Co) and copper (Cu). These elements alter the critical points of iron in a similar way to carbon by raising
the A4 point and lowering the A3 point, thus increasing the range in which austenite is stable,
see above. This will lead to austenitic steels in
the end. They also tend to retard the precipitation of carbides.
Most of these elements have a fcc
crystal lattice like g-iron, and thus tend to be more soluble than in g-austenite
than in a-ferrite. Sitting as a substitutional foreign atom in austenite at least preserves
the kind of environment the atom is accustomed to. |
|
 |
4.
Elements which tend to stabilize ferrite.
Chromium (Cr), tungsten (W), molybdenum
(Mo), vanadium (V) and silicon (Si) belong to this group. Most have a bcc
lattice and thus tend to be more soluble in a-ferrite than in g-austenite.
They diminish the amount of carbon dissolved in the austenite (because there is now less austenite) and thus tend to
increase the volume of carbides in the steel for a given carbon content. They lower the A4 point and raise A3
(although it may be lowered initially), until the two points merge to form a “closed gamma loop”. Above a certain amount of each of these elements the austenite phase then disappears
and ferrite exists from the melting point down to room temperature.
No phase transformation occurs and such steels (e.g.
18% chromium irons) thus cannot be hardened in the usual way. You harden by adding austenite stabilizers from group 3 above.
For example, 2 % of nickel added to a 18 % chromium stainless steel allows "refinement" by normal heat-treatment;
carbon has the same effect. |
 |
You realized, I assume, that in parts the information given is the same as in
the first system, just expressed in different words. |
| |
| |
| |
A few Illustrations, Examples and Graphs |
 |
Let's look at few illustrations now and at an ingenious way to deal with multicomponent
steels without resorting to (typically non-existent and otherwise completely unpractical) multi-dimensional phase diagrams. |
|
 |
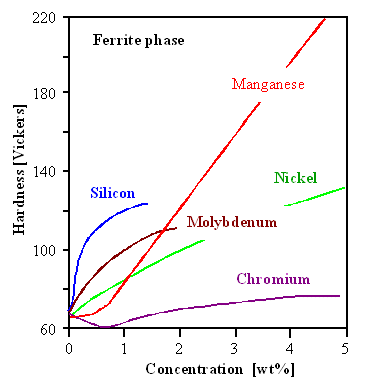
Here is a figure that should be familiar.
It gives the hardness of a steel alloyed with the elements shown. It is assumed that the elements are atomically dissolved,
so we have solid solution hardening. In the link a similar
picture was shown; this one just gives a bit more information. |
|
| |
| |
|
| | |
|
|
 |
In all cases the transformation from austenite to ferrite takes place; the measurements were
done in the ferrite phase at room temperature. Obviously you want to go for manganese (Mn) if hardness is all that counts.
A picture like this, based on extensive experiments, often tells you more than what is visible "at the surface".
For example, the silicon (Si) and molybdenum (Mo) lines end between 1 % and 2 %. That simply tells you that it is not possible
to keep larger concentrations in solid solution. It also tells you that for all those elements and concentrations, you still
have the g ®
a phase transformation. Noticing that, natural questions are coming up:
- At what temperature will we find the g ®
a phase transformation for some alloying element?
We cannot expect that it will be the old
(A1) value of 723 oC (1.333 oF) we know from the plain iron-carbon system.
- At what carbon concentration will we find the eutectoid point of the g
® a
phase transformation?
There is no reason that it should remain at 0.83 % carbon, after all.
|
|
 |
Some answers to these questions are given in the next figure. It shows how
the temperature A1 (upper figure) and the
composition (lower figure) of the eutectoid point changes if various elements are alloyed to a carbon steel. |
| | |
|
|
|
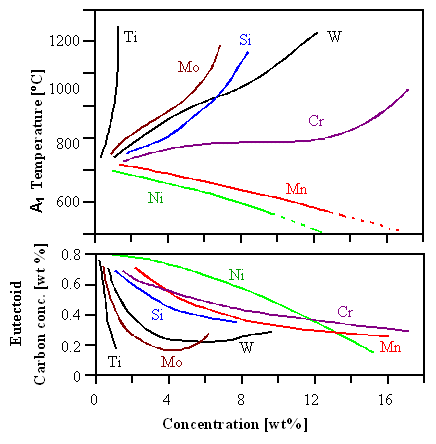 |
| Change in A1 and eutectoid composition by alloying |
| Source: Key to metals |
|
| | |
|
|
 |
To give examples on how to read these curves: Alloying 4 % molybdenum (Mo) increases A1
from its "normal" carbon-steel value of 723 oC (1.333 oF) to almost 900 oC (1.670oF),
and decreases the eutectoid composition from 0.8 % carbon to about 0.2 %. Adding 4 % of manganese brings A1
down to about 700 oC (1.290oF) and lowers the eutectoid composition to about 0.6 %. |
|
 |
There is a lot of information in these curves. Titanium and others (Mo, Si, W) seem to stabilize
the ferrite since the transition to austenite occurs at higher temperatures if these elements are present.
Manganese
(Mn) and nickel (Ni) stabilize the the austenite.
That's exactly what has been stated above - now we have data. There is much more information in the details of the curves
but I will stop at this point. |
 |
Now let's look at a rather ingenious (if approximate) way to deal with steel that
contains many different alloy elements. Anton
L. Schäffler published something like the following diagram in Nov. 1949; W.T. De-Long improved on it in 1974; that's why it is sometime called the Schäffler - De Long diagram. There are two essential insights:
- It is possible to lump all those alloy elements together in groups that either stabilize austenite (like nickel (Ni)),
or ferrite (like chromium (Cr)), respectively. Just add up their weighted percentages
as shown in the diagram. The sum gives you a so-called Ni-equivalent or a Cr-equivalent
concentration, respectively.
- In a Ni-equivalent vs. Cr-equivalent plot, the various phases and phase mixtures can be indicated as shown below (always
assuming some reasonable cooling down process, of course).
|
| | |
| |
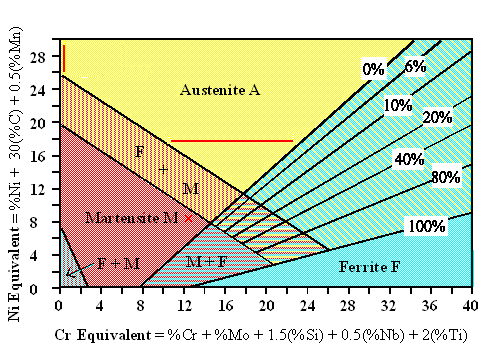 |
| Schäffler - De Long diagram; a rather good guide for high-alloy steels. |
|
| | |
|
|
 |
Three kinds of steel are drawn in either as as red cross or red lines. The percentages at
the black lines indicate the level of ferrite formation.
Let's look at the examples:
- Red cross: A 12% Cr steel containing 0,3% C is martensitic since the 0,3% C gives the steel
a nickel equivalent of 0,3 · 30 = 9.
- Horizontal red line: An 18/8 steel (18% Cr, 8% Ni) is austenitic if it contains 0-0,5%
C and 2% Mn, giving a Cr-equivalent of 9 - 24. Increasing the Cr equivalent brings in an increasing percentage of ferrite.
- Vertical red line: Good old
Hadfield steel with 13% Mn is austenitic due to its
high carbon content of about 1.2 %. Should this be reduced to below about 0,20 %, the steel becomes martensitic.
A lot of high-alloy steels are designed with the help of the Schäffler diagram; this
link gives examples. |
| |
 |
There are other diagrams like the Schaeffler diagram; they all are helpful but must be treated
with a bit of caution. For example, the austenite-stabilizing effect of manganese is nowadays considered to be smaller than
that predicted in the Schaeffler-Delong diagram. Its effect is also dependent on temperature. |
 |
It's time for a few more insights and rules. If we restrict ourselves to steels
with some carbon and process in such a way that the austenite transforms to ferrite and carbide (i.e. slow
cooling), so we leave enough time for alloying atoms to move around, the alloying elements can be divided into three categories:
- Elements which dissolve only in the ferrite phase.
- Elements which form stable carbides and also dissolve in the ferrite phase.
- Elements which form only carbides.
|
|
 |
In the first category there are elements
such as nickel (Ni), copper (Cu), phosphorus (P), and silicon (Si). These elements are normally found in solid solution
in the ferrite phase, their solubility in cementite or in alloy carbides being quite low. |
|
 |
The majority of alloying elements used in steels, however, falls into the second
category. Those elements are essentially carbide formers. At low concentrations they form solid solutions in ferrite but
also go into solid solution in cementite. At higher concentrations most
will form alloy carbides, which are thermodynamically more stable than cementite. Typical examples are manganese (Mn), chromium
(Cr), molybdenum (Mo), vanadium (V).
The carbide-forming elements are usually present in concentration far in excess
of carbon. When the available carbon is used up for carbide formation, the remainder enters into solid solution in the ferrite,
together with the non-carbide forming elements like nickel (Ni) and silicon (Si), if those are around. Some of these elements,
notably titanium (Ti) , tungsten (W), and molybdenum (Mo), then produce substantial solid solution hardening of ferrite,
see above. |
|
 |
The third category contains only a few elements, which
enter predominantly the carbide phase. Nitrogen (N) is the most important element and
it forms carbo-nitrides with iron and many alloying elements. However, in the presence of certain very strong nitride forming
elements, e.g. titanium (Ti) and aluminum (Al), separate alloy nitride phases can occur. |
 |
Finally, a few words to the structures one can expect: |
|
 |
Adding elements like manganese (Mn), silicon (Si) or nickel (Ni) that do not
form carbides (or if they do cannot win against the competition of cementite formation), does not basically alter the microstructure
of plain-carbon steel as formed after transformation.
In the case of strong
carbide-forming elements such as molybdenum (Mo), chromium (Cr) and tungsten
(W), cementite will be replaced by the appropriate alloy carbides, often at relatively low alloying element concentrations.
The microstructure is thus quite different from that of plain carbon steel.
Very strong carbide forming elements,
such as niobium (Nb), titanium (Ti) and vanadium (V), always form alloy carbides, preferentially at alloying concentrations
less than 0.1 %. The microstructures of steels containing these elements will be radically altered.
|
|
 |
Cementite is the only carbide in plain carbon steel. The
wealth of structures found in plain carbon steel results exclusively from the high solubility of carbon in the g-austenite
and its low solubility on the a-ferrite. If we now have many
possible carbides to play with, we first need to know if they form already in the austenite or later in the ferrite.
What was found more or less empirically but is more and more based on calculations, is:
- Niobium (Nb) and titanium (Ti) will form excess alloy carbides which cannot be dissolved in austenite at the highest
solution temperatures if their concentrations exceeds about 0.25 %.
- With vanadium (V) the limit is higher, around 1% - 2 %, and with molybdenum (Mo) it goes up to about 5%.
- Chromium (Cr) has an even higher limit.
|
| |
| |
| |
An Application to "Wootz" Steel? |
 |
I could go on like this for quite a bit but will stop now, except for a last remark
that may explain some of the mystery about wootz steel.
The question
we ask ourselves is: "Where do all those metal carbides nucleate when they first
start to form?". The enticing quote from the "key to metals" article cited above
is: |
|
 |
"Interphase precipitation has been shown to nucleate periodically at
the g/ a interface during the transformation. The precipitate particles
form in bands which are closely parallel to the interface, and which follow the general direction of the interface even
when it changes direction sharply." |
 |
Now consider a slowly solidifying piece of wootz steel in its crucible. It is
an hypereutectoid steel with an extremely high carbon concentration around 2 %, and it contains a bit of strong carbide
formers like vanadium (V). When it cools down and crystallizes, the g / a
interphase is somewhere between the still liquid part and the final pearlite
/ cementite mixture. If the geometry is as shown below, you might get carbide nuclei arranged in sheets as shown. |
| |
|
|
|
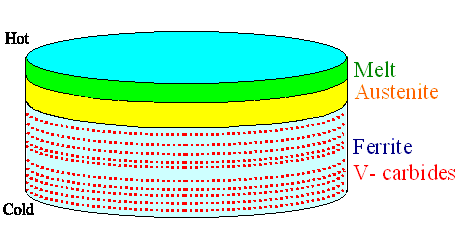 | Idealized and schematic geometry of striated carbide
particle formation in a wootz cake while crystallizing |
|
| |
|
|
 |
So we might find Vanadium-carbide particles in "bands", i.e. in a striated manner, more or less parallel to the surface of the final wootz ingot.
This is schematically illustrated above. These banded or striated tiny metal-carbide particles will supply the nuclei for forming cementite particles later.
If the smith is very careful
in preserving the striated structure in the blade he forges. he can obtain the characteristic "water" pattern
of a wootz sword, essentially showing a cut through the still striated but "wobbly" metal carbide position now
expressed in "large" cementite precipitates. This is outlined in detail in chapter
11.5
Far more could be said to this (especially to the "why" questions coming up now), and I have actually said something rather speculative to the issue. The geometry
shown is rather unlikely to occur without special measures. |
| |
| |
 With frame
With frame

 General Remarks to Literature and Sources
General Remarks to Literature and Sources
 Steel Guys
Steel Guys
 Science of Welding Steel
Science of Welding Steel
 Nucleation Science
Nucleation Science
 Overview of Major Steels
Overview of Major Steels
 9.1.1 Things are Complicated
9.1.1 Things are Complicated
 Phosphorous Steel; 9.4.1 General Remarks
Phosphorous Steel; 9.4.1 General Remarks
 9.2.2 Designing Low Alloy Steels
9.2.2 Designing Low Alloy Steels
 Alloying Elements in Detail
Alloying Elements in Detail
 9.5.2 Kinds of Cast Iron
9.5.2 Kinds of Cast Iron
 Jominy Test and Hardness Depth
Jominy Test and Hardness Depth
 Inhomogeneous Deformation
Inhomogeneous Deformation
 Smelting Science - 1. Furnaces
Smelting Science - 1. Furnaces
 Overview of Major Steels: Scientific Steels
Overview of Major Steels: Scientific Steels
 11.5 Wootz Swords; 11.5.1 The Winner is....
11.5 Wootz Swords; 11.5.1 The Winner is....
 11.5.2 Structure by Dendrites?
11.5.2 Structure by Dendrites?
 Maraging Steel
Maraging Steel
 Segregation at Room Temperature
Segregation at Room Temperature
 8.2.2 Tempering and Ostwald Ripening
8.2.2 Tempering and Ostwald Ripening
 Overview of Major Steels: 1. Classifying Steels
Overview of Major Steels: 1. Classifying Steels
 9.2.1 A Closer Look at Low Alloy Steels
9.2.1 A Closer Look at Low Alloy Steels
 Overview of Major Steels
Overview of Major Steels
 High Alloy steels; 9.3.1 Stainless Steel
High Alloy steels; 9.3.1 Stainless Steel
 Steel Properties
Steel Properties
 The Iron Carbon Phase Diagram
The Iron Carbon Phase Diagram
 Segregation at Room Temperature
Segregation at Room Temperature
 High Alloy Steels and Schäffler Diagram
High Alloy Steels and Schäffler Diagram
 Microsegregation and "Current Burst" Theory
Microsegregation and "Current Burst" Theory
 Alloying Elements and Properties of Steel
Alloying Elements and Properties of Steel
 ChicagoTribune Article
ChicagoTribune Article
 Phase Diagram of Fe-P and Fe-N
Phase Diagram of Fe-P and Fe-N
 Ghost Structures in Phosphorous Steel
Ghost Structures in Phosphorous Steel
 Imaginative Names
Imaginative Names
© H. Föll (Iron, Steel and Swords script)