|
Melt Spinning of Metals |
|
|
| |
|
 |
Melt spinning is one way to make amorphous metals
or "metglas". It always produces thin (around 50 µm) and very long (many m)
ribbons (some mm - several cm in width) of the stuff. In essence, the metal retains more or less its liquid and thus amorphous
structure because it is cooled down with extreme speed - around 1.000.000 °C/s
(1.800.000 °F/s) - making atom movement in the solid well nigh impossible.
The figure shows schematically
how it is done: |
| |
| |
| |
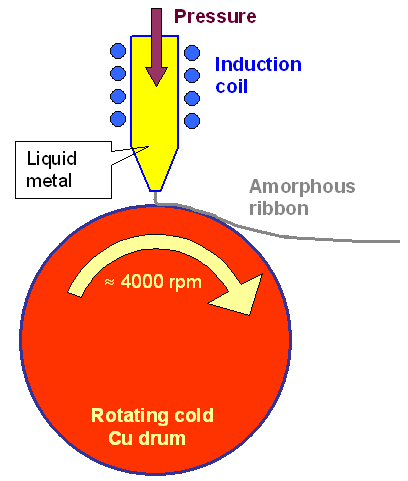 | | Melt spinning principle |
|
| |
| |
 |
Melt spinning has been employed for more than 30 years and can be used for all
kind of metals and alloys. The product may not be fully amorphous but "only" nanocrystalline. Neverthelss, it
has always properties quite different from its normal crystalline form.
Metglas is, of course, a metastable phase that
will change into a crystalline one if enough energy (= temperature) is provided. It only comes as thin ribbon or foil, limiting applications.
|
|
 |
More recently certain alloys, made e.g. by properly mixing lanthanum, magnesium,
zirconium, palladium, iron, copper, and titanium, were found to stay amorphous even as "bulk" material. "Bulk
metallic glasses" are now an active research area in Materials Science and Engineering but have yet to await large
scale use.
So melt spinning is it if you want to make quantities of the stuff at reasonable prices. Here is what a real
(R&D) machine looks like: |
|
| |
| |
 | | Melt spinning apparatus |
| Source: This is a machine operated at the Fraunhofer Institute for Manufacturing Technology
and Advanced Materials, in Dresden, Germany. Thanks for the picture! |
|
| |
| |
 |
Why would you want to make quantities of amorphous metals? Because they have some
remarkable properties, in particular as far as magnetism is
concerned |
|
 |
Ferromagnetic behavior of metglas can be much better than that of its crystalline brethren
(usually iron for large scale uses like transformer cores). In particular, metglas is more energy efficient, i.e. dissipates
much less electrical energy into heat compared to regular iron. China, in particular, has started to install amorphous metal
transformers in a number of energy intensive provinces since 2005; the expected savings in terms of energy, money and reduced
CO2 emissions are enormous. |
|
 |
Thin foils are used for brazing.
It is not so easy to flow a liquid material into a large area joint to be brazed; it's easier to put a thin foil in between
the two metals that melts at convenient temperatures. Desirable materials often cannot be easily produced in the crystalline
structure or might be brittle (and thus hard to use). Metglas may do the job much better, more easily and cheaper. |
|
 |
.Many more applications are emerging but not (yet) making serious money. |
| |
| |
© H. Föll (Iron, Steel and Swords script)

