|
|
| |
A Few General Points |
 |
A solar cell is a device that absorbs light and converts the energy contained
in the light into electrical energy. In order to understand what it is all about, we first need to look at a few numbers.
I will give only rough numbers, as far as possible in the "1" system because
that is good enough to get the basic messages across. Let's ask the following questions:
- How much energy is contained in sun light?
- How much electrical energy do I, personally, need?
- How large is the conversion efficiency of solar cells?
- How many square meters of solar cells do I, personally, need to cover all my electricity needs?
- How much does that cost? Can I afford it?
- How about the times when the sun is not shining?
|
|
 |
Note that at this point we don't need to know how
a solar cell works. Let's go through these points one by one. |
 |
1. How much energy
is contained in sun light?
On this planet the maximum amount of sun light can be had at the equator on a cloudless day. It is - roughly - 1
kW/m2 or 1000 Watts per square meter.
At dawn and in the evening you get less, and at night you
get nothing. The yearly average on that cloudless place at the equator is roughly
200 W/m2. A year (abbreviation: a) has 1 a = 365 · 24 = 8 760 hours (h), so the energy produced
per square meter in one year is 200 W/m2 · 8 760 h = 1 760 000 Wh/m2 = 1 760 kWh/m2
or roughly 2 000 kW/m2.
Where I live, (at a latitude north of Maine) there is far less
sun, a lot of clouds, it's cold, and it rains a lot. So let's say my yearly average is 100 W/m2 or about
1 000 kW/m2
My deplorable lack of sun and surplus of rain and snow is not quite as bad as it appears.
Solar cells work best if they are cold, they encounter serious problems if it is really hot! Rain does decrease light intensities
- but also keeps the surface of solar cells clean and thus has also positive effects. Solar cells covered with leaves, dust,
or bird shit will not produce as much electricity as clean ones |
|
 |
Watt, as you most certainly know, is a unit for power or energy per time. 1 W equals
1 J/s (Joule per second), and a Joule is the basic measure of energy. Now let's
look at power sources you can relate to. If you go full blast on a bicycle, you produce
around 200 W - if you are good. If you go more leisurely and take breaks for sleeping, eating and going to the bath
room, your daily average may be around 50 W. That we can take as the power produced by a typical hard-driven slave,
the ubiquitous and relatively smart power source treasured by most of the great cultures in history.
Looking at power usage, a typical (USA) light bulb consumes 100 W. You need two slaves to keep one
burning. Typical household appliances run from 50 W (small TV) up to a few kW (range, dryer). 746 W, by the
way, equals 1 horsepower. Horses can work harder than slaves, indeed, but are not quite as useful in jobs like digging for
ore, forging swords or sharing beds with. |
|
 |
Just for the hell of it, I give you some precise data now: |
| |
| |
| |
 |
| Average solar energy per year in Germany. |
|
| |
| |
|
 |
Those are measured numbers for Germany. I'm living in
Kiel (black circle, all the way up), so 1 000 kW/m2 is not too bad. The Bavarians down south have a bit
more sun and thus harvest a bit more solar energy. |
 |
2. How much electrical energy do I, personally, need?
I, personally, use electrical energy in two ways. And you
do too. First, I use it at the place where I live, and for that I pay some utility directly.
Second, I use it where I work and hang out, and in the products that I buy. My office
features lights and other electrical devices, and so does the opera theater where I hang out. Making and transporting the
beer I drink needs some electricity, and so on. For all this electrical energy I pay indirectly
through taxes and prices for products and services. |
|
 |
The relation between my direct and indirect electrical energy needs is - roughly - 50:
50, and the total per capita comes to about 5 000 kWh/a or, expressed in
power, 5 000 kWh/8 760 h = 570 W. Double or treble that if you are an American citizen. |
 |
3. How large is the conversion efficiency of solar cells?
An efficiency is simply the relation (in percent) between what comes out relative to what goes in. We know now that up to
1 000 W/m2 light power goes into a 1 m2 solar panel. How much electrical power comes
out?
Good mass-produced solar cells that are on the market now (end of 2012) have a conversion efficiency around 20
%. That is an excellent value, by the way. It means that from my yearly average of 100 W/m2
sun light energy, 20 W/m2 will make it to electrical energy. |
 |
4. How many square meters of solar cells do I, personally,
need to cover all my electricity needs?
The average energy produced in one year per square meter thus is 20 W
· (365 · 24)h = 175.2 kWh/m2. All my direct electrical energy
needs (2 500 Wh) thus can be met by about 15 m2 of solar cells on my roof top; my total
needs, including the indirect use of electricity, demand 30 m2. Americans, of course, need more. Nevertheless: |
| |
| |
| |
Surprise! This is probably far less than you would have guessed.
|
|
| |
| |
 |
5. How much does that cost? Can I afford it?
Presently (May 2013) you pay about 100 $ for 1 m2 modules; or 3 000 $ for the 30
m2 you need. You also must pay for fixing the modules on your roof, hooking up everything, and so on. So
let's say you end up with 8 000 $ - 10 000 $ now and less in the near future. You also need a contract with your
local utility where they pay you when you produce more than you need and bill you when you use more than you produce. Your
utility hates doing this, so you need a government that sees to this part. If done right, income and costs cancel each other |
|
 |
In other words: You will have all the electricity you need and no more electricity bills for
the next 20 - 40 years since (good) modules last about forever. You figure if that is attractive or not. |
 |
6. How about the times when the sun is not shining?
It's dark, man! When the sun is not shining, your solar cells are not producing. There is no choice, you must now get your
electrical energy from other sources.
You might, for example, store enough electrical energy for a few days in some
batteries in your basement. Many companies at present offer suitable batteries but prices are still on the high side. |
|
 |
The smart thing to do, of course, is to create a new nation-wide infrastructure that includes
different ways to produce and store (green) electricity. Besides solar cells you must have wind mills, water power (including
pumped-storage hydropower plants) other storage facilities like compressed air in large cavities, gas turbines, and a "smart"
net. The gas turbines will run on the gas produced whenever there is some surplus wind and solar energy and thus produce
"green" energy. You Americans must also get at least semi-intelligent about energy usage.
Doing this is neither
too difficult nor too expensive. It is just a lot that needs to be done and thus needs
a large amount of money (though less than the average senseless war the USA keeps loosing
on a regular base). Note that a large amount of money needed for something is not the same as something being expensive.
Since the new power structure is an investment for a very long time span, it is not attractive for private enterprise. There
were good reasons why large infrastructures concerning, e.g., transport (street system), electrical power, or communication
(mail and telephone) were run by the state in the good old times. |
 |
The message, I hope is clear. |
| | | |
| |
|
Green electric energy at reasonable costs is not a technical problem anymore. It just needs doing.
|
|
| |
| |
| |
The Working of a Solar Cell |
 |
You may know that a solar cell is just a simple pn-junction or diode, as far as
semiconductor physics is concerned. Well, yes, but physics is never about money. As far as mass-produced cheap but extremely
good solar cells are concerned, they are extremely complex devices - and I'm not going to discuss that here. I'm only going
to discuss a few simple but decisive features of solar cells. |
|
 |
The figure below shows the essentials of a thin-film solar cell (left) and a standard silicon
(Si) solar cell (right). Let's see what we can learn from this |
| | |
|
|
|
 | | Essentials of thin film and Si solar cell |
|
| |
| |
|
 |
The crucial pn-junction in both cases is between the orange and yellow material. |
 |
First, you need a semiconductor material since only
semiconductors can convert light energy into electrical energy. There are a lot of semiconductor materials out there, and
some are better at the job than others. No single material, however, can convert more then - roughly - 30 % of the light
energy into electrical energy for very fundamental reasons. Combining a bunch of different semiconductors can increase this
efficiency to 50 % or beyond - but not cheaply! |
|
 |
The material of choice, however, is only performing well if a lot of other requirements are
met. Most semiconductors will not do that, no matter how much money you are willing to spend. At present only a few are
left that can be made to perform satisfactorily (though never as well as theoretically possible) and
are cheap. In essence we have crystalline silicon (Si), amorphous silicon, cadmium telluride (CdTe) and the "CIGS"
family (copper (Cu) indium (In) gallium (Ga) sulfur (S) or selenide (Se) alloys). Cadmium and selenide are very toxic, gallium,
indium and crystalline silicon are expensive. |
|
 |
The major difference between crystalline Si and the rest (including all variants of amorphous
Si) is how thick the solar cell has to be to absorb all the incoming light. Crystalline silicon needs a thickness of around
20 µm; all others can do with just 1 µm or even less. That means that you can make a "thin film" solar
cell from all contenders except crystalline silicon. That is great because you do not need a lot of expensive material if
you only need a thin film. So why do we have mostly (around 85 %) crystalline Si solar cells out there?
Well, the first law of economics applies. There is
a price to pay. First, thin film solar cells so far are just not as good as solar cells made from crystalline silicon. You
need to cover a larger area with thin film solar cells to harvest the same amount of electrical energy in comparison to
crystalline silicon stuff, and that sets off the advantage of lower prices to some extent. |
| |
|
|
|
 |
| Poly crystalline silicon wafer and solar cell made from it |
|
| |
| |
|
 |
The dark blue color comes from the antireflection layer. The visible lines are the (silver-rich)
metal contacts. |
 |
Next, a solar cell needs to be mechanically stable and that means that it needs
a certain minimum thickness of at least - roughly - 150 µm. Otherwise the machines in your fully automated factory
just can't handle the stuff without breaking it. If you go for crystalline silicon solar cells, you just make them 150 µm
thick and you have no problem except that you are wasting a large part of your (relatively expensive) silicon just for supporting
the active part. |
|
 |
A thin film solar cell needs to be deposited on a thick substrate. You want cheap stuff that
lasts for at least 20 years out there in sun, rain and snow and is compatible with your production requirements. For example,
it must be capable of taking the heat; the high temperatures necessary for depositing all these layers.
Glass is ideal
but not conducting, forcing you to first putting down a metal layer to supply the necessary electrical contact to the backside
of the solar cell. On silicon this is much easier and - big advantage! - you can make the metal layer rather thick and deposit
it with simple methods. It's electrical resistance then will be much lower than in the thin film case, and that is important! |
 |
A solar cell needs a junction between two semiconductors that are of different
conduction type called n (negative) type and p (positive) type. In silicon this is easy to do by a process called doping,
producing n-Si and p-Si. The junction between the two is well understood and rather good for solar cell purposes. |
|
 |
Thin film solar cells need to make the junction by depositing two different materials. That's
not difficult to do but the junctions formed are (so far) not quite as good as the p-typ / n-type Si junctions, and that
is one of the reasons why (mass-produced) thin film solar cells have typical efficiencies around 10 % - 12 % while (mass-produced)
Si solar cells approach 20 %. In the laboratory we can do better - but not (yet) with cheap processes / materials. |
 |
Now you need to contact the front side. Wherever you put down the contact metal,
light will not reach the solar cell below. Obviously, you want to put a system of thin wires down or a transparent conductor.
Once more, metal deposition is easily done on crystalline silicon, and the resistance of the wires is much smaller than
in the case of thin film solar cells where transparent (and rather bad) conductors must be used. |
|
 |
The resistance of the metallization is a serious problem in both cases. For crystalline silicon,
however, we can get away with the simple metal structure as shown above, for thin film solar cells we cannot.
We need
to add some complex additional processes that are not shown in the figure above. You might safely guess, however, that they
cost money. The picture below gives an idea what I'm talking about. |
| | |
|
|
|
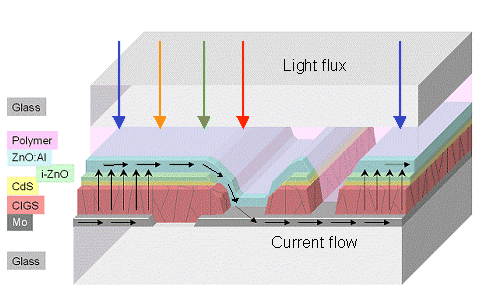 |
| Cross sections through a CIGS kind of solar cell |
| Source: Courtesy of Dr. Powalla; ZWS Stuttgart, © Powalla / ZWS Stuttgart |
|
| |
| |
|
 |
Shown is the complex structure that needs to be introduced about every cm, where single cells
are switched in series in order to overcome the resistance problem. CIGS and CdS (cadmium sulfide) are the power generating
semiconductors, the ZnO ("Intrinsic" zinc oxide) and the ZnO:Al (Al doped ZnO) supply the transparent front side
contact. The backside contact needs molybdenum (Mo). |
 |
Finally we need to put down an anti-reflection (AR) coating since otherwise too
much light will be reflected at the surface of the solar cell. This is standard technology for both kinds of solar cells. |
 |
It only remains to put the solar cells into a sealed frame with an area of about
1 m2; this we call a "module" and that's what you see on roof tops. Now the thin film solar cells have
a big advantage: The are as large as the module since they can be fabricated on large area substrates. |
|
 |
Crystalline Si solar cells, in contrast, are comparatively small (about (12 x 12) cm2)
,and you need to put down and solder together about 100 of them to make a module. That takes some money, of course. |
 |
What can I predict for the future? Well, there are two things I feel rather certain
about:
- Solar energy production will be so cheap that it is going to be big business for many years to come.
- Crystalline silicon will win the competition, at least for the foreseeable future. The reason is that there si a big
potential that suitable silicon can be produced far more cheaply than today, in humongous quantities. All we need to do
is to follow the example of iron and steel, where a similar development as the one I'm predicting here has taken place more
than 100 years ago.
|
| |
| |
© H. Föll (Iron, Steel and Swords script)



