|
When you heat up food with a microwave oven, all you do
is to jiggle the water molecules with a frequency where the dielectric losses
are large. |
|
 |
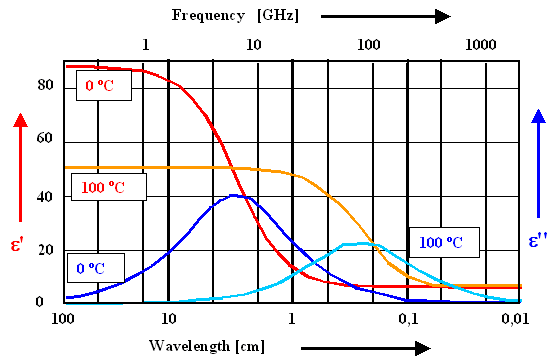
Lets look at the dielectric function of water: |
| |
|
 |
First, we see that the general graph of the frequency dependence is exactly as we would expect
from the theory. |
|
 |
There is a noticeable change of e' and e''
with the temperature. This is as it should be, we had a temperature dependence for the polarization
. |
| |
|
|
 |
Changing the temperature from about 300 K to 400 K thus should reduce '(300 K) »
80 by about ¾; i.e. to e'(400 K) = 60. The observed reduction is somewhat
more severe, because we did not take into account that the water dipoles interact to some extent; and at low temperature
this interaction is stronger than at high temperatures. |
 |
The maximum of the dielectric losses occurs roughly between 5 GHz - 100 GHz., i.e. in
the microwave region of the spectrum |
|
 |
Most microwave ovens work at 2.45 GHz, well below the region of maximum losses. This is intentional
to ensure that the radiation is not totally absorbed by the first layer of water it encounters but may penetrate further
into the foodstuff, heating it more evenly. |
|
 |
Radiation passing through the food items is mostly reflected back, due to the design of the microwave oven,
and absorbed on later passes. |
 |
If the water is frozen, i.e. you have ice, you have problems. The DK of ice at the microwave
frequency is only about 3, and little energy is absorbed |
 |
If your water is salty, the DK goes down, too. On the other hand, the Na+
and Cl ions are jiggled by the electrical field; too (without producing dipoles and thus a DK),
producing frictional heat and increasing the heating rate. |
|
 |
Thus whereas ice is a very poor microwave absorber, and water becomes a poorer microwave absorber with
rising temperature, a lossy salty food such as salt meat becomes a better microwave absorber with rising temperature. |
|
 |
This is particularly noticed on thawing. It may take forever to thaw your frozen butter or steak, but after
it happened it only takes a short time to turn it liquid or mushy, respectively |
|
| |
© H. Föll (Electronic Materials - Script)