|
Global thermal equilibrium at arbitrary
temperatures, i.e. the absolute minimum of the free enthalpy, can only be achieved if
there are mechanisms for the generation and total annihilation of point defects. |
|
 |
This means there must be sources
and sinks for vacancies and (intrinsic) interstitials that operate with small
activation energies - otherwise it will take a long time before global equilibrium will be achieved. |
 |
At this point it is essential to appreciate that an ideal
perfect (= infinitely large) crystal has no sources and sinks - it can never be in thermal equilibrium. |
|
 |
An atom, to be sure, cannot simply disappear leaving a vacancy behind. Even if the crystal
is finite, it cannot simply disappear leaving a vacancy behind and then miraculously appear at the surface, as we assumed
in equilibrium thermodynamics, where it does not matter how a state is reached. |
|
 |
On the other hand, infinitely large perfect crystals do not exist - but semiconductor-grade
dislocation-free single Si crystals with diameters of 300 mm and beyond, and lengths of up to 1 m are
coming reasonably close. These crystals form a special case as far as point defects are concerned but nevertheless incorporate
point defects in equilibirum. |
 |
In real life we need other defects - surfaces,
crystal-melt interfaces, grain boundaries, dislocations, precipitates, and so on, as sources and sinks for point defects.
In regular metals or ceramics and so on, we have almost always plenty of those defects. |
 |
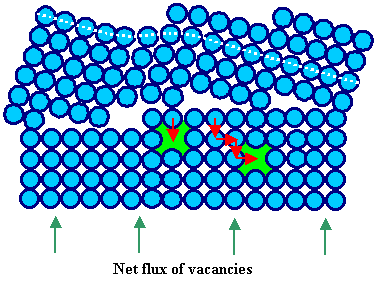
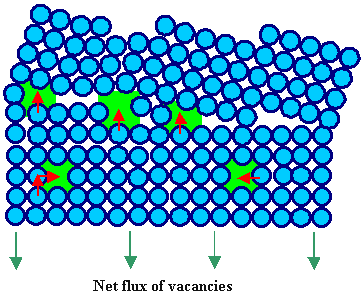
How a grain boundary
can act as source or sink for vacancies is schematically shown in the pictures below. |
|
 |
It is clear from these drawings that the activation energy (which is not
the formation energy of a vacancy!!) needed to emit (not to form from scratch!) a vacancy
from a grain boundary is small. |
|
|
 |
 |
Grain boundary absorbs 1 vacancy, i.e. acts as
sink after one more jump of the proper atom. |
Grain boundary emits 3 vacancies, i.e. acts as
source after one more jump of the 3 proper atoms. |
| The red arrows indicate the jumps of individual atoms. The flux of the vacancies is always
opposite to the flux of diffusing atoms. |
|
 |
We thus may expect that at sufficiently high temperatures (meaning temperatures
large enough to allow diffusion), we will be able to establish global point defect equilibrium
in a real (= non-ideal) crystal, but not really global crystal equilibrium, because
a crystal with dislocations and grain boundaries is never at global equilibrium. |
 |
Sources and sinks are a thus a necessary,
but not a sufficient ingredient for point defect equilibrium. We also must require that
the point defects are able to move, there must be some diffusion - or you must resign yourself to waiting for a long time.
In other words, we must look at the temperature now. |
|
 |
At low temperatures, when all diffusion effectively stops,
nothing goes anymore. Equilibrium is unreachable. For many practical cases however, this is of no consequence. At temperatures
where diffusion gets sluggish, the equilibrium concentration ceq is
so low, that you cannot measure it. For all practical purposes it surely doesn't matter if you really achieve, for example,
ceq = 10–14, or if you have non-equilibrium with the actual concentration c
a thousand times larger than ceq (i.e.
c = 10–11). For all practical purposes we have simply c = 0. |
|
 |
At high temperatures, when diffusion is fast, point defect
equilibrium will be established very quickly in all real crystals with enough sources and sinks. |
 |
The intermediate temperatures thus are of
interest. The mobility is not high enough to allow many point defects to reach convenient sinks, but not yet too small to
find other point defects. |
|
 |
In other words, the average diffusion length or mean
distance covered by a randomly diffusing point defect in the time interval considered, is smaller than the average distance
between sinks, but larger than the average distance between point defects. |
|
 |
This is important, so let's say it once more in yet other words: In the intermediate temperature
range we are considering here, a given vacany will still be able to move around sufficiently to encounter another vacancy,
but not a dislocation, precipitate or grain boundary. |
 |
Global point defect equilibrium as the best state of being is thus unattainable
at medium temperatures. Local equilibrium is
now the second best choice and far preferable to a huge supersaturation of single point defects slowly moving through the crystal in search of sinks.
|
|
 |
Local equilibrium then simply refers to the state with the smallest free enthalpy taking into account the restraints of the system. The most simple restraint is that the total
number of vacancies in vacancy clusters of all sizes (from a single vacancy to large "voids") is constant. This
acknowledges that vacancies cannot be annihilated at sinks under these conditions, but still are able to cluster. |
 |
Let us illustrate this with a relevant example. Consider vacancies in a metal
crystal that is cooled down after it has been formed by casting. |
|
 |
As the temperature decreases, global equilibrium demands that the vacancy concentration decreases
exponentially. As long as the vacancies are very mobile, this is possible by annihilation at internal sinks. |
|
 |
However, at somewhat lower temperatures, the vacancies are less mobile and have not enough
time to reach sinks like grain boundaries, but can still cover distances much larger than their average separation. This
means that divacancies, trivacancies and so on can still form - up to large clusters of vacancies, either in the shape of
a small hole or void, or, in a two-dimensional form, as small dislocation loops. Until they become completely immobile,
the vacancies will be able to cover a distance given by the diffusion length L (which depends, of course,
on how quickly we cool down). |
|
 |
In other words, at intermediate temperatures small vacancy clusters or agglomerates can be
formed. Their maximum size is given by the number of vacancies within a volume that is more or less given by L3
- more vacancies are simply not available for any one cluster. |
|
 |
Obviously, what we will get depends very much on the cooling rate and the mobility or diffusivity
of the vacancies. We will encounter that again; here is a link
looking a bit ahead to the situation where we cool down as fast as we can. |
 |
It remains to find out which mix of single vacancies and vacancy clusters will
have the smallest free enthalpy, assuming that the total number of vacancies - either single or in clusters - stays constant.
This minimum enthalpy for the specific restraint (number of vacancies = const.) and a given temperature then would be the
local equilibrium configuration of the system. |
 |
How do we calculate this? The simplest answer, once more, comes from using the
the mass-action law. We already used it for deriving the equilibrium concentration
of the divacancies. And we did not assume that the vacancy concentration was in global
thermal equilibrium! The mass action law is valid for any starting concentrations of
the ingredients - it simply describes the equilibrium concentrations for the set of reacting particles present. This corresponds
to what we called local equilibrium here. |
|
 |
The reaction equation from sub-chapter 2.2.1 was
1V + 1V Û V2 and in this case this is a valid equation for using the
mass action law. The result obtained for the concentration of divacancies with the single vacancy concentration in global thermal equilibrium was |
|
|
| c2V |
= (c1V)2 · |
z
2 | · exp |
DS2V
k |
· exp |
B2V
kT |
|
|
|
 |
Don't forget that concentrations here are defined as n/N, i.e. in relative units (e.g. c = 3,5 · 10–5) and not in absolute
units, e.g. c = 3,5 · 1015 cm–3. |
|
 |
For arbitrary clusters with n vacancies (1V + 1 V + ...
+ 1V Û Vn) we obtain in an analogous way for the concentration cnV
of clusters with n vacancies |
|
|
| cnV |
= (c1V)n · |
a | · exp |
DSnV
k |
· exp |
BnV
kT |
|
|
|
 |
with BnV = average binding energy between vacancies in an n-cluster,
c1V = const. concentration of the vacancies (and no longer the thermal equilibrium
concentration !), and a = number of possible "orientations" of the
n-cluster divided by the indistinguishable permutations. The value of a
will depend to some extent how we arrange n vacanies: in a row, on a plane or three-dimensionally - but we
won't worry about that because the other factors are far more important. |
|
The essential point now is to realize that these equations still work for local
equilibrium! They now describe the local equilibrium of vacancy clusters if a fixed concentration of vacancies is given. The situation now is totally
different from global equilibrium. If we consider divacancies for example, we have: |
| | |
|
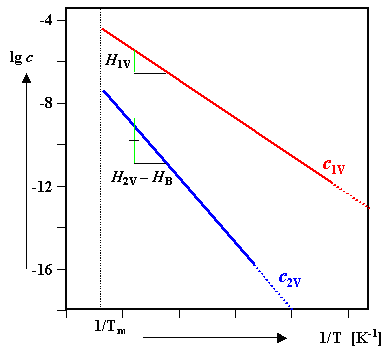
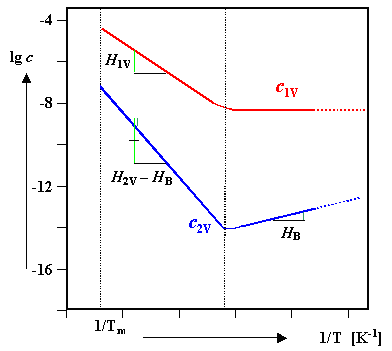
Global equilibrium |
|
Local equilibrium |
|
 |
c2V(eq) << c1V(eq) ; and c2V(eq) rapidly decreases with decreasing temperatures since c1V(eq) decreases. |
|
c2V is increasing
with decreasing T since c1V stays about constant,
but we still have the exp+BnV/ kT term that increases
with T |
| |
| |
|
 |
Whereas the concentration of clusters may still be small, they now contain most
of the vacancies. |
|
 |
Generally speaking, it is always energetically favorable, to put
the surplus vacancies in clusters instead of keeping them in solid solution if there is no possibility to annihilate them
completely. It thus comes as no surprise that in rapidly cooled down crystals with not to many defects that can act as sinks,
we will find some vacancy clusters at room temperature |
 |
It also should come as no surprise that the same is true for impurity atoms -
vacancy clusters. The equations governing this kind of point defect agglomeration are, after all, quite
similar to the equations discussed here. |
|
 |
If you now take the extreme case of a rather perfect Si single
crystal (no sinks for point defects), where we do not just have vacancies at thermal equilibrium, but also some relevant
concentration of interstitials, interstitial oxygen and substitutional carbon, you might well wonder what one will find at room temperature.
|
|
 |
Well - don't wonder! Get to work! It is not all that clear. And even
if that puzzle has been solved before you reach productive scientisthood, there is always GaAs, or InP, or
SiC, or - well, you will find something left to do, don't worry. |
| |
| |
© H. Föll (Defects - Script)