|
Twinning, Shear Deformation and Martensite Formation |
| |
What it is all about |
 |
This was the claim: |
| |
| |
| |
|
| |
| |
|
 |
That is a clear and unambiguous statement. But is it true? "What is truth?" is a
fitting quote in this context. It depends how you look at things, as Pilatus knew. It's not a life-and-death thing here
but good enough for heated discussions. Let's see why. |
 |
Let's start with the experimental observations. Sometimes crystals under stress
deform by what is called twinning on just a few planes, and that looks quite different from the usual mechanisms where dislocations provide for glide on many glide planes.
Some pertinent observations in this respect are:
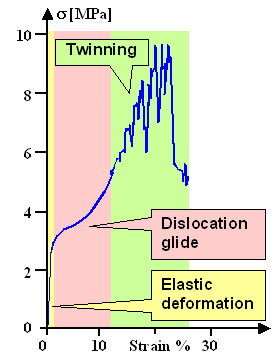
- Twinning happens very quickly. A specimen deforming by twinning often elongates in leaps and bounds, emitting audible
"shrieks". Bend a piece of zinc or tin and you hear it. The stress-strain curves then are serrated or unruly (see
below).
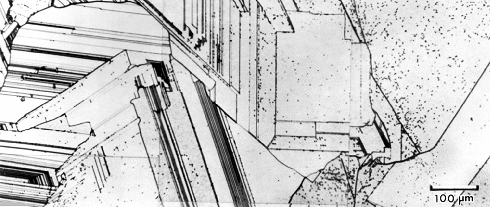
- Twinning leaves typical structures easily recognized. Typically, you see a lot of straight and parallel lines inside
grains (see below or here).
- Deformation by twinning is more likely to occur at low temperatures.
- Deformation by twinning is more likely to occur at high strain rates.
- Deformation by twinning is more likely to occur in crystals with not so many glide
systems. Crystals with fcc, bcc or hex lattices have 12, 12, or 3 glide systems, respectively, and that means that hexagonal crystals like Cd (cadmium), Co (cobalt), Mg (magnesium)
Ti (titanium), Zn (Zinc) or Zr (zirconium) will be more given to twinning than their bcc and fcc brethren.
|
|
|
 |
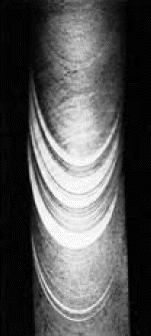 |
Stress-strain diagram for cadmium (Cd) with twinning, and deformed zinc (Zn) crystal showing
very inhomogeneous deformation
including some twinning
This is schematic, the numbers are only approximate.
|
| Source: Textbook stuff, e.g. Meyers and Chawla, and Net at large |
|
| |
| |
 |
So what is twinning? Before I get to this, look at the following series of pictures
. They show twinning in silicon. |
| |
| |
| |
|
| |
| |
|
 |
The straight lines show "twins", while the curved lines denote regular grain boundaries.
Dislocations ending at the surface give rise to the black dots.
As you know, regular grain boundaries form during solidification
when two independently nucleated grains grow and finally meet. Twin boundaries, referring to a very special orientation relationship between the grains, could have formed by chance during solidification
- but this is unlikely. Most of the twins observed above must have been formed by a twinning deformation, induced via the
stress developed during cooling. |
| |
| |
| |
|
| |
| |
|
 |
The bands (some of them striped) show twin boundaries. The picture below shows that "one"
black line of the picture above consists of several twins: |
| | |
|
|
|
 |
| Small part of sample from above in the electron microscope at atomic resolution (HRTEM) |
|
| |
| |
|
 |
Now you see why it is called "twin" boundary. The region between two twin boundaries
is the "twin". Note that the series of 4 twin boundaries / 3 "twins" is just about 20 nm wide and does
not change the orientation of the crystal from the left to part to the right part. You wouldn't see it by light microscopy. |
| |
| |
| |
Deformation by Glide and by Twinning |
 |
Let's look in a simple but systematic way on how you can deform a crystal. I use
an fcc crystal for illustrating this in a <110> projection, i.e. we are looking down the elementary cube as shown
in the figures.
Here is the crystal we start with. It shows nicely the simple recipe for making
fcc crystals: Put a first layer of spheres down, (A-layer). Put a B-layer and then a C-layer on top. Repeat. |
| | |
|
| | |
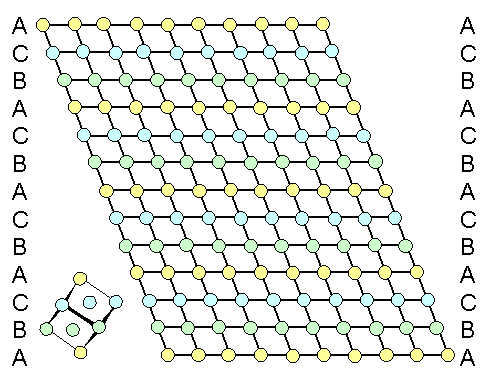 |
| A perfect fcc element crystal projected along a <110> direction. |
|
| |
| |
| |
 |
The inset shows how the elementary cube is oriented. The colored circles symbolize
atoms, all of the same kind but on different planes. The black lines are meaningless and only there to guide the eye. |
 |
Now we put some shear stress on this crystal that tries to make it straight (blue
arrows in the next picture). The necessary deformation we do first in the standard way by moving normal (or "perfect")
dislocations through the crystal. The next picture shows how that works: |
| |
|
| |
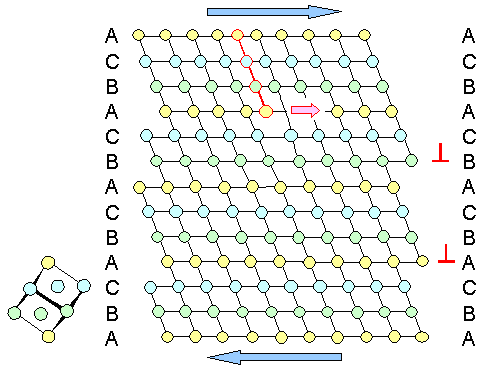 |
| Deformation under shear stress with dislocation glide |
|
| |
|
|
 |
Two edge dislocations have moved completely through the crystal at the positions
marked by the red edge dislocation symbol. One dislocation (marked by the red line) is still moving to the right, so we
only have a step on the left-hand side but not yet at the right-hand exit side.
Three dislocations are enough to "straighten"
our crystal here. On which glide plane the dislocations move is pretty much random. It depends on where the dislocations
come from or could be generated, and that is somewhere to the left of our model crystal, about which we don't know anything. |
 |
Now let's do the deformation by twinning: |
| | |
|
|
|
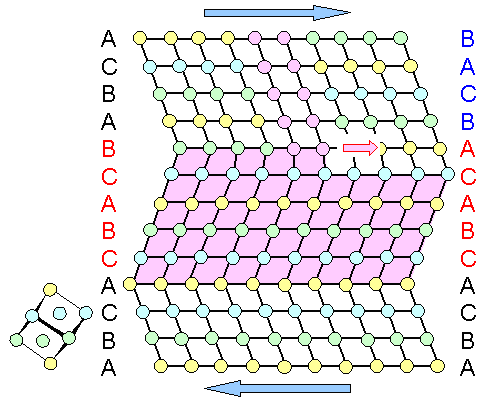 |
| Deformation by twinning |
|
| |
|
 |
This is an interesting picture and there are several things I need to point out
and discuss a bit. |
|
 |
First, it is quite clear that deformation
by twinning is just as good for the task of "straightening" our crystal as deformation by dislocation glide. Since
we can do twinning in several directions, we can make any shape change by twinning just as well as by dislocation glide.
To be sure, there are some differences but on a more macroscopic scale they wouldn't be noticed. |
|
 |
Second, the structure after twinning
occurred is something that could have been made by shearing the layers involved
a certain amount. For the final result it wouldn't matter if you do that by shearing one layer after the other, or all of
them at once. However, that only describes the result of what has happened but not how
it happened. It actually happens in yet a different way. In other words: You cannot deduce the process by knowing the outcome! |
|
 |
Third we notice that there is step in the twinned
region (marked in purple). On the left, the lattice is twinned, on the right it is not yet. In between is actually a dislocation,
but not a normal or perfect dislocation as in the figure farther up, but a so-called partial dislocation
of the Shockley type, to be precise. It is not easily visible because
there aren't some ending lattice planes, but that is not really what makes a dislocation.
Now you must make a decision. Either consult the dislocation modules or just keep reading and skip
over unintelligible details. You will still get a feeling for the gist of the arguments. |
|
|
| |
Anyway, the Shockley partial,
as we like to call it, is located around the place where I colored the atoms purple. The difference
between a regular dislocation and a Shockley partial dislocation is that the latter is schlepping a stacking
fault along as it moves to the right. Doing that is usually not a good idea because the energy of the crystal increases
as the stacking fault area increases. However, if twinning is what the crystal wants, it is exactly the stacking fault that
does the job. On the left to the Shockley partial its stacking fault just adds to the twin. A stacking fault by itself could
also be seen as a kind of one (or two) layer twin.
The long and short of all of that is: Move a Shockley partial from
left to right on adjacent {111} planes and the result is a twinned area. |
 |
Now the big questions come up:
- How does the crystal decide which kind of deformation mechanism it wants to activate? Normal dislocation glide or twinning
by Shockley's. What are the criteria?
- Are there, maybe, other ways to make twins?
- Once a decision has been made, how is it implemented?
|
 |
A lot of experts have spent considerable time to ponder these questions.
I don't think the last word is in yet. The reason is that what I have shown above is highly simplified. Look, for example,
at somewhat more complex crystals than fcc metals and just the geometry of twinning is not all that easy to figure out. |
|
 |
Nevertheless, the basic picture is clear. If a real
crystal is exposed to stress, stress levels vary locally to some extent. At some small surface defect or close to an internal
defect like a little precipitate, at some place at a grain boundary, and so on, the stress might be a bit higher than the
average. Stress might be seen as an energy source for getting dislocations to do something. A second energy source is the
thermal energy contained in the vibrations of the atoms, and this thermal energy always is a statistical entity - it fluctuates
wildly around some fixed average called temperature. This essential adds up to a probability
that something might happen locally. An existing dislocation might pry itself off the
obstacle it was locked to, some dislocation source might become active and spew out a a new dislocation, a grain boundary
might move a bit because some atoms get enough energy to rearrange - a lot can happen in a real
crystal in some particular places.
This is in stark contrast to perfect crystals we love to draw on the black-boards,
or in the many schematic figures here or in other books. For these unreal crystals either nothing
happens or the same thing happens everywhere since no place is different from any other. |
|
 |
Note that I have not yet differentiated between perfect dislocations,
partial dislocations, or whatever else there could be. Whatever there is or can be produced, responds to the local conditions.
If something happens at some place is always "statistical" to some extent. That means that there is, for example,
a certain probability that a dislocation starts moving, but not a certainty.
If you look at the simple picture of standard
dislocation glide above, I have put the three dislocations on the layer No. 3, 7, and 9, respectively. I just as well could
have picked other combinations. You can cast a die - it is a probability thing.
Now note that it is actually easier
to start a Shockley dislocation moving than a perfect dislocation. The Shockley dislocations just will not go very far because
the stacking fault it pulls out tethers it to its starting point like a rubber band. That's why under normal conditions
only perfect dislocations will eventually produce slip and deformation. |
 |
Now consider our real crystal under circumstance where you slowly
increase the outside (shear) stress. The crystal will respond, and the way it does it is akin to what a beleaguered city
will do if put under stress by some enemy ante portas. It will first huddle up, trying to get by without doing anything
drastic and hoping that the enemy will eventually go away and everything returns to what it was. We call that elastic deformation. |
| |
 |
However, if the stress increases, it will become unbearable in some
place and for some people. Some soldiers might run out to encounter the enemy in one-to-one chaotic battle. We call that
the sweeping out of the first perfect dislocations.
Everything now depends on the outcome of that first random encounter
on the battlefield. If the defenders can sweep the attacking army back, their number will increase because other soldiers
and fighters will come out (dislocations multiply). It the defenders get stuck in the fortifications of the attackers (all
kinds of hardening mechanisms become operative), the stress goes up and the city will eventually break (fracture occurs). |
 |
You realized, of course, that I have left out an important option.
The city organizes its defenses. It doesn't rely on more or less independent warriors
or irregulars running out through all those gates to encounter the enemy, but organizes structured units that attack in ordered ranks like this:
|
| | |
|
|
|
 |
| Shockley Romans about to do some twinning. |
|
| | |
|
|
 |
You can go fast and far this way and clear out a whole area leaving
everything whole again in your wake. That's what happens during twin formation. The fighters, excuse me, dislocations, have
to be next to each other and move in a coordinated way. Schematically, it looks like this: |
| | |
|
|
|
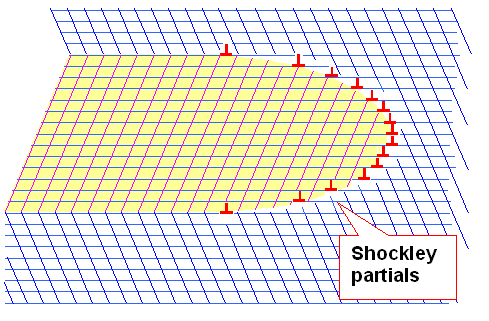 | | How a twin moves |
|
| | |
|
|
 |
The figure explains directly why twins (and, as we shall see, martensite
"lathes"), typically are kind of lens shaped. They simply must have a kind of pointed and drawn-out tip region. |
 |
A twin tip, advancing with the speed of sound, is a formidable force in the deformation game played by crystals and stresses. It is
far more effective than some randomly running perfect dislocations, even so a perfect dislocation, having a big Burgers vector of b = a/2<110> in fcc crystals, could always beat
a meager b = a/6<112> Shockley dislocation if matched one by one. |
|
 |
So, why isn't twinning the rule and not the exception? Well, as you
know, the first law of economics cannot
be beaten. There is no free lunch. It takes more initial energy to organize a bunch of Shockley partials into the right
configuration than having a bunch of perfect dislocations running out at random. Under normal circumstances, the irregulars
will always move first, and if they can repel the enemy (reduce the stress), there is no incentive to organize a bunch of
Shockley Romans. However, whenever the irregulars run into problems, the hour of the professionals may have come. When will
that be?
- Whenever the irregulars can't get out in the right direction. In other words, if there are not enough glide systems
to allow response in the direction of major stress. This is the case for hcp crystals.
- Whenever the irregulars are not very effective because they
are drunk can only move slowly and sluggishly.
In other words: whenever the temperatures is important for dislocation movement and
is low. This describes bcc crystals. |
 |
This fits the observations
concerning twinning. So we only need to discuss one more question: |
| | |
|
|
|
How does a crystal organize a bunch of Shockley dislocations into an ordered formation?
|
|
| | |
|
|
 |
Figuring that out will be your homework till tomorrow.
But seriously
now, this central question is not so easily answered. The crystal, of course, can't organize a bunch of Shockley dislocations
into a fighting squadron anymore than a lawyer can make a microchip. It needs some knowledge and intelligence to do that.
What is going on is known under the term self-organization or synergy. Whatever that
means, the formation of a twin front as shown above is possible as soon as some primary nucleation,
i.e. the formation of a tiny little twin tips, has occurred. That is a probability event that just will happen somehow,
somewhere, and sometime with a probability that depends on circumstances. If it has
happened, things develop more or less automatically and rather fast. The little twin grows and moves by creating more Shockley
partials (there are mechanisms for that) and will keep going until it runs into an obstacle like a grain boundary. |
|
 |
I will not go into more details. Things get rather complicated and
involved. Our twin boundaries actually are grain boundaries, and our Shockley partials
actually are grain
boundary dislocations that "live" in a lattice different from but related to that of the crystal.
A taste treat of what that involves can be found in this link. |
 |
One last word. While the general outline given here is not too far
from the truth, I'm not really sure how close it is either. One of the basic problems with twinning is that nobody has ever
seen it happening. Even if your specimens in the electron microscope would do some twinning, you wouldn't see a thing. The
twin sweeps across the crystal with the speed of sound (easily more that 1000 km/h in a solid), and the few micrometers
you see at high magnifications will be covered in far less time then the quintessential blink of an eye. One moment there
is no twin, the next moment there is one. |
| |
| |
| |
Martensite Formation |
 |
Martensite formation, as we will see later in some detail, can be described by
some shearing of the crystal lattice quite similar to the description
of twinning. It also happens quite suddenly and fast at low temperatures in many kinds of crystals. There is a major difference,
however: |
| | |
|
| |
The crystal does not just deform but changes to another phase
|
|
| | |
|
| |
 |
In other words: a different kind of crystal is formed. Between the crystal and the newly formed
martensite is now a phase boundary and not a grain boundary. That makes
a formal description of how martensite is formed different from that of twin formation and even more involved.
On the
other hand, the martensite phase of a given crystal is made from exactly the same atoms as the crystal itself, just the
arrangement is different. That means that martensite can form if the atoms move to new positions in the right way, and that's
where the similarity to twinning comes in. |
|
 |
I have illustrated that by using the picture from above.
All that has changed is the coloring and the lines "to guide the eye". Instead of a twin we now have a martensite
with a unit cell quite different from that of the crystal (inset on the left). It happens to fit perfectly to the crystal
lattice because that's the way it was drawn. |
| |
| |
| | |
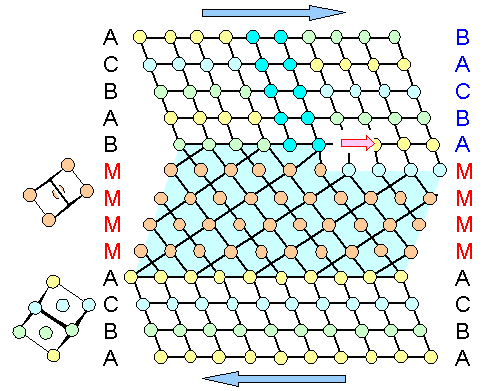 |
| Martensite formation by shear |
|
| |
|
|
 |
For real martensite that will not be the case. There would be stress and strain and possibly
"misfit" dislocations at the phase boundary, it wouldn't have an exact mirror symmetry, and so on. |
 |
Nevertheless, much of what is valid for twinning is also valid - with some adjustments,
perhaps - for martensite formation. The major difference is that the driving force is not so much outside stress but the
need to change the phase. The common denominator, however, is always that the energy of the present situation (high stress
levels, wrong phase, ...) can be lowered by doing something, and that the usual way of doing something is blocked for some
reason (wrong geometry, low temperature, ...). |
| |
| |
© H. Föll (Iron, Steel and Swords script)