| |
11.6.2 Making a Japanese Sword - Part 1 |
| |
The Japanese Way |
 |
Entering sword in a search engine in March
2015 netted 205.000.000 entries. Entering Japanese sword yields 11.100.000. One should
think that it is easy to get a bit of unambiguous information about the basics. |
|
 |
Not so. A lot of the entries with high ranking are of Japanese origin and are
often written in Japanese or in a somewhat questionable English, always with a lot of Japanese terms. And I include papers
found in science journals here. If you do not know, for example, that a "tatara" furnace is just one of the many
variants of the good old bloomery, you won't learn that from most of what you find in the Net or the science literature.
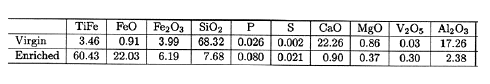
As for the iron sands used as ore; I have given up trying to figure out exactly what that stuff is. Here are examples that
shall go unnamed:
- This material (iron sand), which consists of weathered granite, contains about 58 % iron along with very small amounts of titanium
and other impurities.
- Iron sand is a type of sand with heavy concentrations of iron. It is typically dark grey or blackish
in color. It comprises mainly magnetite, Fe3O4, and also contains small amounts of titanium, silica, manganese,
calcium and vanadium.
- The iron sand used is also of two vastly different types. They are the masa iron sand used chiefly
with the kera-oshi method, and the akome iron sand used for the zuku-oshi pressing method. Masa iron sand comes from a kind
of granite made of acidic rock and has low titanium content. Akome iron sand comes from a kind of diorite made of basic
rock and has high titanium content, with the presence of TiO2 at 5% or greater.
- Iron sand with 2–5% content of iron mined from mountains ... is concentrated to the degree
of 50–60% by a magnet system (...) Such enriched iron sand called masa satetsu contains 8% of pure iron Fe and iron
oxide Fe2O3
(=haematite) with a
very small amount of impurities such as 0.026% P and 0.002% S being injurious for carbon steels. Chemical compositions are
shown in Table 1:
|
| | |
|
| |
| |
|
 |
Huh? Pure iron is contained in iron sand? A lot of TiFe appears after enrichment?
Plus wustite (FeO), something normally not found in nature.
Fe2O3, by the way, is non-magnetic hematite containing 69.9 % iron. The magnetic stuff would be magnetite
with 72.4 % iron by weight. |
 |
And so on. I have yet to find an article that describes the making of a Japanese
sword in the framework and language of modern iron and steel science and technology. That adds mystery, of course, and that
is always good for having something look more interesting and special.
In what follows I will stay mostly within the
terminology used throughout the hyperscript. I do apologize if I "translated" something not quite correctly from
the typical Japanese confusionalese. Much of what follows is based on various Wikipedia articles, Hitachi Metal Internet
sites, and various scientific articles, foremost some of Tatsuo Inoue
1). |
| | |
| |
Ore, Charcoal and Flux |
 |
For feeding your smelter you need ore, charcoal and flux - look it up here if you have forgotten. Everything at smelting needs to be "just right"
but as far as the ingredients are concerned, you must go with what you have. |
|
 |
Ore: The ore is iron sand, whatever exactly that is. It was "mined"
from deposits of sand (probably alluvial fans somewhere) and the heavy iron oxide particles were separated from the lighter
stuff by the usual flotation technique: run the mix through a canal and the heavy stuff separates from the lighter one since
it isn't carried downstream as fast and as far.
Obviously the main ingredient of the iron sand is magnetite, the richest
ore of all. That allows to separate it from the rest also with a strong magnet. That was certainly not the technique used
in ancient times so we don't care about that here.
Part of the confusion may result from describing magnetite as a mixture
of Iron(II) oxide FeO (wustite) and iron(III) oxide Fe2O3.
In contrast to the ore used about
everywhere else, the iron sand ores consist of rather small grains instead of sugar-cube sized lumps and that necessitates
some special care during smelting. |
|
 |
Charcoal:
Whoever smelted iron knew that you need the right kind of charcoal. It needs to be just right for the process you use and
it is thus no surprise that the ancient Japanese iron masters had clear ideas of what they wanted, just as all iron or smelter
masters elsewhere. |
|
 |
Flux:
For an old-fashioned bloomery process you typically need something (=flux) containing silica (SiO2) for forming
the liquid slag that is so important for efficient smelting.
Quite often the flux is simply the gangue, the "rocks" still sticking to the iron oxide or ore. In addition, the
clay used for building the furnace also supplies silica, getting "burned off" in the process. There is then no
need to add flux consciously and that is what the Japanese smelters did.
However, your smelting process than tends to
be less efficient and that is particularly true for the Japanese "tatara" type of bloomery. |
| |
|
| |
The "Tatara"
Bloomery |
 |
"The traditional Japanese furnace, known as a “tatara”,
was a hybrid type of furnace. It incorporated bellows, like the European blast furnace, but was constructed of clay; these
furnaces would eventually be destroyed after the first use" says a Wiki article. Sounds like the tatara was
special. Well, it wasn't. All bloomeries after 1500 BC or so had bellows and were made from clay that would eventually be
destroyed. Except for the bloomeries that were about contemporary with the tataras, the Stückofen
or Catalan forge, for example. They were of
similar size but not completely destroyed after a smelting.
I do not know how the tatara developed, it certainly hasn't
always been as big as the ones that are run now or some hundreds of years ago. So let's look at what one perceives as the
typical (late; say after 1600) tatara and see how it differs from your run-of-the-mill bloomery: |
|
 |
It is rectangular with a footprint as large as 3m × 1m and thus even bigger
than (older) late European bloomeries. That means that several bellows are needed to supply the necessary air. The bloom
(called kera) produced after running the tatara for something like 3 days weighs 1.500
- 1.800 kg. There is then no way to get that monster out of the furnace (like the 150 kg blooms of the Catalan furnace or
even the 900 kg monsters of the 1770 Stückofen.). |
|
 |
The walls of the furnace supply the silicates needed for producing the necessary
slag. The initial furnace wall thickness of 200 mm 400 mm is reduced to 50mm -100 mm. That means that the reaction can not
be homogeneous since in the center of the furnace flux material is not available. Note that there seems to be no defined
tap hole for the slag, it just oozes out at the bottom.
Indeed, the bloom seems to be thicker at the edges and that's
also where the high-carbon steel is found. The non-uniform reaction thus may actually be good for quality of the product
- but it is not so great for the efficiency of the furnace. |
| | |
 |
 | | Old and modern tatara furnaces |
| Source: Internet at large |
|
| |
 |
In some drawings of tataras an elaborate underground structure is shown (obviously
not present in the two furnaces shown above) that is supposed to make sure that no humidity
is encountered. "To produce optimum tamahagane it is important that all moisture is removed.
To combat this, the tatara smelter is built on top of a large 3 metre deep underground structure. The structure includes
air ducts, called ko-bune, through which the moisture is drawn out during the manufacture process", says a source
that shall go unnamed. Why moisture is a concern for Japanese ironmakers but not for their colleagues in wet Europe, and
how the underground structure draws the moisture out, remains mysterious. |
|
 |
Since the ore particles are rather small, conditions are quite different from
a normal smelter where the size of the ore and charcoal pieces should be several percent of the smelter dimension, i.e.
5 cm - 20 cm for a tatara. What that means for the smelting process in a tatara is anybodies guess but the air flow resistance and the wall effect must be a concern.
During the 3 day smelting about 8.000 kg of ore (iron sand) and about
13.000 kg of charcoal are fed to the smelter, producing a bloom around 1.500 kg. The ratio of ore : bloom thus is around
5 : 1, not a great value for a large-size smelter.
Here are some blooms: |
| |
 |
 | Blooms (kera)
produced in tatara
furnaces |
| Source: Internet at large |
|
|
 |
This is not clean stuff but the usual messy sponge-like conglomerate of iron
/ steel with all kinds of carbon concentrations plus enclosed slag, charcoal, and dirt pieces. It is certainly bigger but
not necessarily better than European counterparts. It does not contain phosphorous though, due to the ore used. That is
generally very good but prevents decorative pattern welding that relies on phosphorous steel for color contrast.
For
a piece of not-so-great iron / steel, the bloom produced in a tatara furnace is fantastically expensive. The costs for making a tatara bloom are in the $100.000 region, about 100 times more than what you pay for
the same amount of modern steel. |
| |
| |
| |
Producing the Starting Material |
 |
The first "nihonto", the curved single-edged Japanese
swords, were "koto" or old swords from around 900–1596.That
time period covers 700 years and the swords forged around 900 were of course different from those made 700 years later -
if not much in looks, certainly in composition.
The very early Japanese sword smith forged swords around 1000 AD in
the same way some of his colleagues did that in Europe about 1000 years earlier. He made a blade from one piece of iron
/ steel. He might have used just one bloom but more likely he first made that one piece of iron / steel by fire welding
smaller pieces.
How do I know this? From reading a few short remarks in the few contributions related to the topic
that I could find in the Net. The picture below attests to this: |
| |
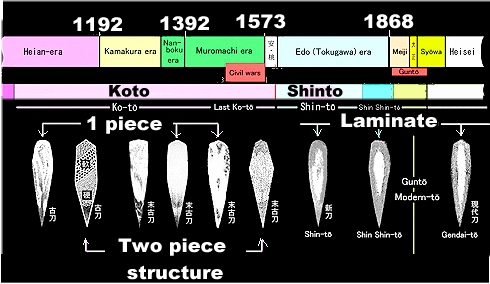 |
Development of the complexity of the Japanese sword blade
Large picture |
| Source: http://ohmura-study.net/994.html.This is not the original source but a student
paper without recognizable references (parts were written in Japanese). I could not find the source of the original picture. |
|
|
 |
Early single-piece blades are not of much interest to us at this point, so let's
go for the more complex ones with a piled construction. That implies that you need at least two different kinds of steel.
European pattern welded blades, for example, used carbon steel and phosphorous steel but the Japanese, being lucky, did
not have to put up with problematic phosphorous steel. They thus needed to separate high and low carbon steel from the blooms
they got. In Japan there was a tatara bloom and the smith (or his co-workers) now needed to do this:
- Sort bloom pieces into low carbon (wrought iron) and high carbon ("tamahagane"). If you have that, you can
make "medium" carbon steel yourself.
- Fire weld the small pieces into a large one and faggot the large pieces extensively.
If all goes right, the smith was now in possession of two largish and halfway homogeneous lumps of different steels.
|
 |
The decisive part of the first point was to find the tamahagane,
the "noble steel, which is spelled as mother of metal in Japanese characters". Tamahagene
is the high-carbon steel that is needed for making the hard parts of the blade. Good tamahagane contains around 1% - 1.5
% of carbon. That is a relatively large carbon concentration; too large actually for the blade to be. The extended forging,
however, will lead to a reduction of the carbon concentration and you must start with concentrations higher than what you
want to have in the end.
I have already pointed out in detail
that a bloomery is likely to produce carbon steel but not necessarily high-carbon steel.
If conditions were chosen to encourage high carbon steel production, there was always a risk that the furnace would switch
to producing (useless) cast iron. Indeed, there are reports that (undesirable) cast iron can be produced in a tatara, and
that you have to pay close attention while running it for avoiding that.
Getting medium and low carbon steel was easy
in comparison. That was the major product of the "civilian" iron industry; you just bought it or found it otherwise.
I'm rather sure that the Japanese were running many "normal" bloomeries parallel to tataras to produce that every-day
commodity. |
|
 |
Now let's assume that a successful smelting has been done in a tatara furnace and that a huge
bloom was produced. The blade smith (or his commercial partner) now needs to bang off pieces with high carbon concentration
and possibly also some with low concentration. How is that done?
Well
- I don't know for sure but from what is written and a bit of logic, a number of points emerge; see also the module "tricks of Smiths":
- Experience. You just know that the high-carbon tamahagane is on the outside of the
bloom, forming part of that jagged ridge you see in the pictures above. Medium and low carbon than is found in more central
regions.
- Looks. There are differences in what the stuff looks like. I have no idea of what
to look for myself but here are some pictures of tamahagane:
|
| |
 | | Tamahagane pieces |
| Source: Internet at large |
|
| |
- Hardness. Bang away with your hammer. Hard steel -surprise! - is harder than soft
steel and you feel that.
- Brittleness. Steel with 1 % or more carbon is perfectly brittle. It will hardly
change shape but will fracture if you bang it hard enough.
- Fracture surfaces. If you managed to fracture a steel piece, the fresh fracture
surface looks somewhat different for high or medium carbon concentrations.
- Specialities. Maybe the colorful parts in the tamahagane pictures above are not
seen in the other steel kinds? Maybe tamahagane rusts a bit more quickly than the low carbon steel when you sprinkle some
vinegar on it? I have no idea myself about that but feel certain that there are some more tricks known to the cognoscenti.
All of these points apply to working with the bloom right at its place of production, possibly while it is still hot.
The smith, after he has obtained pieces with a supposedly known carbon concentration, has a few more ways to check if everything
is right:
- Beat your supposed tamahagane into a thin sheet, then break it into small chips. You will experience "five ways"
of breaking: 1. Difficult to break; just deforms. 2. Breakable with some effort. 3. Easy to break and the fracture surfaces
are smooth. 4. Easy to break and the fracture surfaces are rough. 5. Brittle, shatters into many pieces. Now you have sorted
five grades of steel, from mild steel to brittle high-carbon steel.
- Throw hot pieces into cold water and watch what they do. High carbon steel may shatter. Try the hardness after quenching
with your hammer and do fracture tests once more. Medium and high carbon steel have turned into martensite and are now brittle.
|
|
 |
The essential part, once more, was the high carbon steel, the tamahagane. The softer steels
could also be obtained from other sources, even scrap iron would do: |
| |
 |
| What Miyairi Akihira (1913-1977), sword smith and living national treasure when the pictures
were taken, used to forge a blade |
| Source: Samuraisword.com. |
|
 |
Of course the ancient smiths didn't look for low or high carbon steel; they didn't
know a thing about carbon or martensite after all. They looked for "tamahagane" and whatever they called the other
stuff, for materials having the properties they needed for forging a blade in their particular way. With plenty of experience
they could do a sorting process quite well. I would be very surprised, however, if there wouldn't have been occasional glitches.
In any case, the smith had now piles of little pieces of the various steel kinds. What he needed to do now was to:
- Hammer weld these small pieces into a bigger one.
- Homogenize the bigger pieces by faggoting.
|
|
 |
Yes!
Faggoting! That's what the repeated folding, welding and stretching
is called in English. There is no need to call it "orikaeshi tanren" (Japanese) or "gärben" (German)
when you write in English. The link tells you all there is to know about faggoting. Faggoting was not invented in Japan
and the only question around this process is how the smiths achieves almost perfect weld seams, considering that the "theory" of fire welding seems to be more difficult than the
practice. I have dealt with that extensively so I won't repeat it here.
The Japanese blade smith piled rice straw or rice ash on the pieces to be welded and / or dribbled clay water on them
before welding. I can't see what good that does but then I'm in no position to tell a smith who actually can do fire welding
that he isn't doing it right. Since quite a number of Japanese smiths are forging sword blades right now, there are plenty
of pictures in the Net. Here are some: |
| | |
 |
Miyairi Akihira (1913-1977), living national treasure when the pictures were taken, forges
a blade
Large picture (with additions) |
| Source: Top: Samuraisword.com. |
|
|
 |
You faggot or fold about 10 time, possibly changing the folding directions in between. Ten
foldings would give you 1024 layers as we have asserted before. In a blade that is 5 mm thick, the distance between the
layers then is around 5 µm, just about visible in a high-powered optical microscope. Filing into the blade at a shallow
angle will "magnify" the distance
between the layers: about 10-fold for an angle of 5o and about 30-fold for 2o. Now you can see the
layers with the "naked" eye!
For 6 times folding you get 64 layers at a distance of 78 µm, and 14 foldings
yield 16.384 layers 0.17 µm apart. So around 10 foldings are fine. Your steel will be rather uniform after that but
you still could see a structure on the blade, the "hada". |
| |
 |
Top: "Hada" in cross-section after 16 layers are folded
Bottom: Various "shallow angle" hadas in actual blades |
| Source: Top: FUKUDOME Fusayuki's Japanese Swordsmith Workshop. Bottom: Meiboku.info
site. |
|
 |
The smith can manipulate the hada by making the layers less regular. Fold this
way and that way, make the surface uneven by uneven hammering, and so on. It is quite similar to what wootz sword smith can do. |
|
 |
But I'm getting ahead of myself. We haven't made a blade yet. What we have are two or possibly
three bricks of high, medium and low carbon steel, fairly uniform with respect to the distribution of carbon, slag particles
and whatever else is in there due to the faggoting. Slag particles also tend to be small because all that hammering broke
them into many small pieces.
The medium carbon steel brick we might have made by piling low carbon and high carbon sheets,
fire welding and faggoting. The carbon concentration after 10 foldings will have averaged quite nicely to medium. |
| |
| |
© H. Föll (Iron, Steel and Swords script)