| For this basic module we simply take the suitable
module of the Hyperscript "Introduction to Materials Science II" since it is the newest and updated
version. | ||
| The module as it existed in Sept. '01 is reprocuced below. | ||
Ohm's Law and Classical Physics
| In this subchapter we will look at the classical treatment of the movement of electrons in an electrical field. It is a direct continuation of subchapter 1.1.3 in backbone II (of the hyperscript "Introduction to Materials Science II") and again more closely matched to the actual lecture. | ||
| In this preceding subchapter we obtained the most basic formulation of Ohms law in material
terms. s=q·n·m |
||
| For a homogeneous and isotropic material (e.g. polycrystalline metals or single crystal of cubic semiconductors), the concentration of carriers n and their mobility m do not depend on the coordinates - they have the same value everywhere in the material and the specific conductivity s is a scalar. | ||
| In general terms, we may have more than one kind of carriers (this is the common situation in semiconductors) and n and m could still be more or less complicated functions of the temperature T, the local field strength Eloc resulting from an applied external voltage, the detailed structure of the material (e.g. the defects in the lattice), and so on. | ||
| We will see that these complications are the essence of advanced electronic materials, especially the semiconductors, but in order to make life easy, we now will restrict ourselves to the special class of ohmic materials. We have seen before that this requires n and m to be independent of the local field strength. We still may have a temperature dependence of s ; even commercial ohmic resistors, after all, do show a more or less pronounced temperature dependence which increases roughly linearly with T. | ||
| In short, we are treating metals, characterized by a constant density of one kind of carriers (=electrons) in the order of 1 ...3 electrons per atom in the metal. | ||
Basic Equations and the Nature of the "Frictional Force"
| We consider the electrons in the metal to be "free", i.e. they can move unhindered in any direction. | ||
| The electrical field E then exerts a force F=-e· E on any given electron and thus accelerates the electrons in the field direction (more precisely, opposite to the field direction because the field vector points from + to - whereas the electron moves from - to +). | ||
| In the fly swarm analogy, the electrical field would correspond to a steady airflow - some wind - that moves the swarm about with constant drift velocity. | ||
| Basic mechanics yields for a single particle with momentum p F=dp/dt =m·dv/dt with p=momentum of the electron. Note that p does not have to be zero when the field is switched on. | ||
| If this would be all, the velocity of a given electron would acquire an ever increasing component in field direction and eventually approach infinity. This is obviously not possible, so we have to bring in a mechanism that destroys an unlimited increase in v | ||
| In classical mechanics this is done by introducing a frictional force Ffr=kfr·v with kfr being some friction constant. But this, while mathematically sufficient, is devoid of any physical meaning with regard to the moving electrons. So we have to look for another approach. | ||
| The best way to thing about it, is to assume that the electron, flying along with increasing velocity, will hit something else in its way every now and then, which will change its momentum (and thus the magnitude and the direction of v) as well as its kinetic energy 1/2·m·v 2. | ||
| In other words, we consider collisions with other particles where the total energy and momentum of the particles is preserved, but the individual particles loses its "memory" of its velocity before the collision and starts with a new momentum after every collision. | ||
| What are the "partners" for collisions of an electron, or put in standard language, what are the scattering mechanisms? There are several possibilities: | ||
| Other electrons . While this happens, it is not the important process in most cases. | ||
|
|
Defects, e.g. foreign atoms, point defects or dislocations. This is a more important scattering mechanism and moreover a mechanism where the electron can transfer its surplus energy (obtained through acceleration in the electrical field) to the lattice which means that the material heats up | |
| Phonons, i.e. "localized" lattice vibrations traveling through the crystal. In a quantum mechanical treatment of lattice vibrations it can be shown that these vibrations, which contain the thermal energy of the crystal, show typical properties of (quantum) particles: They have a momentum and an energy given by h·n (h =Plancks constant, n=frequency of the vibration), and treating the interaction of an electron with a lattice vibration as a collision with a phonon gives correct results. This is the most important scattering mechanism. | ||
| It would be far from the truth to assume that only accelerated electrons scatter; scattering happens all the time. If electrons are accelerated in an electrical field and thus gain energy, scattering is the way to transfer this surplus energy to the lattice which then will heat up. Generally, scattering is the mechanism to achieve thermal equilibrium and equidistribution of the energy of the crystal. | ||
| Lets look at some figures illustrating the scattering processes. | |||||
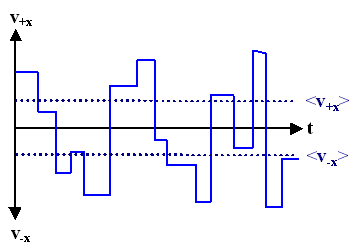 |
Shown here is the magnitude of the velocity of an electron in x and -x direction without an external field. The electron moves with constant velocity until it is scattered, then it continues with a new velocity. | ||||
| The scattering processes, though unpredictable as single events, must lead to the average <v> characteristic for the material and its conditions. | |||||
| Whereas <v>=0, <v> has a finite value and <vx>=- <v-x>. This is a bit tricky since the way we are writing formulas here we cannot easily distinguish between vectors and scalars: Here, to emphasize the point, v is a vector, and v is a scalar (its magnitude). | |||||
| From classical thermodynamics we know that the electron gas in thermal equilibrium with the environment possesses the energy Ekin=(1/2)kT per particle and degree of freedom with k=Boltzmann's constant and T=absolute temperature. | |||||
| We write energies E in magenta to avoid confuison with electrical fields E. | |||||
| The three degrees of freedom are the velocities in x-, y- and z-direction,
so we must have Ekin,x=1/2m<vx> 2=1/2 kT or <vx>=(kT/m)1/2. | |||||
| Similarly, for the total energy Ekin=1/2m<vx> 2 + 1/2m<vy>2 + 1/2m<vz>2=1/2m<v> 2=1/2mv02, we have E kin=1/2m<v>2=1/2mv02=3/2 kT with v0=<v>. | |||||
| Now lets turm on an electrical field E. It will accelerate the electrons between collisions; their velocity increases linearly. | |||||
| In our diagram from above this looks like this: | |||||
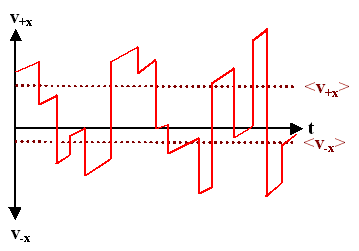 | Here we have an electrical field in x-direction. Between collisions, the electron gains velocity in +x-direction at a constant rate. | ||||
| The average velocity in +x directions, <v+x>, is now larger than in -x direction, <v-x>. For real electrons, however, the difference is very small; the drawing is very exaggerated. | |||||
| The drift velocity is contained in the difference <v+x> - <v-x>; it is completely described by the velocity gain between collisions. We may thus symbolically neglect the velocity right after a collision because it averages to zero anyway, and just plot the velocity gain in a simplified picture; always starting from zero after a collision. | |||||
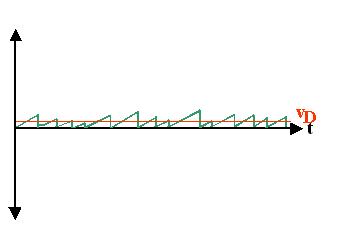 | The picture now looks quite simple; but remember that it contains some not
so simple averaging. | ||||
| At this point it is worthwhile to point out that we can define a new average: The mean time between collisions, or more conventional, the mean time t for reaching the drift velocity vD in the simplified diagram. | |||||
| This is most easily seen by simplifying the scattering diagram once more: We simply use just one time - the average - for the time that elapses between scattering events and obtain. | |||||
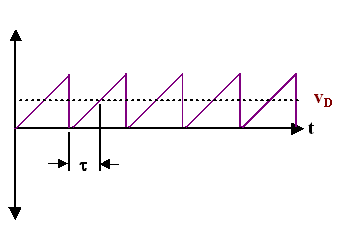 |
This is the standard diagram illustrating the scattering of electrons in a crystal usually found in text books; the definition of the scattering time t is included | ||||
| While this diagram is not wrong, it is a highly abstract rendering of the underlying processes after several averaging procedures. From this diagram only, no conclusion whatsoever can be drawn as to the average velocities of the electrons without the electrical field! | |||||
| With the scattering concept, we now can introduce two new (related) material parameters: | ||
| The mean (scattering) time t between two collisions as defined before, and a directly related quantity: | ||
| The mean free path
L between collisions; i.e. the distance travelled by an electron (on average) before it collides with something
else and changes its momentum. We have L=2t·(v 0 + vD). Note that v0 enters the equation! | ||
| Using t as a new parameter, we can rewrite the mechanics equations: | ||
| dv/dt
can be written as Dv/Dt=vD /t because the velocity change during the time t is just vD. From this we obtain | ||
| vD/t=-E·e/m , or vD=-(E·e·t)/m. | ||
| Inserting this equation for vD in the old definition of the current density , j=-n·e·vD yields | ||
| j=-E·( n·e2t )/m :=s·E
, and thus | ||
| s=(n·e2t )/m | ||
| This is the classical formula for the conductivity of a classical "electron gas" material; i.e. metals. We do not yet know tclass, but we may turm the equation around and use it to calculate the order of magnitude of t class , since we know the order of magnitude for the conductivity of metals. The result is: | ||
|
|
tclass=ca. (10-13 .... 10-15) sec | |
| "Obviously" (as stated in many text books), this is a value that is far too small and thus the classical approach must be wrong. But is it really too small? How can you tell without knowing a lot more about electrons in metals? Well, you can't. So let's look at the mean free path L instead. | ||
| We have t =L/(2(v0 + vD)) or L=2t(v 0 + vD) and v02=(3kT/m) 1/2 . | ||
| This gives us a value v0=ca. 105 m/s at room temperature! Now we
need vD, and this we can estimate from the equation vD/ t=-E·e/m
given above: vD =- E·t·e/m=ca. 1 mm/sec if we use the value for t dictated by the measured conductivities. It is much smaller than v0! | ||
|
|
The mean free path between collisions thus is L=2t (v0 + vD)=ca. 2 tv0=ca. (101..10-1)nm and this is certainly too small!. | |
| Why is a mean free path in the order of the size of an atom too small? Well, think about the scattering mechanisms. The distance between defects is certainly much larger, and a phonon itself is "larger", too. Moreover, consider what happens at temperatures below room temperatures: L would become even smaller - somehow this makes no sense. | ||
| It does not pay to spend more time on this. Whichever way you look at it, whatever tricky devices you introduce to make the approximations better (and physicists have tried very hard!), you will not be able to solve the problem: The mean free paths are never even coming close to what they need to be and the conclusion - maybe reluctant but unavoidable - must be: | ||
| There is no way to describe conductivity (in metals) with classical physics. |
![]() 2.2.1 Intrinsic Properties in Equilibrium
2.2.1 Intrinsic Properties in Equilibrium
![]() 2.2.2 Doping and Carrier Density
2.2.2 Doping and Carrier Density
![]() 2.2.3 Life Time and Diffusion Length
2.2.3 Life Time and Diffusion Length
![]() 5.3.2 Isotype Junctions, Modulation Doping, and Quantum Effects
5.3.2 Isotype Junctions, Modulation Doping, and Quantum Effects
![]() 8.1.2 Basic Time Consuming Processes
8.1.2 Basic Time Consuming Processes
© H. Föll (Semiconductors - Script)