|
|
| |
A closer Look at Stress and Strain |
 |
First we have to look a bit more closely into what the terms stress
and strain actually mean. Yes, I did give you a simple and good definition way back in chapter 3 (and in a related science
module) but we still need to look a bit more closely. Here we will do a few thought experiments to find out what, exactly,
constitutes stress and strain - and what just looks like stress or strain but is something else. The relation between displacement and strain gives an example; it can be found in this
link
I'm going to use the expressions "stress" and "strain" as more or less synonymous because
if you know one you know the other. As longs as only elastic deformation occurs, stress
and strain are simply related by elastic modules like Young's
modulus, shear modulus and so on, or the elastic
coefficients if you want to be fussy.
Since the topic is "thermal stress",
I'm going to play around with temperature a bit. Let's heat up some steel, for example your sword blade. |
 |
Increasing the temperature of a solid will (almost) always increase its size -
we have what is called thermal expansion. In a science
module way back I have actually shown why there is always some thermal expansion
and how one can actually calculate it.
The thermal expansion coefficient
a puts a number on this material property. It is given by |
| |
|
| |
| a | = |
l(T) – l(T0)
l(T0) | · |
1
T – T0 |
| | | |
| | | |
| Relative
length change | |
Temperature
difference |
|
|
| |
| |
|
 |
So the numerical value of the thermal expansion coefficient is simply the relative length change caused by a temperature change of 1 K. Its unit is [a]
= K–1. It is a property of the bonds between the atoms and that means, as in the case of Young's modulus, that all
(low-alloy) steels have pretty much the same thermal expansion coefficient as pure iron:
|
| |
| |
| |
|
airon= 11.8 · 10–6 K–1 or
airon= 11.8 µm/m·K
|
|
| |
| |
| |
|
The last number means that per m length an expansion of 11.8 µm
takes place if you increase the temperature by 1 K or, same thing, by 1 oC. |
 |
Now let's do our simple thought experiment. Take a rod of something and heat it
up. It will get somewhat longer (and a little bit thinner) - provided you don't keep it between two ungiving restraints.
Schematically it looks like this: |
| |
| |
| |

| | Thermal expansion without restraints. |
|
| |
| |
| |
 |
Seen from the outside, you will have a relative length change
Dltherm = a · DT
· l0.
When relative length changes were caused by stresses, i.e. force acting on
the material, we have called that a strain
e, defined by e = (l – l0)/l0.
So do we have something like a thermal strain etherm = a
· DT here? Note that there is no stress whatsoever in our experiment.
No forces were applied.
The answer is: No!!!. There is no strain or stress. You
must be a bit careful about your frame of reference when invoking stress and strain. You
look at your hot sword blade from the outside and compare it to some cold state. That's
fine but there are other ways to look at this. Seen from the inside, or from the viewpoint
of the crystal, there is no strain and no stress because it has the length it likes
to have at the given temperature. In formal terms we have - Outside view:
l0 = l(T0) = constant for all temperatures.
- Inside view:
l0 = l(T) = increasing with T.
It should go without saying: As far as processes like deformation or fracture are concerned, the inside
view is the one that counts. |
 |
Now we repeat the experiment but do not allow our rod to change
its length. We just keep it between massive unmovable restraints. There can be thus no length change and there is thus no
strain as perceived from the outside. In contrast, we now have plenty of stress. The
rod is pressing hard on the unyielding restraints and since they are unyielding they press back with equal force or stress.
It can be visualized like this: |
| |
| |
| |
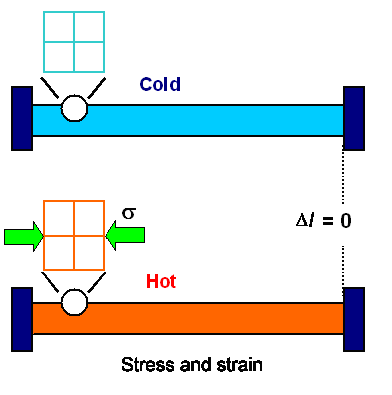
| | Thermal stress and strain with restraints. |
|
| |
| |
|
 |
It is easy to calculate the stress in the sample. You have stherm
= Y · a · DT because this is the
stress you need to counter the thermal expansion, or, in plain words, to compress the expanded rod back to its original
length.
In the inside view you have the same stress but also the strain
that goes with it.
So, as a first conclusion, different temperatures between your
specimen and the environment may or may not cause stress and strain in the specimen - it all depends on the "boundary" conditions.
|
 |
From now on we will look at stress and strain exclusively from the inside view.
This is very easy. All you need to do is to delve inside a crystal (use your brain microscope for this). Look at an elementary cell of the crystal. If is is exactly what it is supposed to be at the local temperature encountered
there, you have neither local stress nor strain. If the elementary cell is not what it is supposed to be, you have stress
and strain.
For iron, the elementary cell is always a cube. The dimension changes smoothly with temperature and abruptly
when you have a fcc Û bcc transition - but it is always well defined. |
|
 |
If you find that the elementary cell (EC) is not what it is supposed to be, you can draw a
few conclusions from its shape (look at the old picture
to "see" it):
- The EC is still a cube but either too large or too small. You have uniform compression or expansion (tension in all
directons) , respectively.
- The EC is a cuboid (longer in one direction). You have uniaxial
tension.
- At least one of the formerly square sides of the EC looks like a parallelogram now. You have shear stresses (and also
normal stresses if the volume has changed, too).
|
| |
Temperature Gradients Cause Stress and Strain |
 |
Now let's do another simple thought experiment. Take a cube of something. Take
out a smaller cube from the interior (easy with your brain knife), heat it to some higher
temperature, and put it back again. We are looking at a hot potato or whatever is hot inside and cold outside. |
|
 |
The hot core won't fit into its original place because it is now larger due to
thermal expansion. You must either squeeze it to its old size, putting it under compressive
stress and strain, or you must pull at the cold piece, elongating it in all directions by the proper amount to make the
inside space large enough. In other words: you must put the outside under tensile stress and strain. Read up about stress
and strain here, if you need a refresher.
In reality, of
course, for a hot core inside a cold shell, the stresses are shared between core and surroundings. The hot inside will be
under compressive stress and the cold outside under tensile stress. If the inside is cold and the outside is hot (this happens
when you put your sample into a hot furnace), it's the other way around. Note that the EC's are not what they are supposed
to be. The ones in the hot core are too small for the temperatures there, the ones at the outside are too big, and in between
it's messy anyway.
The long and short of all of that is simple: |
| | |
|
|
|
Thermal gradients in a specimen always
cause mechanical stress and strain
|
|
| |
| |
|
 |
"Thermal gradient" is a fancy word for "differences in temperature on a small
scale", or dT/dx
¹ 0 in math lingo for one dimension. In full generality for three dimensions the temperature
gradient is a vector with the components dT/dx, dT/dy, dT/dz,
or ÑT in shorthand. |
 |
All of the above is tied to thermal expansion, the change of the lattice constant with temperature. However, we also might
have expansion or compression whenever a phase change occurs. This
is easy to see if we do another simple thought experiment |
|
 |
Take a cube of pure iron and heat it to 912 °C (1.674 °F). It will then be in the
fcc phase. Take out a smaller cube from the interior (easy with your brain knife), cool
it to 908 °C (1.666 °F), and put it back again. The dimensional change due to thermal contraction is so small
that you can neglect it. Your slightly colder piece, however, will not fit because below 910 °C it changed to less
dense bcc iron and thus expanded. Again you need to squeeze it, and so on, so it can be put back inside. What we conclude
is: |
| |
| |
| |
Phase changes in only parts of the specimen always cause mechanical
stress and strain
|
|
| |
| |
|
 |
The specific volume or the density of different phases is generally different, and that means
that local phase changes can also cause a lot of stress and strain. We use that, after
all, to measure phase and TTT diagrams. The "local" is important. If
all of the sample has changed to a different phase, there is no stress and strain anymore
(assuming that there are no ungiving restraints). |
|
 |
For steel that is certainly true. Ferrite (or martensite) has a bcc type lattice and the crystal is thus by definition not as densely packed as the fcc austenite. Ferrite or martensite formation from austenite thus always expands
the lattice. Martensite is also less dense then ferrite since it contains all this carbon that must expand the lattice somewhat
on average. Of course, if we also have cementite in the mix, things get more complicated. |
 |
We now have two mechanisms that can produce stress and strain and it is clear
that both are related. If there are temperature gradients, the more quickly cooling parts of the sample will experience
phase transitions sooner than the more slowly cooling parts, producing local phase changes and the strain / stress going
with that. |
|
 |
This can lead to tricky situations. Assume, for example, that the "lower"
part of your sample cools down more quickly than the upper part (you might imagine the edge of a katana sword blade during
quenching). Let's list what could happen (shown schematically in the figure below):
- Let's assume the blade is pure iron and ferrite everywhere when you start cooling.
It then will stay pure ferrite at all times. During cooling the upper (thicker) part is always hotter than the lower (thinner)
part with the edge. The stress produced by the temperature gradient then always bends the blade downwards
until the temperature is the same everywhere. The larger the gradient, the larger the bending. This is illustrated in the
upper part of the figure below.
- Now let's assume that the blade is still pure iron but austenite everywhere when
you start cooling. The austenite - ferrite transformation temperature then is first reached in the lower part. Ferrite formation
thus starts in the lower part and moves upward. The "local phase change" stress is produced at the phase boundary
where the lattice constants don't match.
The phase transformation austenite / ferrite by itself, while it takes place,
leads to an upwards bending until the transformation is complete everywhere. This is
illustrated in the lower part of the figure below. Note that you don't need large temperature gradients for producing major
effects. It's enough if the lower part is slightly below the transformation temperature and the lower part slightly above.
The "local phase change" induced bending is proportional to the difference in lattice constants between the two
phases.
On top of that you still have the downward bending caused be the temperature gradient and thermal expansion.
It is somewhat different from case 1 because the thermal expansion of ferrite is different to that of austenite - but you
still have temperature gradient induced downwards bending.
The total bending effect
will be given by the superposition of the two mechanisms. It could go either way, depending on "details".
- Now let's assume that the blade is homogeneous steel with some carbon concentration,
and that you start cooling in the austenite state. We have all the effects from case 2 with the added complication that
the phase transformation is to some pearlite plus ferrite or cementite. The "local phase change" induced upward bending part caused be the phase transformation sweeping through the blade will thus have
a more complex behavior. The local bending that we need to consider now, is proportional
to the difference of the (average) lattice constants of the phases encountered locally.
- Take case three and add martensite formation in the edge area where the cooling
rate is fastest. This leads to major stress with respect to the still present austenite there and still some stress to the
eventually forming ferrite / pearlite / bainite. In any case the martensite part would tend to bend the blade upwards.
- Take case four but now for a real katana, with soft steel
inside and hard steel outside, that is pitched into cold water. Extreme temperature
gradients are produced. What will happen during cooling, considering that all kinds of phase transformations are encountered
locally at different times / temperatures, is not all that easy to figure out.
|
 |
Once more: The total strain / stress produced at some given time in any part of
the blade is obtained by summing up all those effects. Which way the blade will bend at any moment in time, what else might
happen, and what the final shape will be, is far from being obvious. |
| | |
|
|
|
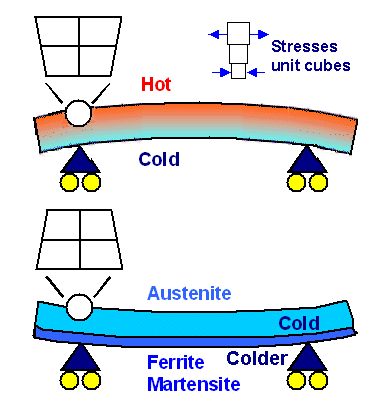 | Thermal stress and strain due to
only temperature gradients or only local phase formation. |
|
| |
| |
 |
In other words: it is easy to quench a hot blade in cold water or some other cold
liquid, but not so easy to figure out beforehand exactly what is going to happen. And we are far from being done yet. |
|
 |
To make things a bit more complicated, it is necessary to point out that the trapezoids showing
the deformation above may reflect the shape of real elementary cells of the crystal but must not be confused with a "unit cube" that has been deformed by the acting stress
tensor. You cannot deform a cube into a trapezoid by one set of forces acting on its
surface. You need to change forces over a sequence of stacked cubes or, in other words, the components of the stress tensor
must vary in a certain way going from the bottom to the top as shown in the upper right hand corner of the figure.
But
that doesn't need to bother us here. We are not going to calculate anything here. It will be sufficient to deduce what is
going to happen in a qualitative way. |
| | |
|
| |
Global, Local, and Microscopic Stress / Strain |
 |
First we need to realize that all of the above is rather general. It would be
true for perfect single crystals and for fully amorphous materials like glass. The silicon people, for example, dealing
with rather perfect single crystals, can tell you a lot about stress and strain produced in silicon wafers by thermal gradients
or by having a different phase (like silicon dioxide (SiO2)) on one side of the specimen.
On the other hand,
a silicon wafer at constant temperature and not molested by other phases is completely
stress and strain free, no matter what the temperature. Another way of saying that is: wherever you look inside the crystal
you find a perfect unit cell of the crystal structure with just the right dimensions for the given temperature. Yet another
way of stating this is to say that a perfect single crystal at constant temperature contains no residual
stress and strain. |
|
 |
Now look at your sword blade or car. Dealing with iron and steel (or just about
any metal), we never ever come even close to a perfect crystal. We have grain boundaries,
dislocations, precipitates and God knows what other defects in there, typically at quite high densities. The density type
and distribution of those defects will typically be different after the thermal treatment to what it was before.
Now
any dislocation has a stress and strain field surrounding its core. Precipitates of any size, grain boundaries, or just
about any crystal lattice defects also have stress and strain fields in their immediate environments, no matter if the specimen
is hot or cold or experiences thermal gradients. "Immediate environment" means
that the stress from some defect is only noticeable close to the defect on a scale of some ten nanometers at best.
|
| |
| |
| |
Defects produce stress and strain on a microscopic scale
|
|
| |
| |
 |
In the figures above I have shown what I called local
stress and strain. It changes when you move across a specimen but on a macroscopic scale. The whole edge of your blade might
experience local strain = strain in the edge = strain on a macroscopic or "global"
scale. It is clear (at least in principle) how thermal gradients and what I now will call "phase distribution gradients" produce large scale if local stress / strain, and that the effects
disappears in a perfect crystal as soon as the gradients are zero. |
|
 |
In the final product we typically only worry about large scale stress and strain. As long
as the defects in there are uniformly distributed, their microscopic stress and strain fields average to about zero on large
scales, on small scales it is a kind of background "noise".
In other words: If your object has an easily
recognizable different shape after the heat treatment (e.g. your blade is now bend), is has experienced large-scale stress
and reacted by changing its shape. If its shape is not changed noticeably on a large scale, a formerly mirror-like surface
might now be a bit dull, because the microscopic strain around defects produced during cooling makes it rough on a small
scale. |
 |
Generalizing a bit, we note: Defects in a crystal are the only
sources of internal stress and strain. A perfect crystal not under any external constraints and thus not experiencing stress
on its outside, is stress-free by definition.
Thinking that through you realize that the global
stress / strain state of a non-constrained specimen at a constant temperature can only
be the sum of all the microscopic stress and strain around its defects. So sum up a lot of tensors and see what you get! |
|
 |
Haha. Nothing like a good joke among friends every now and then. Let's only look at two easy
points:
- If you just look at the stress picture of a dislocation,
you see just as much red as green, or just as much compressive as tensile stress. It's then easy to conceive, I hope, that
the stress and strain fields of a random distribution of random dislocations essentially average to zero. For every compressed
part somewhere, there is a stretched part somewhere else.
That's also true in the spirit for all other kinds of defects.
- Generalizing from that, we can deduce that large-scale or global residual stress
and strain at constant temperature can only results from an non-uniform distribution
of defects (or phases).
|
|
 |
Time to look a bit more closely on what really happens when cooling down a sword blade. |
| |
|
| |
Residual Stress and Strain on Large Scales |
 |
Sooner or later thermal gradients disappear. Your specimen or sword now has the
same temperature and the same phase(s) everywhere. Then all large-scale stress and strain caused by temperature gradients
and local phase differences during its making should be gone. Ignoring the microscopic stuff coming from defects, your specimen
should have its old shape and there should be no residual stress or strain left in
your blade. Is that true?
If that would be always true, I wouldn't need to write a lot more in this module. Major complications
come from the fact, that the quiet little assumption I made all the way up there, is quite
often not correct. It is often not true that only elastic deformation
occurs as a reaction to stress induced by temperature gradients or local phase changes!
Taking this into account, we
might now distinguish between two basic cases: |
|
 |
1.
Local stresses produced during cooling by any one, or any combination, of the possibilities
enumerated above will always (= for at all local temperatures and at all times) be below some critical
stress. Then you will only find elastic strain that disappears if the stress causing factor disappears. To what
extent will the stress causing factors disappear? Let's see by looking at the stress causing factors individually (i.e.
assuming that they are the only ones in some specimen):
- Temperature gradients only. Eventually they disappear completely - and so does the
elastic stress / strain caused by them. If you start with a straight iron blade (or, more to the point, a perfectly flat
silicon wafer), you may observe substantial bending during cooling but always end up with a straight iron blade (perfectly
flat silicon wafer) identical in shape to the one you started with.
- Phase transformations between stable single
phases only (e.g. between austenite and ferrite in pure iron). Eventually they will be finished and no phase boundaries
causing stress are left. If you start with a straight austenite blade, you may observe substantial bending during cooling
but always end up with a somewhat larger straight ferrite blade.
- Phase transformations between stable single and mixed
phases. Eventually they will be finished and lots of phase boundaries are left (e.g. between ferrite and cementite). There
is a lot of stress and strain across the phase boundaries - but on large scales it averages out as long as the phases are
uniformly distributed. If you start with a straight austenite blade, you may observe substantial bending during cooling
but always end up with a somewhat larger and straight pearlite / ferrite or pearlite / cementite blade.
Well - only
up to a point. While you must have the same (average) carbon concentration everywhere, and thus the same amount of ferrite
/ cementite everywhere, the structure might be different. In the parts that cooled faster,
cementite lamellae are closer, or you might even get bainite, compared to the regions with smaller cooling rates. That might
cause some residual stress on large scales but one would not expect large effects.
- Phase transformations that produce metastable phases,
e.g. martensite. If that happens uniformly everywhere in your blade, it is not different
from point 3 above.
However, if it only happens in those parts of the sample with very high cooling rates, e.g. around
the edge of a blade, there will now be major residual elastic stress and strain across
your blade, tied to the distribution of the metastable phase.
If you start with a straight austenite blade, you observe
sudden substantial bending during cooling on top of thermal gradient bending, and end up with a bend blade that contains
martensite only in the edge regions and ferrite / cementite and retained austenite in a non-uniform distribution / structure
in the bulk.
The list could go on. The next point would be to look at composites, e.g. a blade with a low-carbon core and a high-carbon
shell. Then we need to look at different cross sections. A bar of a homogeneous material with a rectangular
cross-section, for example, would not bend because whatever happens in one colder region
close to the surface and causes bending, also happens in exactly the same way on the other side, causing bending in the
opposite direction - the total effect is zero! Single-edge blades, however, have a kind of triangular cross-section and
therefore will bend.
I will not go into this, however. The list above is good for
keeping things straight in your head. It is not extremely useful in practice because for all cases where some substantial
bending of a blade during cooling is expected, you will for almost sure find yourself considering the second
major case: |
|
 |
2.
Local stresses, produced during cooling by any combination of the possibilities enumerated
above, will exceed a critical value at some place and time. Something else besides just
elastic deformation is then going to happen. This something else could be
- Plastic deformation if the yield stress is exceeded.
- Fracture if some critical fracture stress is exceeded.
Both mechanisms have something in common: They relax, i.e. lower the stresses,
preventing that they get much larger than the critical value. That could be good but there is a price to pay:. Stress limitation leads to the destruction or at least the permanent deformation
of the material. For a sword blade that could mean that it shatters into several parts, is still in one part but has cracks,
or is bend but without containing much residual stress. |
 |
Let's look at brittle materials and fracture
first. As far as sword blades are concerned, we don't want that. In fact we almost never want that.
|
|
 |
That is the reason why you must cool all brittle materials
rather slowly. Large chunks of glass, just taken out of the furnace and left to cool in air, will at some point in time
violently explode into thousands of pieces without prior warning. The huge glass mirrors for big telescopes must be cooled
down very slowly in a process that takes many month in order to avoid self-destruction. Of all the mechanisms enumerated
above only temperature gradients apply so it is conceptually easy.
The stress relaxation mechanism is clear. As soon
as the critical stress for fracture is reached, microcracks spread with the speed of sound, sending shock waves through
the material. The freshly created surfaces cool down quickly, causing new temperature gradients and more crack growth -
the process, once started, mushrooms into a full-fledged explosion. The pieces left over, however, will be (almost) completely
stress free. |
 |
Now let's look at ductile materials. In these
cases you simply can never exceed the yield stress very much. This becomes clear if we look at some stress-strain curves
again, slightly redrawn for the purpose here. As soon as the
yield stress is reached somewhere in the sample (about 200 MPa in the figure below), plastic deformation starts and the
stress just won't go up much anymore. |
|
 |
If you need to produce some strain e1 as indicated
to accommodate thermal gradients or whatever, you need far less strain if the material deforms plastically than if it would
only respond elastically as the figure clearly shows. The price to pay for this is that you now have plastically deformed
your specimen. If you remove the stress, it will be longer, shorter, bend - whatever deformation the stress was trying to establish.
|
| |
| |
| |
 |
Stress needed for some strain e1
for elastic and plastic deformation |
|
| |
| |
|
 |
The shape change may be welcome or annoying - but it is far better than fracture! What exactly
happens depends, of course, on the local value of the yield strength at any point in
time. It will be different for different phases and temperatures. |
|
 |
For the same reason, whatever residual stress you might have left at room temperature because
you have an inhomogeneous defect distribution (e.g. a martensite edge, a ferrite body with a little bit of pearlite, and
a bainite outside for a "simple" katana) will be smaller than the local yield stress, otherwise plastic deformation
would occur. In the martensite rich region of the edge the yield stress is rather large, so large residual stresses could
exist in the edge of "quench-hardened" swords but not in their softer interior.
Is this good or bad? Well
- I don't know at this point but I will get back to the issue later. |
 |
Plastic deformation during cooling is sure to happen in all sword
blades that are quenched, and most likely even during normal cooling. Just consider if a piece of glass with the same dimensions
would live through a similar treatment. If it shatters (and it will!) your sword blade would shatter, too, if it couldn't
deform plastically.
Now I can finally make a major point rarely considered in sword making; |
| |
|
| | |
Composite swords have a soft core because otherwise you couldn't make them.
|
|
| |
| |
 |
Damascene technologies of all kinds are usually only discussed in terms of the
better properties they are supposed to have. "A combination of hard steel and soft iron would impart hardness and elasticity
to the blade" says a major source that shall stay unnamed. B.S!
I say. Whatever properties the composite will have, the first reason for using the "soft-hard" combination
is simply that he blade might simply not survive its making.
I would bet all your money that you could not make a katana
without the soft core. It would simply shatter during the quenching. |
|
 |
That is a basic truth concerning most metal objects. While you, the gormless customer only
care about the properties of the material, I, the engineer who has to make it, also care about the making. You
want the body of your car to be hard but ductile. It shouldn't give easily but rebound to its old shape after you hit that
tree. And even if it deforms on a major impact, it should never shatter into 1000 pieces. I
need to deform that piece of sheet metal into a complex shape so it can be part of your car body. The softer the metal ,
the easier the job. The kind of metal you have in mind does exist - it just can't be shaped with ease or simply not at all.
For your sword, as your for your car body and many other objects, we have a simple if largely unknown truth |
| |
| |
| |
You need to compromise between final properties and ease of making!
|
|
| |
| |
|
 |
I'll get back to this when I go into swords and sword making proper! But one thing is clear
already now: What, exactly, is going on during the final quenching of a katana is rather complex. It is small wonder that
you only get a good blade if everything is done "just right". |
| |
| |
© H. Föll (Iron, Steel and Swords script)



